With cognitive disorders increasingly common, clinicians urgently need faster and more accurate tools to classify such disorders and to noninvasively monitor therapeutic interventions. In this review, we provide information on MRI techniques that enable the study of the morphology, neuronal integrity, and metabolism of dementing illnesses. In addition, we explore the usefulness of such techniques as surrogate markers of these diseases.
The incidence of both cognitive disorders and dementing illnesses is rising in our ageing population. As a consequence, imaging modalities are becoming increasingly important for differential diagnosis and monitoring of disease progression in daily clinical practice and for use as surrogate markers in treatment trials. Such technologies as conventional CT, MRI, proton magnetic resonance spectroscopy (MRS), positron emission tomography (PET), and single photon emission CT are currently being applied for this purpose.
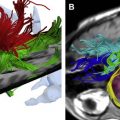
Additionally, diffusion-weighted imaging, diffusion tensor imaging (DTI), perfusion-weighted MRI, magnetic transfer imaging, and a combined visualization of different modalities (eg, MRI/PET) have been proposed for in vivo tracing of biomarkers. We provide a review of original papers as well as published review articles on the role of MRI in various cognitive disorders.
In general, every measurement method runs through different stages of development and validation before it can be used as a routine tool ( Fig. 1 ). Along these lines, we will assess the usefulness of MRI for early diagnosis, for monitoring of disease progression, and for use as a surrogate marker for documenting drug efficacy in therapeutic clinical studies.

Alzheimer’s disease
Conceptually, treatment of Alzheimer’s disease should start early in the disease, ideally in a clinically presymptomatic stage, before widespread synaptic and neuronal loss has occurred. This approach would require tools that already pick up with high sensitivity subtle Alzheimer’s disease–related brain changes.
Structural MRI
Methods to assess brain volume changes in Alzheimer’s disease patients include voxel-based morphometry or the voxel-based specific regional analysis system for Alzheimer’s disease involving transformation to a normalized brain, cortical pattern matching, and brain boundary shift integral measurements. These techniques make it possible for clinicians to discriminate between the magnitude of progression of brain atrophy in normal ageing and the higher rates of volume loss observed in Alzheimer’s disease. Brain boundary shift integral measurements might additionally be useful for assessing subtle regional volume changes. Such assessments have been helpful for demonstrating significant drug effects even in relatively small patient groups.
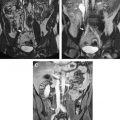
Structural MRI has also been used in several studies to predict progression from mild cognitive impairment to Alzheimer’s disease. Other MRI measurement techniques employed in the assessment of Alzheimer’s disease are manual and semiautomated measurements of regional atrophy in specific regions of interest ( Fig. 2 ). Studies using this approach have focused primarily on the hippocampus and the medial temporal lobes, yet the corpus callosum has also been shown to be vulnerable to Alzheimer’s disease–related brain atrophy. In this context, it is of note that hippocampal atrophy in MRI is strongly related to the histologic finding of neuronal loss and to the severity of Alzheimer’s disease pathology, and that hippocampal atrophy correlates with cognitive impairment.

In even earlier disease stages, tracking shrinkage of the entorhinal cortex may also be useful for predicting Alzheimer’s disease. Atrophy rates have been reported to be larger in entorhinal cortex than in the hippocampus (∼7% per year versus ∼6% per year), but difficulties in unambiguously defining this brain structure on the basis of landmarks make entorhinal cortex measurements highly variable. In general, manual regions-of-interest measurements are both operator-dependent and labor-intensive.
Both regions-of-interest measurements and automated measurement approaches assessing hippocampal volume changes have already proven meaningful and feasible in clinical trials. However, a recent study suggested that whole brain atrophy measures might even be more closely related to clinical and cognitive scores (Mini-Mental State Examination, Dementia Rating Scale, Clinical Dementia Rating) than to the annual change in hippocampal and entorhinal cortex volume.
MRI indices enable assessments of processes linked to disease progression with sensitivity far beyond what would be detectable on the basis of cognitive and clinical scores. Consequently, trials incorporating such MRI markers require fewer subjects, which in turn makes it easier to conduct proof-of-concept studies (for a more detailed review on quantitative MRI of different brain regions see Ramani and colleagues ). As for potential applications of MRI in related clinical trials, a consensus report of the Alzheimer’s Association identified two main areas: (1) for supplementary exclusion and for more stringent stratification criteria to define a homogeneous study population, and (2) for strong correlation with clinical symptoms and neuropathology at earlier stages of the disease and the possibility to trace the progression of the disease.
Nonetheless, because of its lack in specificity, structural MRI cannot currently be considered an established diagnostic tool on an individual basis. Also, for research purposes, standardized acquisition schemes, procedures for regular checks on scanner quality, and standardized protocols for processing and analyzing MRI data in dementia need to be developed.
Diffusion-Weighted Imaging and Diffusion-Tensor Imaging
Several studies have used the apparent diffusion coefficient or diffusion characteristics derived from DTI to gain information on demyelinization and axonal loss in the context of Alzheimer’s disease. The apparent diffusion coefficient provides information on the overall mobility of tissue water while DTI provides additional directional information on this diffusion process.
White matter changes of the brain can be associated with a net loss of barriers restricting the motion of water molecules and tissue anisotropy of white matter. Histopathology of Alzheimer’s disease brains also shows partial loss of myelin, axons, and oligodendrial cells, and reactive gliosis in the white matter. However, diminished fractional anisotropy in normal-appearing white matter may also be caused by other factors, such as membrane damage, edema, reduced axoplasmatic flow related to cytoskeletal dysfunction, and alterations of ion or fluid homeostasis. Several studies set out to define in vivo estimates of Alzheimer’s disease–related structural changes by using DTI techniques and measuring increased apparent diffusion coefficient and decreased fractional anisotropy in the temporal lobe white matter, the hippocampus, the posterior white matter, and the corpus callosum of patients with mild to moderate Alzheimer’s disease. Another approach found a correlation between the lattice index and the Mini-Mental State Examination score in the splenium of the corpus callosum. Together, these measures may reflect very early biophysical alterations linked to the progression of Alzheimer’s disease, although an overlap between patients and groups of healthy controls also has been noted.
Albeit not yet validated to the extent of structural MRI, both diffusion-weighted imaging and DTI appear as promising candidates with potential to support an imaging diagnosis of Alzheimer’s disease. A strong correlation between apparent diffusion coefficient and Mini-Mental State Examination scores underlines a possible role for clinical use. However, some technical issues still must be resolved regarding the optimum diffusion time and the optimal strength of the diffusion gradients (b values). Generally, the use of higher b values has been shown to provide a better lesion-to–normal tissue contrast and a better contrast-to-noise ratio in Alzheimer’s disease. Magnetic resonance units with more powerful gradients should, therefore, be able to markedly improve the signal-to-noise ratio and thus allow higher spatial resolution with less susceptibility effects. However, standardization and quality assurance procedures are still lacking, although endeavors to overcome these problems on different scanner systems have already been proposed.
Magnetization Transfer Ratio
Magnetization transfer imaging can provide pathophysiological information on brain structure on a microscopic level. This notion is supported by magnetic resonance histopathological studies demonstrating a correlation between the magnetization transfer ratio (MTR) and the extent of histologically defined demyelinization and axon loss.
MTR has rarely been used so far to assess Alzheimer’s disease. Further studies are needed to determine the effectiveness of this technique for the clinical diagnosis of Alzheimer’s disease and to assess its value for monitoring disease progression. However, preliminary studies suggest that MTR holds promise as a tool for early diagnosis of Alzheimer’s disease with an even higher specificity for neurodegeneration compared with other surrogate markers. This notion is supported by the observation that MTR in the hippocampus was found to be decreased in Alzheimer’s disease patients and by the detection of MTR changes in gray and white matter in mild cognitive impairment subjects who did not yet show notable brain volume change. MTR changes therefore might precede gross regional atrophy.
An additional future application for magnetic transfer imaging could be the detection of microglia surrounding neuritic plaques in Alzheimer’s disease, as suggested by a 3-T MRI study. Song and colleagues were able to detect exogenously administered microglia noninvasively in rat brains using magnetic transfer imaging, and corroborated their findings by histopathology. Because modern Alzheimer’s disease therapy also aims to affect immunoreactive processes, such as microglia activation in the brain, such a noninvasive way to monitor the potential effects of anti-inflammatory therapies in clinical trials could turn out to be particularly useful.
Magnetic Resonance Spectroscopy
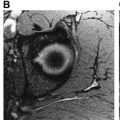
MRS provides biochemical information on the metabolism of the brain. Using MRS, differences in N-acetyl aspartate (NAA) and myoinositol levels between Alzheimer’s disease patients and normal ageing persons have been demonstrated. NAA is thought to represent the neuroal dysfunction and loss, whereas myoinositol is deemed a marker of gliosis ( Fig. 3 ).

The combination of volumetric and spectroscopic magnetic resonance appears superior in terms of sensitivity and specificity to either measure alone. Longitudinal MRS revealed correlations between NAA, choline, and cognitive function in clinical trials on cholinergic agents. This leads some to speculate that MRS might serve to connect traditional measures of brain structure with traditional measures of brain function.
Functional MRI
Functional MRI allows clinicians to noninvasively study changes in neural activity associated after an experimental paradigm (eg, during the performance of a cognitive task in the scanner). Clinical applications of this method mostly rely on the measurement of the so-called “blood oxygenation level–dependent” (BOLD) response, thereby providing a composite surrogate signal of neural activity as defined by the neurovascular coupling hypothesis.
Regarding functional measurements in the field of Alzheimer’s disease, functional MRI conceptually offers the opportunity to study deviations from normal functional processes on a neural system level, ideally and—like other functional techniques, such as PET—even before morphologic changes related to the disease process become detectable by other methods. Such study might finally lead to a definition of “functional phenotypes” of Alzheimer’s disease patients, with individually differential risks for conversion to overt clinical disease, defined by a differential capacity to neural plasticity involved in the partial limitation of the clinical expression of the disease process.
Indeed, in cases with a clinical diagnosis of probable Alzheimer’s disease, functional MRI studies assessing the neural response to different memory tasks relatively consistently reported decreased hippocampal and parahippocampal activation compared with age-matched elderly normal controls. However, other brain regions also have been implicated as relevant in this context. Furthermore, in general, the number of patients studied was small (usually fewer than 10 subjects per study) and different functional MRI tasks have been used (eg, divided attention, subtraction, picture or face encoding, watching novel versus previously learned scenes or items, visual search tasks). Together with the variability in the BOLD response, this might explain some of the inconsistencies in the findings among different studies.
However, preliminary studies suggested that functional MRI might prove particularly useful in short-term, early proof of concept studies, given its potential to evaluate acute und subacute effects of medications on neural activity. This notion is supported by the observation that cholinergic enhancement with a single dose of rivastigmine led to an increase in functional MRI activity both in the fusiform face area during face encoding and in the prefrontal cortex during a working memory task in subjects with mild Alzheimer’s disease.
Functional MRI therefore holds great promise to elucidate mechanisms of functional cortical and subcortical reorganization associated with the progression of the disease, as well as to allow clinicians to visualize how drugs modulate functional networks and potentially to define pharmacologic responders. However, using functional MRI in cohorts of demented or even more cognitively compromised subjects poses obvious challenges, especially regarding issues of reproducibility in longitudinal designs and, even more basically, concerning the applicability and feasibility of more complicated and refined functional MRI paradigms. To overcome some of these problems, resting state functional MRI has been proposed. This enables the study of functional default networks without the necessity for adhering to given tasks. However, the value of this approach in a clinical setting has yet to be determined. Future functional MRI studies will certainly also need to address how much biochemical, (micro)vascular, and morphologic changes associated with the disease process itself potentially affect or confound the surrogate signal under investigation.
Amyloid Imaging
Because Alzheimer’s disease is characterized by aggregates of β-amyloid peptides forming senile plaques, in vivo imaging of these pathologic hallmarks would enable clinicians to monitor progression and effectiveness of amyloid-targeted therapies, such as vaccination, secretase inhibitors, or beta sheet breaker compounds.
The first promising data about the use of such imaging came from studies with transgenic mice that harbor mutated forms of the human amyloid precursor protein as found in familial inherited Alzheimer’s disease. Both PET and MRI were used to assess the amyloid load noninvasively. The theoretic advantages of MRI over PET are its superior spatial resolution, its noninvasive nature, as well as the more widespread availability of MRI scanners compared with PET machines. Imaging strategies varied among the studies from ligand-based detection to the use of the average MRI signal to gain information on the plaques. The latter approach often has been combined with an optimized pulse sequence to be able to visualize individual plaques. Using such an approach, the regional averaged MRI signal has been found to be different between transgenic and nontransgenic littermates and refined pulse sequences have allowed measurements of signal changes corresponding spatially and quantitatively to individual plaques. However, concerns have been raised that these methods might be insensitive to plaques containing little or no iron. Furthermore, iron can be additionally found not only in plaques but also in microhemorrhages, which also occur in Alzheimer’s disease brains. Together, this furthered the renascence of ligand-based approaches, which are less likely to provide misleading findings.
The first ligands used in this context—gadolinium or iron nanoparticles binding to Aβ—necessitated the coadministration of mannitol to open the blood-brain barrier. Most recently, substances with potential to overcome this limitation have been proposed. Thus, 19 fluorine ( 19 F) has been shown to penetrate the brain after intravenous injection, at least in transgenic mice. While mannitol makes the application in humans difficult because of the reasons given, 19 fluorine does not inherit this intrinsic problem.
Skovronsky and colleagues cross-correlated 19 F-containing (E,E)-1-fluoro-2,5-bis(3-hydroxycarbonly-4-hydroxy)styrylbenzene (FSB) MRI maps with FSB binding in postmortem histologic slices, confirming this approach to be a reliable in vivo indicator of amyloid plaques and neurofibrillary tangles. Other researchers detected plaques in T2-weighted images and confirmed their results in Congo-red stained slices histopathologically.
The main technical advances achieved over the last few years concerning MRI scanners were the increase of the magnetic field strength and the availability of high-performance gradient technology and array coils. Together, these achievements in concert with further research efforts could finally result in the in vivo visualization of amyloid plaques in humans using MRI. Compared to other approaches, amyloid imaging will have the advantage of higher specificity because this pathology is typical in Alzheimer’s disease dementia. Future studies should have three goals: (1) the identification of ligands to amyloid plaques safe for use in human studies, (2) exploration of approaches for detecting soluble Aβ species, and (3) determination of methods specific to the second hallmark of Alzheimer’s disease, the neurofibrillary tangles.
Vascular dementia
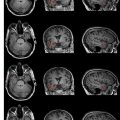
Vascular dementia is a heterogeneous disorder. Various subtypes can be defined clinically. These include poststroke dementia, multi-infarct dementia, dementia with strategically located infarctions, and, the most common type, subcortical ischemic vascular dementia, with white matter lesions and lacunes as hallmark lesions ( Fig. 4 ). A first attempt to define imaging correlates has been provided in the NINDS-AIREN (National Institute of Neurological Disorders and Stroke–Association Internationale pour la Recherche et l’Enseignment en Neurosciences) criteria. Yet, in a study on interrater reliability of these criteria, it became clear that they are very complex and not suited for use by inexperienced raters.

The radiological criteria included in the NINDS-AIREN criteria for vascular dementia have suboptimal reproducibility. Use of operational criteria improves agreement to acceptable levels, but only in experienced readers. Box 1 gives an overview of these operational criteria.
Topography
Large-vessel stroke: Large-vessel stroke is an infarction defined as a parenchymal defect in an arterial territory involving the cortical gray matter.
Anterior cerebral artery: Only bilateral anterior cerebral artery infarcts are sufficient to meet the NINDS-AIREN criteria.
Posterior cerebral artery: Infarcts in the posterior cerebral artery territory can be included only when they involve the following regions:
Paramedian thalamic infarction: The infarct includes the cortical gray matter of the temporal/occipital lobe and extends into the paramedian part (defined as extending to the third ventricle) of the thalamus; the extension may be limited to the gliotic rim of the infarct that surrounds the parenchymal defect.
Inferior medial temporal lobe lesions
Association areas: A middle cerebral artery infarction needs to involve the following regions:
Parietotemporal: The infarct involves both the parietal and temporal lobes (eg, angular gyrus).
Temporo-occipital: The infarct involves both the temporal and occipital lobes.
Watershed carotid territories: A watershed infarction is defined as an infarct in the watershed area between the middle cerebral artery and posterior cerebral artery or the middle cerebral artery and anterior cerebral artery in the following regions:
Superior frontal region
Parietal region
Small-vessel disease
Ischemic pathology resulting from occlusion of small perforating arteries may manifest itself as lacunes or white matter lesions. Lacune is defined as a lesion greater than 2 mm in diameter with cerebrospinal fluid–like intensity on all sequences on MRI (water density on CT) surrounded by white matter or subcortical gray matter. Care should be taken not to include Virchow-Robin spaces, which typically occur at the vertex and around the anterior commisure near the substantia perforata. Ischemic white matter lesions are defined as circumscribed abnormalities with high signal on T2-weighted images not following cerebrospinal fluid signal (mildly hypodense compared with surrounding tissue on CT) with a minimum diameter of 2 mm.
Multiple basal ganglia and frontal white matter lacunes: Criteria are met when at least two lacunes in the basal ganglia region (including thalamus and internal capsule) and at least two lacunes in the frontal white matter are present.
Extensive periventricular white matter lesions: Lesions in the white matter abutting the ventricles and extending irregularly into the deep white matter, or deep/subcortical white matter lesions. Smooth caps and bands by themselves are not sufficient. Gliotic areas surrounding large-vessel strokes should not be included here.
Bilateral thalamic lesions: To meet the criteria, at least one lesion in each thalamus should be present.
Severity
Large-vessel disease of the dominant hemisphere: If there is a large-vessel infarct as defined above, it has to be in the dominant hemisphere to meet the criteria. In the absence of clinical information, the left hemisphere is considered dominant.
Bilateral large-vessel hemispheric strokes: One of the infarcts should involve an area listed under topography but is in the nondominant hemisphere, while the infarct in the dominant hemisphere does not meet the topography criteria.
Leukoencephalopathy involving at least one fourth of the total white matter: Extensive white matter lesions are considered to involve one fourth of the total white matter when they are confluent (grade 3 in the Age-Related White-Matter Changes [ARWMC] scale) in at least two regions of the ARWMC scale and beginning confluent (grade 2 in the ARWMC scale) in two other regions. A lesion is considered confluent when greater than 20 mm in diameter, or consists of two or more smaller lesions fused by more than connecting bridges.
Fulfillment of radiological criteria for probable vascular dementia
Large-vessel disease: Both the topography and severity criteria should be met (a lesion must be scored in at least one subsection of both topography and severity).
Small-vessel disease: for white-matter lesions, both the topography and severity criteria should be met (a lesion must be scored in at least one subsection of both topography and severity); for multiple lacunes and bilateral thalamic lesions, the topography criterion alone is sufficient.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree