The visceral mediastinum contains important vascular and non-vascular structures including the heart, great vessels, lymph nodes, and portions of the esophagus and trachea. Multiple imaging modalities, including chest radiography, computed tomography, MR imaging, and nuclear medicine studies, can be used to detect, diagnose, and characterize masses in this compartment. Lymphadenopathy is the most common process involving the visceral mediastinum and can be seen with a wide variety of diseases. Less commonly seen entities include foregut duplication cysts, neoplasms and other lesions arising from the trachea and esophagus, paragangliomas as well as other mesenchymal tumors.
Key points
- •
The middle or visceral mediastinum contains the heart, great vessels, lymph nodes, the esophagus and portions of the trachea.
- •
Computed tomography scanning is the primary imaging modality used to identify and characterize mediastinal abnormalities. MR imaging and nuclear medicine studies can help with problem solving and refining the differential diagnosis.
- •
Lymphadenopathy is the most common disease process involving the visceral mediastinum and occurs with a wide variety of benign and malignant processes.
- •
Other abnormalities that can be diagnosed on imaging include foregut duplication cysts, neoplasms and other lesions arising from the trachea and esophagus, and paragangliomas.
- •
In addition to identifying, localizing, and characterizing lesions in the visceral mediastinum, imaging plays an important role in directing invasive diagnostic procedures and monitoring response to treatment.
Introduction
The middle or visceral mediastinum is notable for containing the heart, great vessels, lymph nodes, and portions of the esophagus and trachea. In terms of pathology, the most common abnormalities involving this compartment include lymphadenopathy, foregut duplication cysts such as bronchogenic and esophageal duplication cysts, tracheal and esophageal neoplasms, and other diseases. Imaging plays a key role in identifying and diagnosing these conditions as well as guiding biopsy or surgery if needed.
Normal anatomy and imaging technique
The mediastinum contains several vascular and nonvascular structures and its division into different compartments is crucial for localization and development of an accurate differential diagnosis. The International Thymic Malignancy Interest Group has developed a classification of mediastinal compartments based on cross-sectional imaging to help with characterization of mediastinal lesions and facilitate communication among medical specialties. This classification is accepted as a standard. ,
The visceral mediastinal compartment is characterized by the following boundaries: (1) the thoracic inlet superiorly, (2) the diaphragm inferiorly, (3) the anterior aspect of the pericardium anteriorly, and (4) a vertical line situated 1 cm posterior to the anterior margin of each vertebral body posteriorly. Thus, it includes several important vascular structures such as the heart, thoracic aorta, intrapericardial pulmonary arteries, superior vena cava, and thoracic duct. Nonvascular structures incorporated in this compartment include portions of the trachea, including the carina, the esophagus, and the lymph nodes. ,
Anomalies of the visceral mediastinum may be initially detected on chest radiography. Although the identification of small lesions is often difficult, careful examination of the mediastinum on chest radiography may reveal unsuspected abnormalities in otherwise asymptomatic individuals. Mediastinal masses typically present with focal contour abnormalities of the mediastinum or diffuse widening of the mediastinum. Indirect signs with displacement of mediastinal lines and stripes can also be seen. In the case of a hilar or perihilar mass, the hilum overlay sign, which is defined as a visibility of the hilar vasculature through the opacity, suggests that the mass is either within the anterior or posterior compartments. A different sign, the hilum convergence sign, can help to distinguish between pulmonary artery enlargement (manifesting as pulmonary vessels that converge toward the edge of the mass-like opacity) versus a nonvascular hilar mass, such as lung malignancy or lymphadenopathy (in which vessels do not converge toward the edge of the mass-like opacity). If only a frontal radiograph is available, obtaining a lateral radiograph may be helpful in localizing mediastinal anomalies to the proper compartment. After the initial detection of a potential mediastinal lesion, further characterization with cross-sectional imaging is warranted.
Computed tomography (CT) scanning remains the imaging modality of choice to evaluate most mediastinal abnormalities owing to its widespread availability, ability to localize lesions to the correct compartment, and excellent spatial resolution. It can also be used for tissue characterization with detection of macroscopic fat, cystic components, calcification, hemorrhage, and hypervascularity. , Intravenous (IV) iodinated contrast is usually used, because it allows for an assessment of lesion enhancement and helps in differentiating potential masses from vascular structures.
MR imaging is beneficial for its excellent tissue characterization and lack of ionizing radiation. Thoracic MR imaging provides greater diagnostic precision in the evaluation of indeterminate mediastinal masses on CT scan, such as complex cystic masses or masses containing intralesional fat or hemorrhage. , It can also be used as a staging study in patients with contrast allergy or renal failure, in whom a contrast-enhanced CT scan may be contraindicated.
Finally, several nuclear medicine studies may be used as additional problem-solving modalities. A PET/CT scan with fluorodeoxyglucose can be used to identify distant metastases in the setting of suspected malignancy. 123 I-Metaiodobenzylguanidine, In-111octreotide scintigraphy, and a PET/CT scan with fluorodeoxyglucose can be used in patients with suspected paragangliomas to further narrow the differential diagnosis. 99mTc pertechnetate scans have a role in the further evaluation of suspected esophageal duplication cysts, where it can accurately localize functioning ectopic gastric mucosa.
Imaging findings and pathology
Lymphadenopathy
Stations
The visceral compartment of the mediastinum includes multiple lymph node stations, including upper and lower paratracheal, subaortic, para-aortic, subcarinal, paraesophageal, and pulmonary ligamental lymph nodes. The enlargement of 1 or more of these lymph nodes may be seen in a wide variety of benign and malignant processes and is the most common cause of a visceral mediastinal mass. A CT scan has greater sensitivity than chest radiography for the detection of lymph node enlargement. A PET/CT scan with fluorodeoxyglucose also plays an important role in evaluation of lymphadenopathy and in directing endobronchial ultrasound-guided biopsies.
Appearance and size
On a CT scan, lymph nodes typically appear as oval or round homogeneous masses of soft tissue attenuation in their expected anatomic location. The size criteria for abnormal lymph node enlargement have been discussed in the literature and measurements range from 10 to 15 mm depending on the location. , The sensitivity and specificity of a CT scan are limited in differentiating malignant and benign lymph node enlargement. However, a CT scan can be used to guide invasive diagnostic procedures and its accessibility makes it a useful modality for monitoring treatment response.
Causes of nodal enlargement
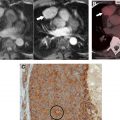
Malignant lymphadenopathy can be seen in metastatic disease (from thoracic or extrathoracic primary neoplasms) or lymphoproliferative disorders. Benign enlargement may be seen in the context of pneumonia or pulmonary edema, an inflammatory process like sarcoidosis ( Fig. 1 ), or granulomatous infection, or rarer entities like angiofollicular lymph node hyperplasia (Castleman disease, discussed in detail elsewhere in this article). The differential diagnosis can be narrowed by measuring the density of the lymph nodes and evaluating their distribution ( Tables 1 and 2 ).

| Category | Possible Causes |
|---|---|
| Neoplastic | Metastases, lymphoproliferative disorders |
| Infectious | Mycobacterial and fungal infection |
| Inflammatory | Sarcoidosis |
| Pneumoconioses | Silicosis, coal worker’s pneumoconiosis, berylliosis |
| Reactive | Pulmonary edema, interstitial lung disease |
| Lymph Node Density | Possible Causes |
|---|---|
| Hypoattenuating | Cystic or necrotic metastases, mycobacterial and fungal infections, lymphoma |
| Hyperattenuating | Sarcoidosis, silicosis and other pneumoconiosis (berylliosis), prior granulomatous infection, amyloidosis, treated lymphoma |
| Hyperenhancing | Hypervascular metastases, Castleman disease, Kaposi sarcoma |
Lymph Node Attenuation
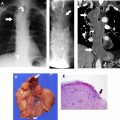
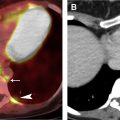
Hypoattenuating lymph nodes usually represent necrosis and are commonly seen in metastatic disease ( Fig. 2 ) or atypical infectious processes caused by fungal or mycobacterial agents. , Much rarer entities include sprue and necrotizing lymphadenitis (Kikuchi–Fujimoto disease).

A hyperattenuating appearance of the lymph nodes may be due to the presence of calcification or mineral dust or due to hypervascularity. The presence of calcifications or other dense inorganic materials can be seen in sarcoidosis, remote granulomatous infection (most commonly tuberculosis and histoplasmosis), treated lymphoproliferative disorders, amyloidosis, and pneumoconioses, such as silicosis and berylliosis. Metastases from thyroid carcinoma and mucinous adenocarcinoma may also calcify.
Hypervascular lymphadenopathy, characterized by brisk enhancement after IV contrast administration, can be seen in metastatic disease, usually from a select group of tumors, including renal cell carcinoma, melanoma, papillary thyroid carcinoma, sarcoma, and neuroendocrine tumors ( Box 1 , Table 3 ). Rarer entities include angiofollicular lymph node hyperplasia (Castleman disease) and Kaposi sarcoma. , Kikuchi–Fujimoto and Kimura diseases, commonly presenting with cervical lymphadenopathy, can also cause mediastinal lymph node enlargement. ,
Narrows differential diagnosis to a small group of entities.
Entails propensity to bleed and need for careful preprocedure planning, including embolization before any procedure.
Percutaneous biopsy may not be safe; surgical biopsy allowing for better control of hemorrhage may be preferred.
| Benign | Malignant or Malignant Potential | Metastases |
|---|---|---|
| Castleman’s disease Ectopic parathyroid adenoma Vascular malformations | Sarcomas Paraganglioma Carcinoid Castleman’s disease | Renal cell carcinoma Thyroid carcinoma Melanoma Pheochromocytoma Carcinoid Choriocarcinoma |
Patterns of Lymph Node Enlargement in Neoplastic and Non-neoplastic Entities
An understanding of lymphatic drainage pathways is helpful in directing attention to relevant stations when interpreting an imaging examination, especially for the staging of cancer. The distribution of lymphadenopathy can also give clues as to the site of the primary tumor in the setting of metastatic disease or suggest alternative diagnoses.
Lung neoplasms usually drain first to the hilar lymph nodes, although direct mediastinal involvement is also possible. Subsequent drainage pathways in the mediastinum depend on the lobe of origin. Right upper lobe tumors drain into the right paratracheal and anterior mediastinal lymph nodes, whereas lesions of the middle and lower lobes drain into the subcarinal lymph nodes before continuing to the right paratracheal lymph nodes. Left upper lobe tumors drain preferentially to the para-aortic and subaortic lymph nodes, whereas those in the left lower lobe favor the subcarinal and subaortic stations.
The upper two-thirds of the esophagus generally drain cranially, whereas the lower one-third drains caudally. Therefore, neoplasia involving the upper two-thirds tend to involve the paratracheal lymph nodes, whereas tumors of the lower one-third preferentially involve the gastrohepatic lymph nodes. The esophageal lymphatics communicate with the thoracic duct; distant nodal metastases are also possible.
Enlargement of the lymph nodes involving multiple mediastinal stations and bilateral hila can be seen in lymphoproliferative disorders. Hodgkin lymphoma in particular involves the mediastinum in up to 85% of cases, especially the superior paratracheal stations and prevascular mediastinum. Non-Hodgkin lymphoma may also involve the mediastinum but does so less frequently and sometimes present as lymphadenopathy in a single station.
Sarcoidosis is a common cause of bilateral hilar lymph node enlargement and should be a leading differential in the absence of known malignancy. Involvement of the paratracheal and subcarinal lymph nodes is also frequent. Pneumoconioses such as silicosis and coal worker’s disease can also manifest with a similar distribution, as well as fungal and mycobacterial infections. As outlined elsewhere in this article, measuring the internal density of these lymph nodes may help to narrow the differential diagnosis. Finally, mild diffuse lymph node enlargement can also be seen in reaction to diffuse lung processes, such as pulmonary edema and interstitial lung disease.
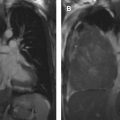
Castleman Disease
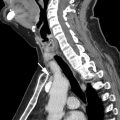
Angiofollicular lymph node hyperplasia, also known as Castleman disease ( Fig. 3 ), is a rare benign lymphoproliferative disease that frequently presents as a mediastinal mass. Although it can also affect the cervical and abdominal lymph nodes, approximately 70% occur in the chest, and the visceral mediastinum is the most common location. , ,