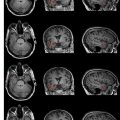
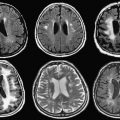
Imaging plays a crucial role in staging and the assessment of treatment response in patients who have lymphoma of the musculoskeletal system. This article reviews imaging features of lymphoma of bone, muscles, cutaneous, and subcutaneous tissue. At radiography, lymphoma of the bone is most commonly lytic, but the affected bone also can appear deceivingly normal, even when a large tumor is present. At CT, lymphoma of muscle can be homogenous in attenuation, and it may not show contrast enhancement, making tumor detection more difficult. Post-treatment changes often are encountered at MR imaging and positron emission tomography, and when considered in light of the patient’s therapy regimen (eg, radiation therapy and granulocyte-colony stimulating factor), they usually can be differentiated from tumor. Post-treatment changes include diffuse FDG uptake in marrow after chemotherapy, indicating rebound of normal marrow, and MR imaging signal abnormalities that may persist for anywhere from a few months to years after treatment.
Musculoskeletal involvement by lymphoma may occur as a part of disseminated disease or an isolated manifestation (ie, primary lymphoma of bone or muscle). Clinical features, prognosis, and treatment of lymphoma differ for isolated, primary lymphoma and disseminated disease, but their imaging features are similar. This article reviews imaging features of lymphoma of bone, muscles, and subcutaneous tissue. Evaluation of bone marrow changes with MR imaging and positron emission tomography (PET) will be discussed as well as post-treatment changes.
The evaluation of musculoskeletal lymphoma often requires more than one imaging modality and may include radiography, bone scintigraphy, CT, MR imaging, and/or PET. Lymphoma is a heterogenous disease, and thus its imaging features vary among the different modalities. Radiography, bone scintigraphy, CT, PET, and MR imaging each provide different information about the disease process. Radiography and CT depict bone destruction. Bone scintigraphy depicts the osteoblastic reaction and bone formation. PET depicts 2-(18F) fluoro-2-deoxy-D-glucose (FDG)-avid lesions in the bone marrow, and MR imaging detects changes by tumor and treatment in the bone marrow composition. For example, marrow involvement of lymphomas is detected better with PET and MR imaging, whereas cortical bone destruction and pathologic fractures are visualized easily with radiography and CT. Plasma cell myeloma and plasmocytoma are included in the 2001 World Health Organization (WHO) Classification Scheme for Lymphoma but will not be discussed explicitly in this article.
Lymphoma of the bone
Primary lymphoma of bone (PLB) is rare, comprising less than 1% of all lymphomas, 5% of extranodal non-Hodgkin lymphoma (NHL) and 3% to 5% of all primary bone tumors. PLB initially was described as reticulum cell sarcoma by Oberling in 1928, to distinguish it from other primary malignant bone tumors. Parker and Jackson reported the first case series of NHL in adults in 1939. The term PLB was introduced by Ivins and Dahlin in 1963. The diagnostic criteria for PLB include lymphoma within a single bone with or without regional nodal metastases and the absence of distal lesions within 6 months following the diagnosis. When PLB involves more than one bone without distal metastases, it is recognized as a subgroup of PLB and termed primary multifocal osseous lymphoma or multifocal primary lymphoma of bone. PLB has a predilection for the pelvis and appendicular skeleton, with the femur and tibia being the most common sites. Multifocal PLB also tends to involve vertebrae more often than PLB. Within long bones, diaphyses and metaphyses commonly are involved. The location of the involved bone does not appear to have a statistically significant effect on the overall survival rate of PLB.
Multiorgan involvement with involvement of the bone or relapsed lymphoma with involvement of the bone has imaging and immunocytologic features similar to those of PLB and will not be discussed separately.
Clinical Features
The most common symptom is bone pain (61%), but B symptoms (fever, night sweat, and weight loss) are rare (13%) in PLB. Palpable masses and an elevated level of lactate dehydrogenase (LDH) (31%) may be present. Most cases of PLB and multifocal osseous lymphoma involvement result from NHL. PLB is more common in males (male-to-female ratio 1.5-2.3:1). The median age is 42 to 54 years. PBL is rare in children younger than 10 years of age. The overall survival rate for PLB is better than the survival rates for other primary bone tumors. For example, in a study of 82 patients who had PLB, Beal and colleagues reported that the 5-year survival rate (with combined radiation and chemotherapy, radiation alone or chemotherapy alone) was 88%, and in a study of 77 patients who had PLB, Barbieri and colleagues reported that the 15-year survival rate (with radiation, with or without chemotherapy) was 88.3%. In contrast, a study of 1702 patients who had osteosarcoma by Bielack and colleagues found 5-year and 15-year survival rates of 65% and 57%, respectively. In adults, favorable prognostic factors for overall survival include age younger than 40 years, lack of B symptoms, normal LDH level, and female gender.
Imaging
Lymphoma can occur in bone marrow, cortical bone, or both, and also may extend outside of bone. When tumor is confined only to bone marrow, infiltration of the bone marrow with lymphoma cells may have no imaging correlate on MR imaging or PET and often is diagnosed by bone marrow biopsy. When a cortical lesion is associated with an extraosseous soft tissue mass, it may be difficult to distinguish lymphoma of bone with extraosseous extension from soft tissue lymphoma invading the bone. This distinction, however, does not influence staging or treatment significantly.
Radiography
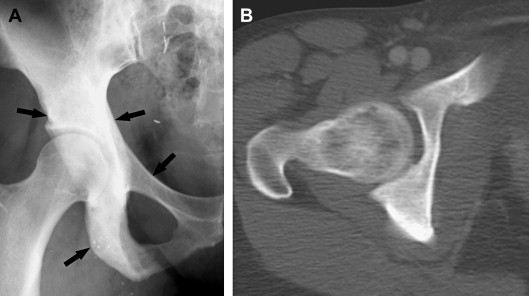
Radiographic manifestation of osseous lymphoma is variable and nonspecific and tends to under-represent the extent of osseous lesions, especially when lesions are confined to the marrow cavity. Therefore, additional imaging such as MR imaging or PET usually is obtained for further evaluation. The radiographic pattern can be normal, predominantly lytic, sclerotic, or mixed lytic–sclerotic. In particular, osseous lymphoma is known for having a deceivingly normal appearance on radiography while displaying extensive abnormality on cross-sectional modalities and bone scintigraphy ( Figs. 1–3 ). When it is visible on radiography, it most often displays a lytic pattern. Lytic osseous lymphoma may be well circumscribed, or it may be characterized by a wide zone of transition (permeative or moth-eaten). Cortical destruction, extraosseous soft-tissue masses, and periosteal reaction indicate advanced local disease ( Fig. 4 ). Periosteal reactions are usually aggressive patterns, including linear, lamellated, or disrupted, and disrupted periosteal bone formation is considered to be an indicator of poor prognosis. Pathologic fractures are not rare at the time of diagnosis (17% to 22%). Differential diagnostic considerations of lytic osseous lymphoma include both malignant and benign categories. In adults, they may include metastases, chondroblastic osteosarcoma, multiple myeloma, plasmacytoma, osteomyelitis, and giant cell tumor (especially in the end of long bones). In children, differential diagnostic considerations may include Ewing sarcoma, primitive neuroectodermal tumor (PNET), infection, and eosinophilic granuloma.
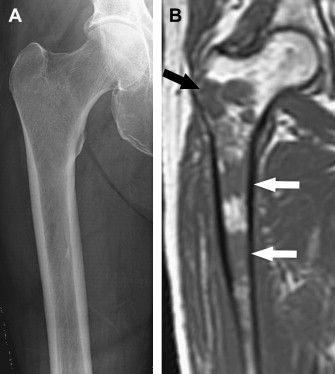
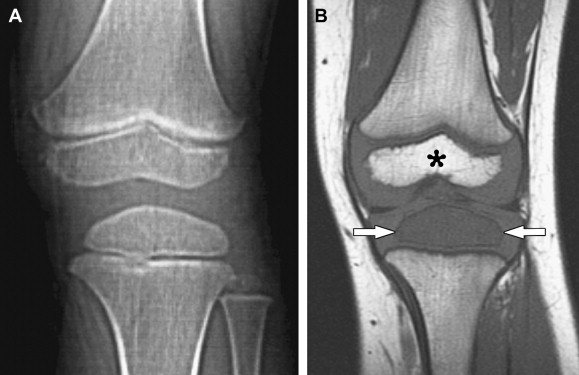
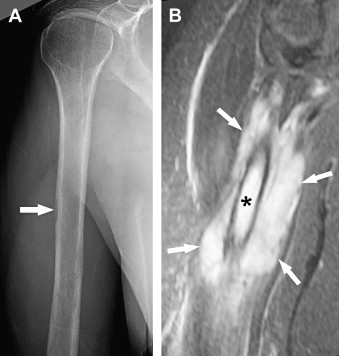
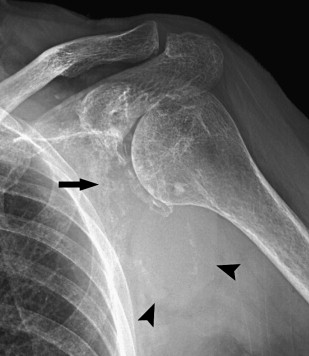
A sclerotic pattern of osseous lymphoma was reported in 2% of PLB in a study of 237 patients ; it is seen more frequently in Hodgkin lymphoma (HL) ( Fig. 5 ) and reported in 24% of 15 patients whose radiography was performed in one study. Diffuse sclerosis may be related to fibrosis, with dense ivory vertebrae being a classic presentation, and sclerosis can develop following chemotherapy or radiation. Diffuse sclerotic osseous lesions also have been associated with other neoplastic and non-neoplastic processes such as primary bone tumor (osteosarcoma), metastases (from prostate or breast cancer), Paget’s disease, and sarcoidosis.