Etiology
Acute pancreatitis is an acute inflammatory disorder of the pancreas that has numerous causes ( Box 48-1 ). The most common risk factors are chronic alcohol consumption and choledocholithiasis. In 20% of cases no cause can be found.
- •
Gallstones (45%)
- •
Alcohol (35%)
- •
Others (10%)
- •
Medications
- •
Hypercalcemia
- •
Hypertriglyceridemia
- •
Duct obstruction (e.g., tumor)
- •
Post–endoscopic retrograde cholangiopancreatography
- •
Hereditary
- •
Trauma
- •
Viral
- •
Post cardiac bypass
- •
Idiopathic (10% to 20%)
Prevalence and Epidemiology
In the United States, up to 210,000 patients per year are admitted to a hospital for acute pancreatitis. The spectrum of acute pancreatitis ranges from mild to severe and fatal.
In 1992, the International Symposium on Acute Pancreatitis in Atlanta, Georgia, established a clinical-based classification and defined certain terminologies commonly associated with acute pancreatitis. Acute pancreatitis is classified as mild and severe based on the presence of local complications and organ failure. This classification helped identify patients with severe disease who required close monitoring and intensive unit care. Mild acute pancreatitis has a mortality rate of less than 1%, whereas the death rate for severe pancreatitis is much higher—10% with sterile and 30% with infected pancreatic necrosis.
Prevalence and Epidemiology
In the United States, up to 210,000 patients per year are admitted to a hospital for acute pancreatitis. The spectrum of acute pancreatitis ranges from mild to severe and fatal.
In 1992, the International Symposium on Acute Pancreatitis in Atlanta, Georgia, established a clinical-based classification and defined certain terminologies commonly associated with acute pancreatitis. Acute pancreatitis is classified as mild and severe based on the presence of local complications and organ failure. This classification helped identify patients with severe disease who required close monitoring and intensive unit care. Mild acute pancreatitis has a mortality rate of less than 1%, whereas the death rate for severe pancreatitis is much higher—10% with sterile and 30% with infected pancreatic necrosis.
Clinical Presentation
The hallmark symptom of acute pancreatitis is the acute onset of persistent upper abdominal pain, usually with nausea and vomiting. The pain may radiate to the back, chest, flanks, and lower abdomen. Physical examination findings include fever, hypotension, severe abdominal tenderness, guarding, respiratory distress, and abdominal distention.
Pathophysiology
The inflammatory process in acute pancreatitis is triggered by the premature activation of pancreatic enzymes with resultant autodigestion of the pancreatic parenchyma. The inflammatory process may remain localized to the pancreas, spread to regional tissues, or even involve remote organ systems, resulting in multiple organ failure and occasionally death.
Mild acute pancreatitis (also known as interstitial or edematous pancreatitis) is more common and is a self-limiting disease with minimal organ dysfunction and an uneventful recovery. Pathologically, the mild form of acute pancreatitis is characterized by interstitial edema and infrequently by microscopic areas of parenchymal necrosis.
Severe acute pancreatitis (also known as necrotizing pancreatitis) occurs in 20% to 30% of all patients and is associated with organ failure and/or local complications, such as necrosis, abscess, or pseudocyst formation.
Imaging
Computed tomography (CT) and abdominal ultrasonography are routinely used in the setting of an acute abdomen to identify the source of pain. CT can help confirm the diagnosis of acute pancreatitis and exclude other causes of acute abdomen such as gastrointestinal perforation, acute cholecystitis, acute aortic dissection, and mesenteric artery occlusion, which can clinically mimic acute pancreatitis. In established cases of acute pancreatitis, contrast-enhanced CT is considered the gold standard for evaluating morphologic changes of acute pancreatitis, particularly in the assessment of pancreatic necrosis. Magnetic resonance imaging (MRI) with MR cholangiopancreatography (MRCP), endoscopic retrograde cholangiopancreatography (ERCP), endoscopic ultrasonography, and angiography have specific indications in a patient with known acute pancreatitis.
Radiography
Plain abdominal radiographs are often normal in patients with acute pancreatitis. Air in the duodenal C-loop, a “sentinel loop” (focally dilated jejunal loop in the left upper quadrant) or the “colon cutoff” sign (distention of the colon to the transverse colon with a paucity of gas distal to the splenic flexure) may be seen on plain radiographs in patients with pancreatitis. However, these findings are never sufficiently enough to confirm the diagnosis.
Computed Tomography
Role of Computed Tomography in Acute Pancreatitis
CT can establish the diagnosis of acute pancreatitis. It helps determine the underlying cause of acute pancreatitis (identifies choledocholithiasis and biliary ductal dilatation associated with biliary pancreatitis). It grades the severity of the disease and detects complications such as pancreatic necrosis, abscess, or pseudocysts.
Optimal Time to Perform Computed Tomography
In established cases of severe acute pancreatitis, contrast-enhanced CT helps in grading the severity of the disease and determining the extent of necrosis. Because necrotic areas of pancreatic parenchyma become better defined 2 to 3 days after the onset of symptoms, contrast-enhanced CT performed 48 to 72 hours after the onset of an acute attack gives more reliable information. CT findings can be equivocal if the scan is obtained during the initial 12 hours.
Scanning Technique
Multidetector CT scanners allow high-resolution, multiphase imaging of the pancreas performed using short scanning times. Table 48-1 depicts the CT scanning parameters used in our institution.
| Parameters | Vascular Phase (40-45 sec) | Portal Venous Phase (65-70 sec) |
|---|---|---|
| Intravenous contrast | 4-5 cm 3 /s | 3 cm 3 /s |
| Range | Celiac through entire pancreas | Dome of diaphragm to symphysis pubis |
| Automated bolus tracking | + (150-HU threshold in the aorta at the level of celiac artery and 8- to 10-sec diagnostic delay) | + (55-HU threshold in the right lobe of liver at the level of the right portal vein) |
| Slice thickness | 1-3 mm | 5 mm |
| Spacing | 1-3 mm | 5 mm |
| Kilovoltage peak | 120-140 | 120-140 |
| Milliamperage | 240-280 | 240-280 |
| Time | 0.5-0.8 sec | 0.5-0.8 sec |
| Field of view | 28 | Based on size of patient |
A single-phase contrast-enhanced CT in the portal venous phase (~70 seconds after the injection of intravenous contrast) is usually adequate to make the diagnosis and assess the complications of acute pancreatitis. Normal pancreatic tissue should demonstrate a homogeneous increase in attenuation to 100 to 150 Hounsfield units (HU) after contrast administration. Lack of enhancement or minimal enhancement (<30 HU) of a portion of the pancreas or the entire pancreas indicates decreased blood perfusion and necrosis. In the absence of pancreatic necrosis, the pancreas and spleen should be similar in attenuation on the portal venous phase.
When vascular complications are suspected, an additional vascular phase can be added to the imaging protocol, which is performed approximately 45 seconds after the injection of the contrast agent. Automated bolus tracking can be used to determine optimal timing of image acquisition. Unenhanced CT may be added to the imaging protocol if there is a strong clinical suspicion of hemorrhage.
Imaging Findings
As mentioned earlier, CT is not required in mild, self-limiting cases of acute pancreatitis. In these cases, CT of the pancreas may be normal or the pancreas may be enlarged or low in attenuation, indicating interstitial edema ( Figure 48-1, A ). The role of CT is primarily in patients with severe acute pancreatitis to assess the degree of pancreatic necrosis and detect complications such as pseudocyst and abscess formation.
The definitions, pathogenesis, and imaging appearance of the different types of fluid collections seen in acute pancreatitis are discussed in this section. These terminologies were standardized by the 1992 International Symposium on Acute Pancreatitis and should be included in the radiology reports.
Acute Fluid Collections.
Acute fluid collections occur early in the course of acute pancreatitis (within 48 hours). These consist of enzyme-rich pancreatic juices and lack a wall of fibrous or granulation tissue. These usually occur in or near the pancreas and may dissect in the lesser sac, anterior pararenal spaces (commonly on the left), transverse mesocolon, and mesenteric root (see Figure 48-1, B ). Acute fluid collections are seen in 30% to 50% of cases and resolve spontaneously in approximately 50% of patients. In the remainder, they can get walled off and progress to become pseudocysts or abscesses.
Acute Pseudocyst.
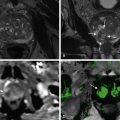
A pseudocyst is defined as a collection of pancreatic juices enclosed by a wall of fibrous or granulation tissue. Pseudocysts are formed approximately 4 weeks after the onset of acute pancreatitis. On CT, a pseudocyst appears as a well-circumscribed, low-attenuation collection commonly occurring in the vicinity of the pancreas ( Figure 48-2 ). On rare occasions they can be seen in unusual locations such as the mediastinum and groin ( Figure 48-3 ). A typical pseudocyst is sterile. If pus is present in the pseudocyst, the lesion is termed a pancreatic abscess.
Pancreatic Abscess.
A pancreatic abscess consists of a circumscribed intra-abdominal collection of pus, usually in proximity to the pancreas, and contains little or no pancreatic necrosis. Like pseudocysts, pancreatic abscesses occur later in the course of severe acute pancreatitis, often 4 weeks or more after the onset. On contrast-enhanced CT, the presence of a thick, irregular wall helps differentiate a pancreatic abscess from a pseudocyst, which generally has a thin, well-delineated wall ( Figure 48-4 ).
Pancreatic Necrosis.
Pancreatic necrosis is defined as focal or diffuse areas of nonviable pancreatic parenchyma and is typically associated with peripancreatic fat necrosis. On contrast-enhanced CT, pancreatic necrosis appears as one or more focal areas of nonenhancing pancreatic parenchyma ( Figure 48-5 ). Pancreatic necrosis is often hemorrhagic because of leakage from small veins and is seen on CT as areas of increased attenuation within the pancreas ( Figure 48-6 ). Retroperitoneal fat necrosis is invariably seen in patients with pancreatic necrosis, but the converse is not true. CT cannot reliably diagnose retroperitoneal fat necrosis, and thus all heterogeneous peripancreatic collections should be considered as areas of fat necrosis until proved otherwise ( Figure 48-7 ).
In individuals with fatty infiltration of the pancreas and in those with edematous or interstitial pancreatitis, the decreased enhancement of the pancreas should not be mistaken for pancreatic necrosis. Also, small, focal intrapancreatic fluid collections that are sometimes seen in acute pancreatitis should not be mistaken for focal necrosis. This distinction can be difficult in the absence of prior imaging.
Imaging Differentiation of Sterile Versus Infected Necrosis
The differentiation of sterile from infected pancreatic necrosis is important from the management perspective because the latter necessitates necrosectomy. The mortality rates increase from 10% in those with sterile necrosis to 30% in the presence of infected necrosis. Distinguishing sterile from infected necrosis based solely on imaging is virtually impossible, the only exception being the presence of gas bubbles within the collection suggesting the presence of infection ( Figure 48-8 ). Percutaneous needle aspiration under CT or ultrasound guidance is helpful in detecting the presence of infection.
Computed Tomography Severity Index
Balthazar and colleagues graded the severity of acute pancreatitis based on CT findings and described the term CT severity index (CTSI). CT grading of acute pancreatitis is depicted in Table 48-2 . These investigators reported 0% and 48% mortality and morbidity rates, respectively, in patients who had less than 30% necrosis on CT. Larger areas of necrosis (30% to 50% and >50%) were associated with a morbidity rate of 75% to 100% and a mortality rate of 11% to 25%. Additionally, patients with a CTSI of 0 to 3 showed a 3% mortality rate and an 8% morbidity rate, whereas in patients with a CTSI of 7 to 10, the mortality and morbidity rates were 17% and 92%, respectively.
| Grade | Description |
|---|---|
| A | Normal-appearing pancreas |
| B | Focal or diffuse enlargement of the pancreas |
| C | Pancreatic gland abnormalities accompanied by mild peripancreatic inflammatory changes |
| D | Fluid collection in a single location |
| E | Two or more fluid collections near the pancreas or gas in or adjacent to the pancreas |
| CT Grade | Score | Necrosis (%) | Score |
|---|---|---|---|
| A | 0 | 0 | 0 |
| B | 1 | <30 | 2 |
| C | 2 | 30-50 | 4 |
| D | 3 | >50 | 6 |
| E | 4 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree