Etiology
Chronic pancreatitis is defined as an ongoing prolonged inflammatory disease characterized by progressive irreversible structural changes resulting in permanent loss of endocrine and exocrine function. The Cambridge classification of 1983 acknowledged that chronic pancreatitis is typically associated with abdominal pain but occasionally can be painless and may recur. According to the revised pancreatic classification of pancreatitis from the Marseille symposium of 1984, acute and chronic pancreatitis are very different diseases and acute pancreatitis only rarely leads to chronic pancreatitis.
Prolonged alcohol abuse is the most common cause (70%) of chronic pancreatitis in the United States. Other causes are familial occurrence with hyperlipidemia, hyperparathyroidism, cystic fibrosis, trauma, cholelithiasis, and pancreas divisum. The familial form of pancreatitis, hereditary pancreatitis, is thought to be inherited in an autosomal dominant fashion with variable penetrance.
Of patients with chronic pancreatitis, 30% to 40% have no apparent underlying cause and are considered to have idiopathic chronic pancreatitis.
Patients with idiopathic chronic pancreatitis have been noted to cluster in a younger group (peak incidence, 15 to 30 years of age) and an older group (peak incidence, 50 to 70 years of age). Autoimmune chronic pancreatitis is an increasingly recognized condition frequently coexisting with other autoimmune diseases such as Sjögren’s syndrome and primary sclerosing cholangitis. An independent effect of tobacco smoking on the development of chronic pancreatitis has been suggested by several epidemiologic studies. In certain parts of developing regions such as Africa, India, and South America a particular type of chronic pancreatitis occurs in children and adolescents. It is thought to be caused by dietary toxins and micronutrient deficiencies and results in tropical pancreatitis endemic in these regions ( Table 49-1 ).
| Classification | Risk Factors | Comments |
|---|---|---|
| Toxic-metabolic (70%) | Alcoholic | |
| Tobacco smoking | ||
| Hypercalcemia | Associated hyperparathyroidism | |
| Hyperlipidemia | ||
| Chronic renal failure | ||
| Medications | Example: Phenacetin abuse | |
| Toxins | Example: Organotin compounds | |
| Idiopathic (20%) | Early onset | |
| Late onset | ||
| Tropical | Tropical calcific and fibrocalculous pancreatic diabetes | |
| Other | ||
| Others (10%) | ||
| Genetic | Autosomal dominant | Cationic trypsinogen gene |
| Autosomal recessive/modifier genes | CFTR mutations, SPINK1 mutations, cationic trypsinogen, alpha-1 antitrypsin deficiency (possible) | |
| Autoimmune | Isolated autoimmune chronic pancreatitis | |
| Syndromic | Sjögren’s syndrome, inflammatory bowel disease, primary biliary cirrhosis, sclerosing cholangitis | |
| Recurrent and severe acute pancreatitis | Postnecrotic | Severe acute pancreatitis |
| Recurrent acute pancreatitis | ||
| Vascular disease/ischemic | ||
| Radiation injury | ||
| Obstructive | Pancreas divisum | |
| Sphincter of Oddi disorders | Not universally accepted | |
| Duct obstruction | Example: Tumor | |
| Periampullary | ||
| Duodenal wall cysts | ||
| Posttraumatic | ||
| Pancreatic duct scars |
Prevalence and Epidemiology
Because of the varied clinical manifestation, recent increase in alcohol consumption, and improved sensitivity of diagnostic tests, the true prevalence of chronic pancreatitis is unknown, although estimates range from 0.04% to 5%. Earlier reports from Copenhagen, the United States, and Mexico City are nearly the same, with the incidence of chronic pancreatitis reported as 4 cases per year per 100,000 inhabitants. Several retrospective studies have reported an annual incidence of 3 to 9 cases per year per 100,000 population.
Prevalence and Epidemiology
Because of the varied clinical manifestation, recent increase in alcohol consumption, and improved sensitivity of diagnostic tests, the true prevalence of chronic pancreatitis is unknown, although estimates range from 0.04% to 5%. Earlier reports from Copenhagen, the United States, and Mexico City are nearly the same, with the incidence of chronic pancreatitis reported as 4 cases per year per 100,000 inhabitants. Several retrospective studies have reported an annual incidence of 3 to 9 cases per year per 100,000 population.
Clinical Presentation
Chronic abdominal pain is the most frequent presentation in most patients with alcohol-induced disease. The most common site of pain is the epigastrium, but some individuals report pain in the left or right upper quadrants that radiates to the back. The pain is described as severe, deep, and penetrating and is characteristically relieved by assuming a stooped or jack-knife posture. In some patients there is spontaneous remission of pain as organ failure sets in (burnout).
Exacerbation of pain may be associated with nausea and vomiting. Two patterns of pain have been described in chronic pancreatitis, as follows :
- •
Type A: Short relapsing episodes lasting days to weeks, separated by a pain-free interval
- •
Type B: Prolonged, severe, unrelenting pain
As the disease advances, approximately one fifth of patients present without pain but with pancreatic exocrine (fat and vitamin malabsorption) or endocrine failure, manifesting as steatorrhea, diabetes mellitus, or weight loss.
Structural complications of chronic pancreatitis such as pseudocysts and stricture of the common bile duct (CBD) in the pancreatic head are frequently seen. Thrombosis of the splenic or the portal vein with gastric or esophageal varices, arterial pseudoaneurysm formation, pancreatic abscess, ascites, and duodenal obstruction are seen less frequently.
Pathophysiology and Pathology
The pathophysiology of chronic pancreatitis includes chronic inflammation, irregular and patchy loss of acinar and ductal tissue, glandular atrophy, duct changes, and fibrosis. The pathogenesis of chronic pancreatitis is not well elucidated. Several theories have been developed for alcoholic pancreatitis.
- 1.
Stone and duct obstruction: Disturbance of acinar and ductular function with formation of intraductal protein plugs, stones, and duct obstruction with subsequent inflammation and fibrosis.
- 2.
Toxic metabolic theory: Results in early- and late-phase inflammatory responses and production of profibrotic cells, including stellate cells, with progressive lipid deposition, periacinar fibrosis, and inflammatory and fibrotic changes.
- 3.
Oxidative stress theory: Excess free radicals ultimately result in peroxidation of lipid components of the membrane within the pancreatic acinar cell leading to mast cell degranulation, platelet activation, and an inflammatory response.
- 4.
Necrosis-fibrosis sequence: Chronic contact of the stones with duct epithelial cells produces ulceration and scarring. Eventually, atrophy and fibrosis result from chronic obstruction of the acini. The necrosis-fibrosis theory emphasizes that acute and chronic pancreatitis represent a spectrum of disease. New research and discoveries about hereditary pancreatitis have supported the necrosis-fibrosis sequence.
Imaging
Radiography
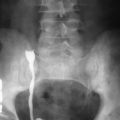
On abdominal radiographs calcifications can be found in 30% to 70% of patients at presentation, which suggests the diagnosis with 95% confidence. Pancreatic calcification is nearly 10% specific but poorly sensitive (30% to 70%) for the diagnosis of chronic pancreatitis. It is important to differentiate parenchymal or ductal calcification from calcified pancreatic cysts or occasionally from heavily calcified peripancreatic vasculature ( Figure 49-1 ).
Ultrasonography
Transabdominal ultrasonography is a noninvasive, easily accessible imaging modality for evaluation of the pancreas. However, it is limited in its evaluation of the pancreas owing to overlying bowel gas and patient body habitus and is operator dependent. Most ultrasound findings of chronic pancreatitis are not sensitive or specific for the diagnosis. Early diagnosis of chronic pancreatitis is difficult. The size of the gland may be normal, enlarged, or reduced, depending on the amount of active inflammation and fibrosis. Other ultrasound features include altered echotexture of the gland, parenchymal and ductal calcifications, pancreatic duct dilatation and irregularity, biliary dilatation, and, occasionally, pseudocysts.
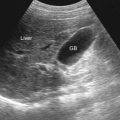
Pancreatic calcifications are a classic ultrasound finding and appear as multiple punctate, hyperechoic foci, which may be focal or distributed throughout the gland, seen in 40% of patients ( Figure 49-2 ). Pancreatic calcification is believed to be the most specific ultrasound finding for chronic pancreatitis. As the disease progresses, the pancreatic parenchyma demonstrates a heterogeneous echotexture with areas of increased and decreased echogenicity. Areas of increased echogenicity result from fibrosis and calcification, whereas areas of decreased echogenicity result from inflammation. The pancreas may be normal in size, enlarged, or atrophic depending on the amount of fibrosis and active inflammation. Most commonly, the gland atrophies in advanced disease. Ultrasound has been described as 97% sensitive for pseudocysts larger than 3 cm. Pancreatic pseudocysts are reported in 25% to 40% of patients. Doppler ultrasonography is useful for vascular complications such as portal and splenic vein thrombosis, varices, and splenic artery pseudoaneurysm.

Endoscopic Ultrasonography
Endoscopic ultrasonography was introduced in the early 1980s, and with advances in its technology it has emerged as a front-line diagnostic tool for detection of early chronic pancreatitis. The proximity of the pancreas to the stomach and duodenum allows acquisition of high-resolution imaging, thus overcoming the limitations of transabdominal ultrasonography—that is, bowel gas and body habitus. The normal pancreas has homogeneous echogenicity with fine granularity and reticulation and increased echogenicity relative to the liver. The margins are smooth without side-branch ectasia. The average pancreatic duct diameter in the body is 1.9 mm, and normal side branches are visible in 32% of patients.
The parenchymal abnormalities demonstrated with endoscopic ultrasonography are heterogeneous echogenicity with hyperechoic foci, prominent interlobular septa appearing as echogenic strands (secondary to fibrosis), hypoechoic foci (1 to 3 mm) resulting from small cystic changes in the parenchyma, lobular outer gland margins, and large echo-poor cavities (>5 mm). The ductal abnormalities include main pancreatic duct (MPD) dilatation more than 3 mm, irregularity, intraductal echogenic foci, hyperechoic margins of the pancreatic duct, side-branch ectasia, and intraductal calculi ( Table 49-2 ).
| Parenchymal Features | Ductal Features |
|---|---|
| Gland atrophy | Narrowing |
| Hyperechoic foci | Dilation |
| Hyperechoic stranding | Irregularity |
| Cysts | Calculi |
| Lobularity | Side branch dilation |
| Hyperechoic walls |
Computed Tomography
Contrast-enhanced computed tomography (CT) of the abdomen is the imaging modality of choice in the diagnosis of chronic pancreatitis. The CT features of chronic pancreatitis are scattered coarse parenchymal calcifications, intraductal calcifications, ductal dilatation, and parenchymal atrophy. In a retrospective study by Luetmer and colleagues, pancreatic ductal dilatation was reported in 68%, parenchymal atrophy in 54%, and pancreatic calcification in 50%.
Dilatation of the MPD and side branch ducts is the most common feature of chronic pancreatitis. Ductal dilatation can be smooth, beaded, or irregular; no single pattern is predominant, and more than one pattern can be seen in the same gland. Ductal dilatation is nonspecific as an isolated finding because it is also seen with distal CBD cholangiocarcinoma and pancreatic and ampullary carcinoma.
Altered glandular size is another key finding in chronic pancreatitis. Parenchymal atrophy was seen in 54% in a retrospective study conducted by Luetmer and colleagues. Parenchymal atrophy is frequently seen in association with ductal dilatation. Most patients with exocrine insufficiency have parenchymal atrophy or ductal dilatation. Atrophy, however, is a less sensitive finding in the elderly, in whom it may represent a normal aging process. Occasionally, either focal or diffuse enlargement of the pancreas is present.
A more reliable and the most specific sign of chronic pancreatitis is pancreatic calculi. Intraductal calcifications result in obstruction, ductal ectasia, and periductal fibrosis. Intraductal calculi range in size from microscopic to greater than 1 cm in diameter. Calcifications result from inspissation of pancreatic secretions within the duct and subsequent deposition of calcium carbonate on the intraductal protein plugs. Parenchymal calcifications vary in size and can be fine and stippled to large and coarse. The distribution may be focal, involving only one portion of the gland or diffuse throughout the gland. The head of the pancreas is more prominently involved than the tail. Pancreatic calcifications, when present, are virtually diagnostic of chronic pancreatitis and occur late in the course of the disease. However, in hereditary pancreatitis, calculi are seen early in the course of disease. They are large and rounded, and in the dilated MPD they tend to be arranged in a linear pattern.
CT also has the advantage of detecting complications of chronic pancreatitis. Fluid collections are seen in 30% and may be within the pancreas or adjacent to the pancreas. They may be seen in the retroperitoneum and rarely in distant sites. Extrapancreatic fluid collections are mostly seen in the lesser sac or anterior pararenal spaces. Most fluid collections in chronic pancreatitis are well-encapsulated mature pseudocysts that are present in 25% of cases. Ductal dilatation and intraductal calculi favor chronic pancreatitis as the underlying cause. Unencapsulated fluid collections are seen only with associated superimposed acute pancreatitis. Most pseudocysts resolve spontaneously but can be complicated by hemorrhage, superinfection, and spontaneous rupture.
Dilatation of the MPD is strongly associated. The dilated CBD demonstrates smooth gradual tapering as opposed to the abrupt cutoff shown in malignancy. It is often associated with an inflammatory mass in the pancreatic head.
Arterial pseudoaneurysms (pancreatic duodenal and splenic arteries) and splenic vein thrombosis are the most frequently encountered vascular complications. Pseudoaneurysms are seen near the pancreatic head and splenic hilum. They are caused by destruction of the vascular wall by pancreatic inflammation and appear as rounded areas isoattenuating to vascular structures. They also can be diagnosed with magnetic resonance imaging (MRI) and pulsed Doppler ultrasonography without intravenous administration of a contrast agent. The attenuation is similar to that of hematoma on noncontrast CT. Chronic pancreatitis is also associated with splenic, portal, and superior mesenteric vein thrombosis and accounts for 65% of cases of splenic vein thrombosis. Thrombus is seen as a filling defect in the vein. Venous thrombosis can result in prehepatic portal hypertension with venous collaterals and gastric varices.
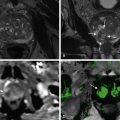
Pancreaticopleural fistulas are associated with chronic alcoholic pancreatitis. Patients present with chest symptoms more often than abdominal symptoms. Pancreatic secretions from a ruptured pancreatic duct dissect through the aortic and esophageal hiatus or through the diaphragm and gain access to the pleural cavity and mediastinum. CT and MRI with MR cholangiopancreatography (MRCP) can depict the fistula and ductal anatomy in these patients. Endoscopic retrograde cholangiopancreatography (ERCP) combined with CT can delineate the cause of recurrent pleural effusion and rare complications such as pancreaticobronchial fistula ( Figures 49-3 to 49-8 ).