Chest radiograph or x-ray is one of the most commonly performed imaging tests. It is a high-yield test, providing significant clinical information rapidly, at low cost, and with low radiation exposure, but many examinations are nonetheless unnecessary. In this chapter, we discuss the indications for chest x-ray, features of the chest x-ray technique and their effect on the resulting image, basic principles of chest x-ray interpretation, and a systematic algorithm for image interpretation. Following this, we present extensive figures with chest x-ray images depicting a spectrum of emergency findings. When available, corresponding computed tomography (CT) images are shown. These clarify chest x-ray findings, demonstrate the limitations of plain chest radiography, and illustrate the occasional need for advanced imaging for further delineation of pathology. This chapter focuses on chest x-ray in the nontrauma patient, although the principles also apply to patients with traumatic injuries, discussed in detail in Chapter 6 .
Chest CT, ventilation–perfusion (VQ) scan, and thoracic ultrasound are discussed in detail in additional chapters focused on chest trauma (see Chapter 6 ), pulmonary embolism and aortic disease (see Chapter 7 ), and cardiac imaging (see Chapter 8 ).
Chest Imaging Modalities
Modalities for thoracic imaging include chest radiograph (x-ray), CT, VQ scan, echocardiography, magnetic resonance (MR), fluoroscopy (used for pulmonary angiography, aortography, and upper gastrointestinal imaging), and positron emission tomography (PET). MR has a limited role in the emergency department currently, largely because of issues of cost and availability, but is used in selected patients in diagnosis of conditions such as pulmonary embolism and aortic disease. PET scanning is used primarily in detection of metabolically active neoplastic disease and is therefore not routinely used in the emergency department. Characteristics of these imaging modalities are shown in Table 5-1 . Issues related to CT and MR such as iodinated contrast nephropathy and allergy, radiation, and gadolinium-associated nephrogenic systemic fibrosis are discussed in Chapter 6 , Chapter 7 , Chapter 8 , Chapter 15 .
| Modality | Applications | Radiation Exposure | Cost | Contraindications |
|---|---|---|---|---|
| X-ray (radiograph) | Broad utility in chest pain, dyspnea, fever, hemoptysis, pneumoperitoneum, foreign body, trauma | 0.01-0.02 mSv (~1 day of background radiation on Earth) | $50-$200 | None. X-ray is safe in pregnancy, with or without abdominal shielding. |
| Computed Tomography (CT) | Pulmonary embolism, aortic disease, coronary artery disease, trauma, abscesses, malignancy, further delineation of chest x-ray abnormalities | 8-20 mSv, depending on factors such as electrocardiogram gating (see Chapter 8 ) | $1000-$2000 | IV contrast used for CT angiography is contraindicated by renal insufficiency or allergy. Radiation dose is high, raising concerns in young patients and women due to breast exposure. |
| Ventilation-Perfusion (VQ) Scan, a nuclear scintigraphy technique | Pulmonary embolism; also used in nonemergent settings to evaluate lung ventilation | 8 mSv | $1000-$2000 | Use in pregnancy and nonpregnant women has been advocated as an alternative to CT, based on some studies showing lower radiation exposures from VQ. Use is contraindicated in patients with extensive lung disease, e.g., COPD or asthma, due to high likelihood of nondiagnostic results in PE evaluation. |
| Echocardiography | Pericardial effusion, cardiac output, valvular abnormalities, congenital heart disease, pulmonary embolism, cardiac arrest | None | $500; bedside echo by emergency physicians may or may not be billed depending on institution | None. |
| Magnetic resonance imaging (MRI) | Aortic disease, pulmonary embolism (not standard) | None | $3000-$4000 | Pacemakers and other electronic devices are a contraindication, although some devices are safe (check device- and MRI machine–specific information). Gadolinium contrast used in many chest applications is contraindicated in patients with severe renal disease. |
| Fluoroscopy | Pulmonary angiography, aortography, esophagography | 4-20 mSv; high radiation exposure due to total duration of “beam on” time | Extremely variable; may exceed $10,000 for aortography or pulmonary angiography if used for diagnosis and treatment | Radiation exposure is a relative contraindication in young patients. Injected contrast agents are contraindicated by allergy or renal insufficiency. |
| Positron Emission Tomography (PET) | Detection of metabolically active neoplastic disease | Not routinely used in emergency department patients | ||
Cost of Chest X-ray
Chest x-ray is one of the most cost-effective imaging examinations, with the cost to patients being between $50 and $220 in many medical systems. However, charges for chest x-ray vary considerably, even in the same geographic region. According to data provided by health care facilities, the U.S. national average cost of chest x-ray was $370 in 2010. Evidence-based guidelines for imaging should be followed when available and applicable, as the total cost of common, individually inexpensive tests such as chest radiography to the U.S. medical system is enormous.
Radiation Exposure From Chest X-ray
Chest x-ray provides minimal radiation exposure, around 0.01 to 0.02 mSv. This is the equivalent of approximately a single day’s background radiation exposure at most locations on the Earth. It equals the additional radiation exposure incurred by taking a commercial airline flight at 35,000 feet from New York to Los Angeles. The radiation exposure is focused and is quite safe in pregnant patients, with negligible scatter radiation to the abdomen and pelvis. Abdominal shielding has not been shown to be necessary to reduce fetal exposure substantially, though it remains a recommended action for the psychological well-being of the pregnant patient. Radiation exposures from chest CT are far greater, by as much as 500 fold, and are discussed in detail in Chapter 7 , Chapter 7 .
Are Health Care Workers at Risk From Chest X-ray Radiation Exposure?
The exposure to health care workers from portable chest x-rays is very low if simple precautions are taken; at a distance of 15 inches from the x-ray beam, the scatter radiation exposure is minimal (around 0.0008 mSv), requiring exposure to more than 1200 radiographs to equal annual background radiation exposure. Multiple other studies suggest trivial exposures to health care workers using simple precautions to distance themselves from the x-ray source. If an appropriate safe distance cannot be achieved because of patient care requirements, simple shielding devices such as thyroid and torso aprons should be used.
Clinical Indications for Chest X-ray
For reasons of time, cost, and radiation exposure as described earlier, best-evidence guidelines should be applied when possible to guide the use of chest x-ray. Chest x-ray is useful in evaluation of a multitude of patient presentations, including cough and fever, chest pain, dyspnea, hemoptysis, weight loss, hypoxia, suspected subdiaphragmatic pneumoperitoneum, even Horner’s syndrome. Given the incredible range of possible patient presentations, no single clinical decision rule can govern chest x-ray use for all cases, and the emergency physician must make clinical decisions for radiography based on the individual patient presentation. However, the value of chest x-ray in common clinical scenarios has been reviewed extensively by the American College of Radiology (ACR) in its appropriateness criteria. These criteria, published on the ACR website, are expert consensus guidelines based on review of medical literature by radiologists and experts from other medical specialties. Summaries of ACR guidelines relevant to thoracic imaging are presented in Tables 5-2 and 5-3 . The ACR rates imaging studies using a 1-to-9 scale, with 9 being “most appropriate” and 1 being “least appropriate.” Although the use of plain chest radiography may appear “harmless” given its speed, relatively low cost and low radiation exposure, collective costs of unnecessary radiographs and other preoperative tests are believed to total in the billions of dollars in the United States annually. Other potential harms from unnecessary radiography include delays in surgery prompted by the detection of incidental findings such as pulmonary nodules and costs and morbidity of additional diagnostic procedures triggered by chest x-ray findings. Consequently, chest x-ray should be ordered with attention to the evidence supporting its use for a given clinical presentation. Unfortunately, as we will review, high quality evidence to guide clinical decisions is lacking for many potential indications for chest x-ray.
| Clinical Scenario | ACR Rating for X-ray Chest | ACR Rating for CT Chest |
|---|---|---|
| Acute chest pain | ||
| 9 | CTA chest (noncoronary): 6—Useful in ruling out other causes of chest pain such as aortic dissection, pulmonary embolism, pneumothorax, pneumonia. |
| 9—Should be performed if readily available at the bedside and does not cause delay in obtaining a CT or MRI. Alternative causes of chest pain may be discovered. Not the definitive test for aortic dissection. | CTA chest and abdomen: 9—Recommended as the definitive test in most patients with suspicion of aortic dissection. |
| 9—to exclude other causes of chest pain | CTA chest (noncoronary): 9—Current standard of care for detection of PE. |
| Chronic chest pain | ||
| 3—Usual initial imaging study in cardiac patients. Although used frequently, chest radiographs can neither establish nor exclude chronic ischemic heart disease. Insensitive for detecting coronary arterial calcification. Limited value in patients with high risk of CAD. | CTA coronary arteries: 7—Very good accuracy and negative predictive value in low to intermediate risk groups. However, may have false negatives in high-risk group, and negative studies may still require further diagnostic testing. Coronary calcification often found in older high-risk patients (especially males) can limit coronary luminal assessment. |
| 9 | CTA chest (noncoronary) with contrast: 8— Important examination for pulmonary embolism and thoracic aortic aneurysm/dissection. To rule out PE and evaluate lung pathology. Appropriate for chronic anginal chest pain.CTA coronary arteries with contrast: 8—Can be used to assess for coronary atherosclerosis,anomalous coronary artery, and pericardial disease. High negative predictive value will exclude coronary artery disease and allow triage management to focus on more likely diagnoses. To eliminate unnecessary catheterizations. |
| CHF | ||
| 9 | 2—readily diagnosed on CT obtained for other indications |
| 4 | |
| Dyspnea—suspected cardiac origin | 8 | CTA coronary arteries: 6CTA coronary arteries with advanced low dose techniques: 6CTA chest (noncoronary): 6 |
| Suspected bacterial endocarditis | 9 | CT heart function and morphology with contrast: 6—Multidetector with maximal temporal and spatial resolution. Probably indicated to rule out paravalvular abscess and/or psedoaneurysm. Emerging technology. |
| Known or Suspected congenital heart disease in the adult | 7 | CT heart function and morphology with contrast: 7. May be alternative to MRI and TTE/TEE. High spatial resolution to evaluate small and tortuous vessels. For evaluation of airway. CTA coronary arteries with contrast: 6. When there is a high index of suspicion for fistula or anomalous coronaries.CTA coronary arteries with contrast with advanced low dose techniques: 6. Important in young to middle-age adults because of reduced radiation dose. |
| Acute respiratory illness | Varies—see Table 5-3 | Varies |
| Acute respiratory illness in HIV-positive patients | 9 | CT chest without contrast: 8—When negative, equivocal, or nonspecific chest radiograph. |
| Chronic dyspnea—suspected pulmonary origin | 8-9—depends on age and examination | Any age, nonrevealing or nondiagnostic clinical, standard radiography, and laboratory studies. In the setting of chronic dyspnea, the most appropriate imaging study is a thin section high resolution chest CT: 9. If a patient has dyspnea not clearly of pulmonary origin, other entities such as chronic or acute pulmonary embolism may need to be excluded. In that setting, a thin section chest CT with intravenous contrast is appropriate. See the ACR Appropriateness Criteria® topic on “Acute Chest Pain–Suspected Pulmonary Embolism”. |
| Hemoptysis,Two risk factors (>40 years old and >40 pack-year history) | 9 | without contrast: 6 (useful for patients with renal failure or contrast allergy.); with contrast: 8 (optimal study shows enhancement of the systemic arteries.) |
| Rib fractures, adult < 65 years of age | 8—PA view. Rib views are given a rating of 2. | 2—with or without contrast |
| Routine admission and preoperative chest radiography | NA | |
| 2 (preoperative or routine) | |
| 9 (preoperative or routine) | |
| 6 (preoperative), 4 (routine admission) | |
| 8 (preoperative or routine) | |
| Routine chest radiographs in uncomplicated hypertension (asymptomatic) | NA | |
| 1 | NA |
| 5 | NA |
| Screening for pulmonary metastases | 8-9—depends on malignancy type | 7-9—depends on malignancy type; noncontrast CT |
| Solitary pulmonary nodule | NA—chest x-ray presumably already performed, demonstrating lesion | CT chest with contrast: 6CT chest without contrast: 8 |
| Staging of bronchogenic carcinoma |
| CT chest with or without contrast (including upper abdomen): 9—CT with contrast is preferred if there are no strong contraindications. |
| Blunt chest trauma—suspected aortic injury | 9 |
|
| Clinical Variant | ACR Rating for X-ray Chest |
|---|---|
| >Age 40 | 8 |
| Dementia, any age | 8 |
| <Age 40, negative physical exam, and no other signs, symptoms, or risk factors | 4 |
| <Age 40, positive physical exam or other risk factors | 9 |
| Complicated pneumonia | 9 |
| Suspected severe acute respiratory syndrome | 9 |
| Suspected anthrax | 9 |
| Febrile, neutropenic | 9 |
| Acute asthma | |
| 4 |
| 9 |
| Acute exacerbation of COPD | |
| 4 |
| 9 |
∗ This is a truncated version of the full ACR Appropriateness Criteria, which also includes ratings for chest CT in each of the Clinical Variants.Reprinted with permission of the American College of Radiology, Reston, VA. No other representation of this material is authorized without expressed, written permission from the American College of Radiology. Refer to the ACR website at www.acr.org/ac for the most current and complete version of the ACR Appropriateness Criteria.
† Acute respiratory illness is defined by the presence of one or more factors including cough, sputum production, chest pain, and dyspnea, with or without fever.
Is a Chest X-ray Routinely Indicated for Chest Pain?
A multitude of causes can result in chest pain, including a number of abnormalities visible on chest x-ray. Pneumonia, pneumothorax, pneumomediastinum, neoplasms, rib fractures, and aortic dissection are just a few of the disease processes that can result in chest pain and that the chest x-ray may diagnose or suggest. Pulmonary embolism may have associated chest x-ray abnormalities such as pleural effusion or pulmonary infarct, although sensitivity and specificity of chest x-ray are too poor for clinical diagnosis of this entity. The ACR recommends chest radiography as the initial imaging study for patients with chest pain and a low probability of cardiac ischemia, given the range of possible diagnoses and the low cost and radiation exposure of chest x-ray. The American College of Emergency Physicians (ACEP) recommended chest x-ray for many scenarios described in its 1995 “Clinical Policy for the Initial Approach to Adults Presenting With a Chief Complaint of Chest Pain, With No History of Trauma,” which has since been retired (all ACEP clinical policies are subject to retirement if a new review of the literature is not conducted within a prescribed time frame). Systematic prospective studies on the value of chest x-ray in undifferentiated chest pain are limited. In a retrospective study of 5000 portable or posterior–anterior (PA) and lateral chest radiographs performed on emergency department patients, Buenger et al. discovered that “serious disease” was found in 35% of those with chest symptoms, based on the x-ray requisition. Requisition information may not reliably characterize a patient’s symptoms, so the validity of this study remains in doubt.
Is a Chest X-ray Routinely Indicated for Stroke?
Sagar et al. performed a retrospective review of 435 patients admitted with a diagnosis of acute stroke. They found that 86% had an admission chest x-ray performed and 77.5% of radiographs were technically inadequate, mostly because of poor patient positioning and poor inspiratory effort. Radiographic abnormalities were seen in 61 patients (16.4% of those undergoing radiography), and clinical management appeared to have been altered in 14 patients (3.8% of those undergoing radiography). The authors conclude that chest radiography is not routinely indicated for patients with acute stroke in the absence of suggestive clinical findings or complaints. As with many other clinical presentations, prospective studies for chest x-ray in stroke are lacking, and little evidence exists regarding the role of chest x-ray in presentations such as altered mental status (rather than obvious stroke syndromes such as hemiparesis).
Is a Chest X-ray Routinely Indicated for Gastrointestinal Bleeding?
Tobin et al. reviewed 202 adult patients admitted to intensive care units for gastrointestinal hemorrhage. They found that 161 (80%) underwent admission chest x-ray. Review of radiographs by a radiologist blinded to the study purpose found 14.3% with minor radiographic abnormalities, none of which resulted in a therapeutic or diagnostic intervention. Major radiographic abnormalities were found in 13% of patients, with an intervention in only 5.6% of patients. Predictors of major abnormalities included history of lung disease and abnormal lung physical examination at presentation (sensitivity = 79%, negative predictive value = 96%). These variables also predicted major radiographic abnormalities associated with interventions (sensitivity 89%, negative predictive value = 99%). The authors found that selective radiography based on these criteria would have reduced the charges from chest radiography by more than $500 per major abnormality detected and by more than $1000 per major abnormality associated with an intervention. These findings have not been externally validated in a separate population but do suggest that routine chest x-ray is unnecessary for this clinical presentation.
Is a Chest X-ray Routinely Indicated for Acute Respiratory Illness With Features Such as Productive Cough?
Acute respiratory illness is defined by the presence of one or more factors, including cough, sputum production, chest pain, and dyspnea, with or without fever. A multitude of studies have examined the role of chest x-ray, examining varying populations including healthy hosts, immunocompromised patients, the elderly, and patients with asthma and chronic obstructive pulmonary disease (COPD). The ACR divides its recommendations based on these and other patient factors as shown in Table 5-3 . Benacerraf et al. studied 1102 outpatients with acute respiratory illness and found that in patients below 40 years 96% of chest radiographs had no acute abnormalities. Limiting radiography in patients below age 40 to those with abnormal physical examination or hemoptysis would have reduced radiography by 58% and missed only 2.3% of acute abnormalities. Fever, cough, and the presence of even purulent sputum were not predictive of chest x-ray abnormalities in patients under the age of 40. Patients above the age of 40 and immunocompromised patients, such as those with HIV or neutropenia, represent higher risk groups for which radiography is recommended by the ACR for signs and symptoms of acute respiratory illness. The emergency physician should incorporate other factors into the decision to obtain radiography and to treat the patient, including access to care, contacts with high-risk (immunocompromised) patients, endemic or epidemic disease, and risk factors for tuberculosis or undiagnosed HIV.
Is a Chest X-ray Routinely Indicated for Suspected Pneumonia?
When the emergency physician specifically suspects pneumonia, is radiography warranted? As discussed earlier, patients below the age of 40 with normal physical examination and no hemoptysis have a very low rate of radiographic abnormalities and should not routinely undergo chest x-ray. Does this mean that patients not meeting these “low risk” criteria must undergo chest x-ray? The ACR points out in its appropriateness criteria for acute respiratory illness some important disparities between clinical practice and professional society recommendations in the diagnosis of pneumonia. The Infectious Diseases Society of America and the American Thoracic Society recommend that chest radiography be obtained when pneumonia is suspected in adults to assist in distinguishing community-acquired pneumonia from other common causes of cough and fever, such as acute bronchitis. However, some clinicians may choose to treat patients empirically for pneumonia, without obtaining radiography, when a high clinical suspicion exists. This is an appropriate evidence-based strategy, as a negative chest x-ray does not rule out pneumonia. When a high pretest probability exists, a negative chest x-ray is inadequate to exclude pneumonia and should not be used as the sole basis to determine management.
Is a Chest X-ray Routinely Indicated for Asthma?
In patients with asthma without features suggesting complications such as pneumonia or pneumothorax, x-ray likely has little role. Findley and Sahn studied 90 adult patients with asthma undergoing chest radiograph and found only 1 patient with an acute infiltrate; normal (55%), hyperinflation (37%), and unchanged minimal interstitial infiltrates (7%) were common patterns. Chest x-ray interpretation did not correlate with admission, suggesting that radiographs in asthma play little role in management. Gershel et al. studied 371 consecutive children over the age of 1 year with first-time wheezing and found that 94.3% had x-ray findings compatible with uncomplicated asthma (negative). Of the 371 patients studied, 21 (5.7%) had positive chest x-ray findings, including atelectasis and pneumonia in 7, segmental atelectasis in 6, pneumonia in 5, multiple areas of subsegmental atelectasis in 2, and pneumomediastinum in 1. Although the authors call this 5.7% “positive,” it is not clear that findings of atelectasis or even pneumomediastinum alter management in a clinically beneficial way, as no specific therapy is required for these abnormalities. When only pneumonia is considered a positive finding, only 3.9% of first-time wheezing children had positive chest x-rays. The authors found that 95% of abnormal x-rays could be predicted based on the presence of tachypnea greater than 60 breaths per minute, pulse greater than 160 beats per minute, and localized rales or diminished breath sounds, before or after treatment—suggesting that even in first-time pediatric wheezing, routine x-ray may not be necessary. Aronson et al. reported on 125 consecutive adult asthma admissions and found no chest x-ray abnormalities in 81 patients characterized as uncomplicated, defined as having a prior history of asthma, presenting with dyspnea and wheezing, and having no history of fever or chills, immunosuppression, cancer, intravenous (IV) drug abuse, prior thoracic surgery, cardiac disease, or pulmonary disease such as COPD or granulomatous disease.
Is a Chest X-ray Routinely Indicated for Chronic Obstructive Pulmonary Disease?
In COPD, the ACR recommendations are somewhat at odds with work published in the emergency medicine literature. According to the ACR, in COPD patients without fever, leukocytosis, chest pain, or history of congestive heart failure (CHF) or coronary artery disease (CAD), chest x-ray has a low diagnostic utility, with an appropriateness rating of four out of nine. These recommendations are concordant with the work of Sherman et al., who retrospectively reviewed 242 patients with admission chest x-rays for COPD. Although 14% had radiographic abnormalities, the authors concluded that only 4.5% had appropriate and clinically important management changes resulting from chest x-ray findings. They found that leukocytosis (white blood cell count >15 × 10 9 per liter and neutrophil count >8 × 10 9 per liter), history of CHF or CAD, chest pain, and extremity edema were a “high risk” criteria predictive of important acute radiographic abnormalities.
Emerman and Cydulka attempted to validate these high-risk criteria in a retrospective series of 685 emergency department visits for acute COPD in which a chest x-ray was performed. In this series, 16% of patients had significant chest x-ray abnormalities: 88 with new infiltrates, 2 with new lung masses, 1 with pneumothorax, and 20 with evidence of pulmonary edema. Abnormalities were associated with history of CHF, fever, rales, pedal edema, and jugular venous distension. In contrast, white blood cell count, temperature in the emergency department, history of CAD, and complaints of chest pain or sputum production were not associated with chest x-ray abnormalities. The sensitivity of the high-risk criteria was only 76%, with a specificity of only 41%. The authors recommended routine x-ray in patients with apparent acute COPD.
Is a Chest X-ray Routinely Indicated for Asymptomatic Hypertension?
According to the ACR reviews, patients with asymptomatic hypertension should not routinely undergo chest x-ray, although debate remains about the value of radiography with rising levels of hypertension. In theory, chest x-ray might be of value in patients with asymptomatic hypertension by revealing cardiomegaly or left ventricular hypertrophy, aortic enlargement or coarctation, or pulmonary vascular congestion. Although a number of authors have suggested these benefits, others have argued that the management of hypertension is not positively influenced by chest x-ray findings; regardless of the presence or absence of chest x-ray abnormalities, blood pressure control should be attempted. Moreover, studies indicate that chest x-ray is insensitive and nonspecific for detection of left ventricular hypertrophy, which is better assessed with echocardiography.
Is a Chest X-ray Routinely Indicated for Admitted Patients?
The ACR found that routine admission or preoperative chest x-ray is not indicated in the patient with no cardiopulmonary findings by history or physical examination, with an appropriateness of two out of nine. In patients with acute cardiopulmonary findings by history or physical, chest x-ray is highly appropriate, with a rating of nine. Because of a lack of evidence against the use of routine chest x-ray, in elderly patients (70 years or older) with chronic cardiopulmonary disease, the ACR rates chest x-ray as appropriate (eight of nine), even in the absence of history or physical findings, when a chest x-ray has not been performed within the past 6 months. The value of chest x-ray is less clear in this asymptomatic group when a recent chest x-ray (within 6 months) is available for review (ACR rating four to six of nine).
What is the basis for the ACR recommendations in these cases? The ACR cites multiple studies addressing the utility of routine chest x-ray in patients admitted for gastrointestinal hemorrhage, acute stroke, and other conditions affecting the elderly. Gupta, Gibbins, and Sen prospectively studied 1000 consecutive admissions to an acute geriatric ward and found that 35% to 50% of patients had no apparent indication for chest x-ray. In this group without chest x-ray indications, 5.5% had radiographic abnormalities but less than 1% had clinically important findings. In an exhaustive systematic review, Munro, Booth, and Nicholl found that no randomized controlled trials of the effectiveness of routine preadmission or preoperative chest x-rays had been published, and most studies do not (or cannot, because of study design) assess the effect on clinical management of detected radiographic abnormalities. Estimates of the frequency of changes in clinical management suggest patient benefit in fewer than 5% of cases.
We have discussed some common evidence-based indications for chest x-ray. Now we consider basic principles of chest x-ray technique relevant to emergency medicine and key principles of image interpretation. We finish with findings of specific pathology and a systematic approach to the interpretation of chest x-ray.
Interpretation of Chest X-ray: Basic Principles
Interpretation of chest x-ray requires several elements:
- •
An understanding of some basic principles and terminology of radiography
- •
A knowledge of the normal appearance of the chest x-ray
- •
A familiarity with the appearance of important pathologic findings
- •
A systematic approach to allow all key findings to be observed
We start with some basic principles. If you are already familiar with basic radiographic concepts and the normal appearance of the chest x-ray, move on to the section on pathology. But a few minutes familiarizing yourself with the ideas in this section can pay big dividends some day when you encounter an unfamiliar chest x-ray that does not fit any of the patterns of common pathology discussed in this chapter. Understanding basic principles may allow you to deduce the nature of the abnormality and avoid common pitfalls. A number of outstanding texts are devoted entirely to chest radiography. We have drawn on numerous sources for the following discussion.
Chest X-ray Techniques and Effects on Resulting Images
Familiarity with the chest x-ray technique is important for the emergency physician because it can affect the appearance of important diagnostic findings and the sensitivity and specificity of a chest x-ray for these diagnoses. In some cases, a given chest x-ray technique may falsely simulate pathology; in other cases, the technique many hide important abnormalities. Here, we describe the most common chest x-ray techniques, along with pitfalls of each. The most common variations of the frontal chest x-ray used in the emergency department are the anterior–posterior (AP) view and the PA view. The AP view is commonly performed as a portable study in the patient’s room, and it may be performed with the patient in a supine or upright position. The PA frontal view is performed in the radiology suite, and often a lateral projection PA chest x-ray is obtained at the same time.
Chest x-rays can be characterized by the following:
- •
The direction of the x-ray beam as it passes through the patient (posterior to anterior or anterior to posterior)
- •
The distances between the x-ray source, the patient, and the x-ray detector
- •
The position of the patient (upright, supine, lateral decubitus, lordotic, or oblique)
- •
The x-ray exposure
Each of these variables has important effects on the resulting image. In some cases, the technique is manipulated intentionally to achieve a desired diagnostic effect. In other cases, the patient’s clinical condition limits the x-ray technique, and we are forced to accept a suboptimal diagnostic image. Understanding the effects of the x-ray technique on the resulting image is invaluable in helping the emergency physician to avoid misinterpretation of the image and misdiagnosis of the patient.
Direction of the X-ray Beam With Respect to the Detector: Posterior–Anterior Versus Anterior–Posterior Technique
When a frontal projection chest x-ray is obtained, the direction of the x-ray beam as it passes through the patient to the detector can be from patient posterior to anterior or from patient anterior to posterior ( Figure 5-1 ). To restate this another way:

In the case of the PA x-ray, the patient is oriented with the x-ray film or detector in contact with the anterior surface of the thorax. The x-ray beam passes through the patient’s posterior thorax to the detector. The patient faces toward the detector, away from the x-ray source ( Figure 5-1 ).
In the case of the AP x-ray, the patient is oriented with the x-ray film or detector, in contact with the posterior surface of the thorax. The x-ray beam passes through the patient’s anterior thorax to the detector. The patient faces toward the x-ray source, away from the detector.
Why does the direction of the x-ray beam through the patient to the x-ray detector matter? We return to this question in a moment, but first we describe a characteristic difference in the PA and AP x-ray techniques, which contributes to differences in the resulting image.
The PA chest x-ray is typically acquired in a radiology suite, with the x-ray source positioned 6 feet from the x-ray detector. In comparison, the AP chest x-ray is typically acquired as a portable examination, with only 3 feet separating the x-ray source and the detector.
Distance of the X-ray Beam With Respect to the Detector: Posterior–Anterior Versus Anterior–Posterior Technique
Why does the distance from the x-ray source to the detector matter? The answer becomes obvious if we consider the analogy of the children’s game of creating shadows on a wall with a light source ( Figures 5-2 and 5-3 ). You can try our example at home (use a light, not an x-ray) if it is unclear. Place a lamp 6 feet from a wall. Position your hand close to the lamp, and the shadow on the wall appears larger than the actual size of your hand, though rather indistinct at its edges. Most people have used this trick for their amusement, making a hand appear as large as a head, for example. Without moving the lamp, move your hand closer to the wall; the resulting shadow becomes smaller (closer to its actual size), denser (darker), and sharper at its edges. We have now defined one of the two variables: with a light (or x-ray) source a fixed distance from a detector, positioning the object to be imaged close to the detector results in a sharper, truer image without false magnification. Therefore, when performing medical imaging, we should place the body part to be imaged as close to the detector as possible to achieve a sharp, unmagnified image. Now consider the same scenario, with a twist. Imagine that you cannot put your hand close to the wall because of furniture obstructing your path. How can you sharpen the image and reduce magnification? The answer is simple: move the lamp farther from your hand and the wall. Try this experiment, and you will find that the shadow of your hand becomes sharper and less magnified, truer to its actual size. Why would this scenario occur with medical imaging? Imagine placing your thorax against the wall; your ribs prevent you from moving your internal organs closer to the wall, although by facing toward or away from the wall you can position your heart (anterior in your chest) closer to the wall. However, you can easily control the distance of the light source from the wall to reduce magnification and improve the image sharpness. Therefore, when performing medical imaging, we should position the x-ray source as far as possible (within reason) from the body part to be imaged, to improve sharpness and reduce magnification.


Magnification: Asset or Artifact?
Although magnification may sound advantageous, it can introduce clinically deceptive artifacts if some body parts are magnified to a greater degree than others. The most clinically accurate x-ray has no magnification. Why would we not want magnification? Wouldn’t magnification potentially assist in identifying pathology? The problem again can be resolved by considering the shadow experiment. In medicine, we need an accurate estimate of the actual size of body parts and an accurate measure of their size relative to one another. Consider the example of the hand shadow and the shadow of the human head. Although it may be amusing to have your hand mimic a giant dog biting a head, this scenario of varying magnification is highly undesirable in medical imaging. We need to know whether a mass has increased in size or if the heart and mediastinum are enlarged relative to the surrounding thoracic cavity. Our imaging technique must avoid false magnification, particularly when differential magnification of objects in the same image might occur.
Hopefully, we have convincingly demonstrated how the distances from the body part to the detector (assuming a fixed light source) and from the light source to the body part (assuming a fixed distance from body part to detector) affect the resulting images. Let’s briefly consider why this is the case. Figures 5-4 through 5-6 illustrate the effect of distance on object magnification. The angle created between the x-ray source and an object’s edges is determined by the distance from the x-ray source to the object. The shorter the distance from the x-ray source to the object, the greater the angle. Greater angles lead to greater magnification; therefore, a shorter distance from the x-ray source to the imaged object leads to increased magnification. Although increased magnification may appear to be a benefit, in most clinical scenarios this can lead to a false appearance of cardiomegaly or a widened mediastinum. This is particularly a problem if very short distances are employed, as thoracic structures nearer to the x-ray source will be magnified to a greater degree than structures farther from the source. Positioning the x-ray source at a greater distance from the object to be imaged reduces this relative magnification, leading to a truer representation of the object. The AP portable examination places the x-ray source 3 feet from the patient, rather than the 6 feet usually used for a PA examination. Consequently, the AP portable chest x-ray examination typically has a greater degree of false magnification of the heart and mediastinum compared with the PA technique.



We asked earlier why the direction of the beam (AP vs. PA) matters to the resulting image. As we described in our shadow experiment earlier, sometimes we cannot fully control the location of objects relative to the x-ray detector. We cannot change the anterior location of the heart in the chest; however, by positioning the detector on the anterior aspect of the chest, we bring the heart and the detector closer together, reducing magnification of the heart. Consequently, the PA x-ray technique results in less magnification of anterior structures compared with the AP technique. Remember this when viewing AP portable chest x-rays, which are more prone to false appearance of cardiomegaly or mediastinal widening ( Figure 5-7 ).

Patient Positioning for Chest X-ray: Supine Versus Upright Technique
Now let’s also consider the differences among a true upright, a supine, and an intermediate lordotic x-ray ( Table 5-4 ). Although occasionally other positions of the patient are desirable in specific clinical scenarios, for most applications, a perfectly upright patient position is desirable during chest x-ray acquisition. When the patient is positioned perfectly upright and perpendicular to the direction of the x-ray source, thoracic structures are positioned equal distances from the x-ray source, ensuring equal magnification on the resulting x-ray image. In comparison, if the patient is positioned in a lordotic position with the upper and lower thorax at different distances from the x-ray source, the magnification of superior thoracic structures will differ from that of the lower thoracic structures.
| Supine Chest X-ray Finding | Possible Misdiagnosis |
|---|---|
| Heart appears large | Simulates cardiomegaly |
| Mediastinum appears wide | Simulates aortic or mediastinal abnormality |
| Increased vascular markings in upper lung zones | Simulates pulmonary edema |
| Poor inspiration | Simulates pulmonary edema and may hide small nodules |
| Layering of fluid in plane of x-ray detector | May prevent recognition of pleural fluid |
| Distribution of air to anterior chest and abdomen | May prevent recognition of pneumoperitoneum or pneumothorax |
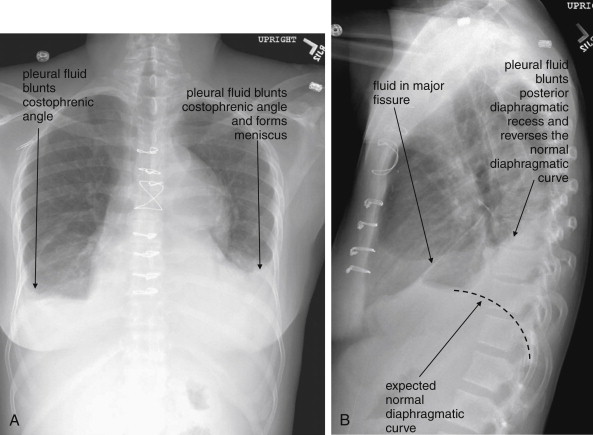
When the patient is positioned in a fully upright position, fluid within structures of the chest will generally reside due to gravity in a dependent position, forming fluid levels. In comparison, if the patient is positioned supine, the horizontal plane in which fluid will spread is parallel to the x-ray film or detector beneath the patient, and no fluid levels will be seen. (see Chapter 6 , Figure 6-2 ). The resulting appearance may be a diffuse increase in the density of the entire affected thorax, sometimes called a “veiling opacity.” This may be mistaken for an increased parenchymal density, rather than being recognized as a broadly layered pleural effusion.
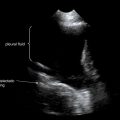
Pleural fluid and fluid within collections such as lung abscesses typically are not visible on an image obtained with a supine patient but are visible on an image obtained with an upright position. In a patient positioned upright, the upper surface of fluid within the potential pleural space usually forms a curved line, higher along the lateral chest wall than at its intersection with the mediastinum. This appearance is termed the meniscus sign ( Figures 5-8 and 5-9 ). In contrast, fluid within an air-filled cavity usually forms a straight horizontal line without a lateral meniscus. Examples include fluid within an air-filled abscess cavity or fluid within a hemopneumothorax ( Figure 5-10 ; see also Figure 5-8 ).


In an upright patient, air within the peritoneal cavity collects beneath the diaphragm, making it visible on x-ray because of the contrast between the density of air and diaphragmatic soft tissue. ( Figures 5-11 through 5-14 ). In a supine patient, air within the peritoneal cavity may collect in the midline anterior abdomen rather than in a subdiaphragmatic position and in addition will spread in the same horizontal plane as the x-ray detector beneath the patient, preventing the air from being visible. An x-ray obtained with an upright patient position is therefore more sensitive for detection of pneumoperitoneum.




Another advantage of the upright chest x-ray is that the patient typically is able to accomplish a greater degree of inspiration. With greater inspiration, blood vessels become more widely spaced, allowing other abnormalities to be recognized. Radiologists have sometimes compared this with seeing a bird in a tree, which is more easily accomplished in a tree with widely spaced branches. Small lung masses can be more easily seen as a result. A well-expanded chest on an upright view also gives a truer estimate of the heart size. Rarely, a chest x-ray obtained at end-expiration provides diagnostic benefits, which we will review later.
In comparison, a supine chest x-ray is usually characterized by higher diaphragms, with poorer lung expansion and clumping of pulmonary vessels. In addition, blood flow to the upper lung is relatively increased by gravity, and the heart appears larger than on an upright x-ray. This is due to the portable AP technique but also because of the relatively unexpanded chest, which makes the heart appear comparatively larger. In combination, these findings may mimic CHF (see Figure 5-7 ).
Supine and semilordotic views are often obtained in emergency department patients not by design but because patient condition prohibits the desired technique. Trauma patients, unstable medical patients, and intoxicated or neurologically disabled patients are just a few examples of patients who often undergo imaging in supine or semilordotic positions because of their inability to be positioned upright. The emergency physician must be aware of the limitations of these examinations and should recognize the patient position when interpreting the resulting images.
Lordotic and decubitus positions are sometimes obtained intentionally for specific diagnostic purposes, described later under “Additional Chest X-ray Views.”
Normal Appearance of a Chest X-ray
Recognizing pathology requires a strong familiarity with the normal appearance of the chest x-ray. Classic abnormalities are often recognized by their distinct differences from the normal chest x-ray appearance. More subtle abnormalities may be missed by an inexperienced observer, but an experienced reader may immediately recognize that the chest x-ray differs from the norm even before characterizing and articulating the abnormality. Radiologists understand that recognizing familiar patterns of normal and abnormal requires constant exposure, much like recognizing a familiar relative. This pattern recognition approach has been dubbed the “Aunt Minnie” effect for this similarity. We briefly describe the appearance of a normal chest x-ray here. In the sections that follow, we present numerous examples of important pathology. Even with several examples of each type, you will only begin the exposure required to “recognize Aunt Minnie.” Make a habit of looking at your patients’ chest x-rays, even if the radiologist has already rendered an interpretation.
Frontal (Posterior–Anterior or Anterior–Posterior) Upright Chest X-ray View
The normal frontal upright chest x-ray ( Figure 5-15 ) has the following features:
- •
The airway is midline.
- •
The bones show no fractures or lytic lesions.
- •
The cardiac silhouette occupies less than half of the transverse diameter of the thoracic cavity. The cardiac silhouette is crisp, with no adjacent pleural fluid or parenchymal opacities to disrupt the normal silhouette. The mediastinal width is less than 8 cm, and the aortic knob is well defined. The normal appearance of the aorta is described in detail in Chapter 6 , Chapter 7 .
- •
The hemidiaphragms are visible as smooth curves bilaterally. No air is seen beneath them; the upper surface is not obscured by pleural effusion or infiltrates. The costophrenic angles are not blunted by pleural effusions. The right diaphragm is slightly higher than the left.
- •
The lung fields are clear, without opacities to suggest pleural effusion, parenchymal disease such as infectious infiltrate, or mass lesion. Lung vascular markings are visible to the periphery, without evidence of pneumothorax. Lung markings are less prominent in the lung apices because gravity diverts blood flow to the lung bases.
- •
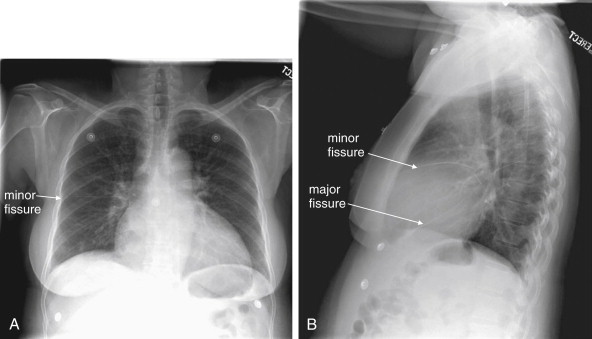
The minor fissure is invisible or subtle in appearance, without significant thickening to suggest fluid accumulation in this potential space.

Lateral Upright Chest X-ray View: Retrosternal Space, Retrocardiac Space, and the Spine Sign
The lateral chest x-ray provides important diagnostic information. Unfortunately, this view is usually not obtained when a portable x-ray examination is performed—another good reason to send the patient to the radiology suite for imaging if the clinical condition permits this. The lateral view ( Figure 5-16 ) reveals the retrosternal space, which overlies the heart and mediastinum on a frontal projection. This space is usually quite lucent (black) because of the presence of a low-density epicardial fat pad and sometimes lung segments—but when occupied by a soft-tissue mass, this space may appear radiodense (white) ( Figure 5-17 ). The lateral chest x-ray also reveals the retrocardiac space. This space normally should be quite lucent (black) (see Figure 5-16 ). Lower lobe pneumonias may be evident on the lateral view as an abnormally dense retrocardiac region ( Figure 5-18 ). On the lateral view, the diaphragms usually form smooth curves descending from anterior to posterior. The space above the diaphragms is usually lucent (black), as it contains low-density lung tissue. Pleural effusions may be evident on lateral view as dense (white) layering opacities replacing the normal curve of the diaphragm in this space (see Figure 5-9 ). Sometimes pleural effusions form a meniscus against the posterior wall of the thorax, actually reversing the normal curve of the diaphragm. In addition, air beneath the diaphragm (pneumoperitoneum) may be visible on the lateral view (see Figure 5-14 ).



The thoracic spine also is visible on a lateral chest x-ray. The normal appearance of the spine is a gradually more lucent (blacker) appearance moving from cephalad to caudad (see Figure 5-16 ). This is not a result of decreasing spinal density but rather is a normal artifact of the examination technique. When this progressively more lucent appearance is lost, it implies the presence of an abnormal density in the retrocardiac space. This is called the spine sign and is a pathologic abnormality that can be a clue to disease. Remember that the increasing density has a differential diagnosis, including infectious infiltrate, pulmonary edema, pleural effusion, mass, and atelectasis. Other radiographic findings and the patient’s clinical presentation must be used to sort through this differential diagnosis, and additional imaging may be necessary. Nonetheless, this finding can confirm a pneumonia not seen on the frontal projection x-ray. The lateral x-ray is often neglected but is a key additional view that should be obtained whenever possible and carefully reviewed.
Additional Chest X-ray Views
Early radiologists became extremely skilled at deducing clinically relevant information from additional chest x-ray views. The advent of cross-sectional imaging with CT scan has made some of these views less common, as CT imaging is able to determine the three-dimensional location of objects with high accuracy. However, in some cases, the detailed information provided by CT is unnecessary, and more limited information from x-ray may be sufficient for clinical action such as draining a pleural effusion. Although CT could be used in this scenario, the radiation dose from CT is approximately 500 times that of a single additional chest radiograph, and the cost is approximately 10 times higher. In addition, for chest angiography protocols, chest CT requires IV contrast administration, with risks of allergy or contrast nephropathy. X-ray avoids these issues. On an individual basis, the emergency physician should consider the information needed for clinical decision-making. If it is obvious that CT will be required for specific information, such as evaluation of pulmonary embolism or aortic pathology, additional x-rays should generally not be obtained. However, if the information required can be provided by radiographs, these are a better choice for reasons of cost, radiation, and contrast exposure.
Lateral Decubitus Views
Lateral decubitus views allow assessment of radiographically visible mobile fluid collections and foreign bodies, as well as inferences about the presence of radiographically invisible foreign bodies. In a lateral decubitus view, the patient is positioned with one side of the thorax (right or left) in a dependent position ( Figures 5-19 and 5-20 ). The view is labeled based on the side of the chest that is dependent. Thus an x-ray obtained with the patient positioned with the left side of the thorax in a dependent position is a “left lateral decubitus” view. Usually, an x-ray is then obtained using an AP projection, as described earlier.


Decubitus views can be useful in the following scenarios:
- •
Differentiation of parenchymal consolidation from pleural effusion. When opacities are visible on an upright or supine frontal projection (PA or AP), a lateral decubitus view can differentiate parenchymal consolidation from pleural effusion. A parenchymal opacity will not change in position relative to the patient when the patient is moved to a lateral decubitus position. A pleural fluid collection will layer with gravity if it is not loculated. (see Figures 5-19 and 5-20 ). Alternative means of differentiating these two entities include ultrasound, which readily identifies fluid as a hypoechoic collection, and CT.
- •
Identification of loculated versus freely mobile pleural fluid collections. When pleural fluid is seen on standard radiographic views, decubitus views can reveal mobile collections which will maintain their position relative to the patient. Ultrasound and CT can also differentiate these.
- •
Identification of mobile radiopaque foreign bodies. In some cases, the location of a visible foreign body may be further delineated by repositioning the patient. Airway foreign bodies may shift from the midline, while foreign bodies within the esophagus will remain midline despite dependent positioning.
- •
Identification of radiolucent (invisible) foreign bodies. Some aspirated foreign bodies such as peanuts or plastic are too low in density to be visible on standard radiographs. Their presence can sometimes be deduced from their effect on pulmonary inflation. A foreign body lodged in an airway can occlude the airway in a ball-valve fashion, trapping air within a lung segment and causing relative hyperinflation. Normally, when a patient is placed in a lateral decubitus position, the dependent lung appears hypoinflated because of the weight of other thoracic structures resting upon it. In the case of an obstructing airway, foreign body, air trapped within a lung segment will make the affected lung remain fully inflated despite lateral decubitus positioning. Of course, a foreign body that does not create a ball-valve effect would not be detected by this method.
- •
Identification of a pneumothorax in a patient who cannot tolerate an upright position. Remember that a pneumothorax may not be visible on a supine x-ray, because air will collect in the anterior chest rather than near the thoracic inlet or lung apices. The patient should be placed in a decubitus position opposite to the side of the suspected pneumothorax (hemithorax with the suspected pneumothorax should be higher), allowing air within the pleural space to rise to the lateral chest wall. As a consequence, the pleural line and absence of lung markings typical of pneumothorax may then be more visible.
Lordotic Views
In a lordotic view, the patient is positioned in a semiupright position relative to the x-ray source. The resulting image provides more detail of upper thoracic structures by altering the position of upper ribs and clavicles relative to the upper lung. Normally, these bony structures obstruct the view of the upper lung; a lordotic position can allow assessment of upper lung masses, infiltrates, or apical abnormalities such as pneumothoraces. The ribs appear more horizontal on a lordotic view. Although lordotic positioning is sometimes used for intentional diagnostic benefit, often lordotic positioning is an undesired effect in an emergency department patient undergoing an AP portable chest x-ray. Artifacts of this technique include magnification of the heart and mediastinum, simulating cardiomegaly, mediastinal mass, or aortic aneurysm. In addition, lordotic positioning may increase the amount of soft tissue projected over the lower abdomen, resulting in increased apparent opacity at the lung bases. This may be mistaken for basilar infectious infiltrates, atelectasis, or pulmonary edema.
Oblique Views
Oblique views, though rarely used in the emergency department, use parallax to identify the location of objects or structures within the lung. Two structures that overlie each other on lateral or frontal projection views can be distinguished by an oblique view. More often in the emergency department, an oblique view is inadvertently obtained because of rotation of the patient. This commonly occurs when AP x-rays are obtained in patients with kyphosis, contractures, or neurologic deficits. Rotated or oblique views can introduce a number of artifacts. Among them, the mediastinum often appears artifactually widened, and the heart and mediastinum may appear shifted ( Figure 5-21 ). Opacities in the lung bases may be hidden if the cardiac silhouette is projected over them because of rotation.

Phase of Respiration: Inspiratory and Expiratory Views
Normally, a chest x-ray is obtained at full inspiration, although in many cases patients may fail to inspire deeply because of chest pain or may be unable to hold their breath in this position because of dyspnea. The chest x-ray captures a frozen, static moment in time, but thoracic structures are actually in motion with patient respiration and cardiac activity. The apparent density of lung tissue varies with the phase of respiration. At end-expiration, lung volumes are very low, the diaphragms appear high, and lung tissue appears dense (whiter). Blood vessels in the lungs appear crowded together, contributing to the apparent density of lung tissue. If the phase of respiration is not considered, this appearance may be mistaken for pulmonary edema. In contrast, a chest x-ray obtained at end-inspiration shows well-inflated lungs, diaphragms that have descended fully, and widely spaced pulmonary vessels. Lung parenchyma under these conditions appears less dense. A clue to the phase of respiration is the number of ribs visible. A rule of thumb is that the diaphragm should lie at the level of the posterior 8th to 10th rib for an adequate respiratory effort in a good-quality chest x-ray. Abnormally dense lung parenchyma such as seen with pneumonia is more visible against the backdrop of fully inflated and lucent (black) lungs. Patients with COPD or asthma may have hyperexpanded lungs with low-density parenchyma and more than 10 ribs visible.
Occasionally, chest x-rays are intentionally obtained in other phases of respiration. Examples include:
- 1.
Pneumothorax: An end-expiratory chest x-ray is sometimes used to exaggerate the appearance of a pneumothorax. At end-expiration, lung parenchyma is deflated and occupies a smaller percentage of the thorax. In contrast, air in the pleural space will not decrease in volume during expiration. As a consequence, a pneumothorax will appear to occupy a larger percentage of the thoracic cavity.
- 2.
Foreign body. Low-density foreign bodies may be difficult to see on chest x-ray. However, they may exert a ball-valve effect by lodging in small airways. As a consequence, they may trap air within lung segments. During end-expiration, a normal lung will decrease in size. A lung with a partially obstructed bronchus due to a foreign body exerting a ball-valve effect may be unable to deflate during expiration. The abnormal lung or lung segment will therefore appear larger and more lucent than the normal side on an end-expiratory radiograph.
Chest X-ray Exposure (Penetration)
The exposure of chest x-ray is particularly critical, as the chest contains structures across a wide range of densities, from air to bone. A fully exposed x-ray film or detector results in a completely black image. For example, x-ray passes readily through air outside the patient, making the background around the patient appear black. Lungs are normally composed mostly of air, with a small density contribution from pulmonary blood vessels, and appear nearly black with a good exposure. A pneumothorax is even less dense, like air outside of the patient, and appears almost completely black. Metals and bones prevent transmission of much x-ray to the detector with a normal exposure; consequently, the detector is not exposed and the image appears white.
From these principles, we can extrapolate that an overexposed chest x-ray will appear black (the entire detector is fully exposed). An underexposed chest x-ray will appear nearly white, as the detector is not exposed. Although the appearance of tissues can be adjusted (brightness and contrast) on a digital picture archiving and communication system (PACS) display, no amount of image manipulation can overcome a badly over- or underexposed image. For example, in a badly overexposed image, the entire detector is fully exposed, and all pixels are completely black. Adjusting the contrast and brightness simply makes the entire image blacker or whiter, without revealing tissue detail. In a badly underexposed image, the entire detector fails to be exposed, and all pixels are completely white. Adjustment of brightness and contrast is not useful in this case, either. As the exposure level is increased above an “optimal exposure,” lung tissue becomes “burned out” (black), and fine details of lung architecture, such as bulla, fissures, pulmonary vascularity, and pneumothorax, are lost. This loss comes with a gain in the visibility of bony detail, as x-ray can now penetrate through less dense regions of bone, including fracture zones. If exposure is lower than an “optimal level,” detail of bone is lost, as no x-ray can penetrate through dense bone. At the same time, soft tissues become more visible, as the lower exposure prevents x-ray from fully exposing the detector behind them. Depending on the clinical presentation, overexposure- or underexposure may be intentionally performed to highlight either bone or soft-tissue detail.
Beware of some common scenarios in which poor exposure can simulate pathology:
- •
In the obese patient, underexposure is common. The detector is in effect “shielded” from the x-ray beam by overlying soft tissues. Consequently, the entire image, including lung tissues, appears brighter. This appearance can be mistaken for pulmonary edema if the overall image exposure is not considered.
- •
On a frontal projection, underexposure makes retrocardiac structures less visible, including the left lower lobe, which extends behind the heart.
- •
On a frontal projection, breast tissue can increase attenuation of the x-ray beam in the lower lung zones, resulting in relative underexposure of the lung in these regions. Consequently, the lower lung fields may appear brighter white, simulating bibasilar infiltrates, pulmonary edema, or atelectasis.
- •
In a patient with a unilateral mastectomy, an asymmetrical appearance of the lower lung zones can lead to false diagnosis of unilateral basilar pneumonia. Pay attention to this finding to avoid misdiagnosis. Use other x-ray findings such as the silhouette sign (described later) to assess for pathology in the lower lung fields.
Artifacts Outside of the Patient
Opacities and lucencies on the x-ray image may not originate within the patient ( Figures 5-22 through 5-25 ). Overlying skin folds and clothing can create lines simulating pathology such as the pleural line of pneumothorax. Foreign bodies may appear to lie within the patient but may be discerned to be outside of the patient if a lateral view is obtained. Two orthogonal views are needed to confirm an object’s location. Whenever possible, extraneous external foreign bodies should be removed before obtaining x-ray to prevent them from obscuring internal anatomy or being mistaken for objects within the patient. Sometimes surface anatomy such as nipples can simulate internal pathology such as lung nodules. When this is in doubt, marking the surface anatomy with a radiopaque tag can clarify the source of the chest x-ray finding.




Tissue Densities and Chest X-ray
On chest x-ray, only four basic tissue densities are present ( Box 5-1 ): air, fat, water (typical of both fluids and most solid body organs, such as liver and muscle, which are more than 90% water), and metal. This includes calcium, which you will recall from chemistry class is actually a metal. Some texts distinguish bone from metal. A key principle of chest x-ray interpretation is that two tissues in direct physical contact with each other and sharing the same basic density are indistinguishable. For this reason, the diaphragm cannot normally be discerned from the abutting liver, because both are of water density. Similarly, blood (water density) cannot be seen within the heart (also water density). Pericardial fluid cannot be distinguished from myocardium, because both are of water density, but pericardial air can be distinguished from myocardium. A pleural effusion lying above the diaphragm shares the same density with the subjacent diaphragm and liver and cannot be directly distinguished. FLOAT NOT FOUND
Normal lung tissue (air density) can be distinguished readily from adjacent normal soft tissues (water density), because the two tissues do not share the same density. However, when normally air-filled alveoli become filled with water density, the border between that lung tissue and the adjacent heart disappears. The cause of the alveolar water density does not matter: pneumonia, hemorrhage, and aspirated material all have the same effect, increasing the density of the lung from air to water. Consequently, if the abnormal lung abuts another water density structure such as the heart or diaphragm, the junction between the two tissues cannot be discerned. A little anatomy illustrates the clinical utility of this fact. The right middle lobe abuts the lower right heart border, and the right lower lobe abuts the diaphragm. For this reason, a right middle lobe pneumonia obscures the lower right heart border, and a right lower lobe pneumonia obscures the diaphragm. On the left side, the lingula abuts the lower left heart border, and the left lower lobe abuts the diaphragm. Pneumonias in these areas obscure the left heart border and left diaphragm, respectively.
Silhouette Sign
As we have just discussed, two objects with the same basic chest x-ray density (air, fat, water, or metal/bone) abutting one another cannot be distinguished. The corollary of this is that when two structures of different basic density abut each other, their junction can be discerned on x-ray. This is called the silhouette sign ( Figure 5-26 ). Loss of the normal silhouette sign along the heart borders or diaphragm is an important pathologic finding, though it does not always signify pneumonia, as we explore further. Any change that causes the abnormal juxtaposition of two tissues of the same density results in loss of the silhouette sign. Examples include a solid lung mass adjacent to the heart, a pleural effusion adjacent to the heart or diaphragm, dense pulmonary edema, or atelectasis of lung adjacent to the heart or diaphragm, in which case the “collapse” of lung tissue results in it taking on a solid organ density (water density). Box 5-2 outlines the differential diagnosis of increased lung parenchymal opacity.

In other cases, the pathologic change is not the loss of the silhouette sign but rather the introduction of a silhouette sign when one is not normally present. This occurs when a new, abnormal tissue density becomes interposed between two adjacent tissues that share the same density and normally are not distinguishable. For example, the junction between the liver and the diaphragm is not normally visible, as the two tissues are in direct physical contact and share the same density (water or soft-tissue density). If pneumoperitoneum develops, air can become interposed between the liver and the diaphragm, making this interface visible (see Figure 5-11 ). Air can also cause a pathologic silhouette sign when it is present within the pericardium (between the soft tissues of the heart and the pericardial sac) or in the abdomen when air is visible between two adjacent loops of bowel, making the external surface of the bowel wall visible.
- •
Air: black or dark
- •
Fat: dark gray
- •
Water (blood, soft tissue): bright gray
- •
Metal or bone: white
Air Bronchograms: A Manifestation of the Silhouette Sign
The silhouette sign can sometimes be used to distinguish otherwise similar-appearing chest x-ray findings. For example, a pleural effusion and a dense pulmonary infiltrate may have similar appearances on upright chest x-ray. Because both conditions are water density and both may abut the heart and mediastinum, both may cause loss of the normal silhouette sign. However, a pleural effusion appears uniformly dense, whereas a pulmonary alveolar infiltrate may result in the appearance of a new, abnormal silhouette called an air bronchogram ( Figure 5-27 ). Normally, bronchi within the lung parenchyma are not visible on chest x-ray. This is because the bronchi contain air and immediately abut alveoli, which also contain air. The bronchial wall itself is too thin to attenuate x-ray to a significant degree and is therefore invisible, except when it is seen end-on. In these cases, it may be visible as a small, circular density with a hollow, lucent (black) center. This normal scenario is disrupted when alveolar fluid is present—whether that fluid is pus (pneumonia), serous fluid (pulmonary edema), blood (pulmonary hemorrhage), or lunch (aspirated food material). Under these conditions, fluid-filled alveoli become visible as water density immediately adjacent to air-filled bronchi, and the silhouette of air-filled bronchi may be seen. At extremes of the conditions described, pus, pulmonary edema fluid, blood, or aspirated material may fill bronchi as well, in which case air bronchograms may not be seen. When present, air bronchograms prove alveolar opacification. When air bronchograms are absent, either pleural fluid or alveolar and bronchial fluid may be present.

Silhouette Sign and Lateral Chest X-ray
We discussed the lateral chest x-ray earlier in some detail in the section on x-ray technique and views. However, this topic is important enough to revisit in the context of the silhouette sign.
Retrosternal Space
The retrosternal space is invisible on the frontal chest x-ray, as it lies directly anterior to the soft tissues of the heart and upper mediastinum. On the lateral x-ray, this space is normally lucent (black) because of the presence of aerated lung tissue and epicardial fat; it forms a normal silhouette against the soft tissues of the heart and upper mediastinum. A
Normally, the heart and diaphragm (water density) abut low density lung tissue (air density) and thus are silhouetted against it. Loss of this silhouette implies that lung density has been replaced by water density. Causes include:
Alveolar consolidation (pulmonary edema, infectious infiltrate, pulmonary hemorrhage, aspirated material)
Atelectasis or lobar collapse
Herniated abdominal solid organs
Pleural fluid (effusion, empyema, or hemothorax)
- •
Solid mass pulmonary lesion
Retrocardiac Space
The normal retrocardiac space is lucent (black) on the lateral chest x-ray because of aerated lung in this location; it forms a normal silhouette with the soft tissues of the heart anteriorly, the diaphragm inferiorly, and the bones of the thoracic spine posteriorly. The space is hidden on the frontal projection by the presence of the heart. When tissue increases in density in this area, the appearance is a brighter white—a possible indication of a retrocardiac infectious infiltrate or other cause of increased parenchymal or pleural opacity, such as mass or edema. Inspection of the retrocardiac space on the lateral chest x-ray is therefore an important step in assessing the x-ray. Loss of the normal silhouette sign in this location, or the presence of an abnormal density, is evidence of pneumonia, mass, pulmonary edema, or pleural effusion (see Figures 5-16 and 5-18 ).
Fissures
The major and minor fissures can provide anatomic landmarks, revealing important disease when present ( Figures 5-28 through 5-30 ). The fissures represent pleural boundaries and are actually a “sandwich” composed of the distinct pleura lining two adjacent pulmonary lobes and the potential space between. Thickening of a fissure on chest x-ray can be the result of thickening of the pleura itself, fluid accumulating in the potential space between the two pleural layers, or disease such as malignancy. The minor fissure (also called the transverse fissure) is usually visible as a thin horizontal line on both frontal (PA or AP) and lateral chest x-ray projections. It separates the upper and middle lobes of the right lung. The major fissure is not visible on the frontal chest x-ray projection. On the lateral projection, it is visible as a thin line running from the inferior portion of the anterior thorax to the superior aspect of the posterior thorax. It is actually composed of two fissures, superimposed on the lateral projection. On the left, it divides the upper and lower lobes of the left lung. On the right, it divides the upper and middle lobes from the lower lobe of the right lung.
Five Terrible Ts
- •
Thyroid mass (goiter or malignancy)
- •
Thymoma
- •
Teratoma
- •
“Terrible” lymphoma
- •
Thoracic aortic aneurysm






Changes in Volume and Pressure on Chest X-ray
Changes in volume and pressure can be seen on frontal chest x-ray and can indicate immediately life-threatening pathology ( Figure 5-35 ). Mediastinal shift is seen with both increased and decreased pressure and volume. Increased pressure and volume on the side with pathology push the mediastinum away from the abnormal side. Decreased pressure and volume pull the mediastinum toward the abnormal side. In both cases, tracheal deviation may be seen, in the same direction as the mediastinal shift.

Increased Pressure and Volume
Increased pressure and volume, as seen in a tension pneumothorax ( Figure 5-36 ; see also Figure 5-35 ), result in the diaphragm on the abnormal side being pushed down. A tension pneumothorax on the right side may be notable for the right hemidiaphragm being lower than the left—a reversal of the normal pattern. Normally, the right hemidiaphragm is higher than the left because of the liver (right side) being larger than the spleen (left side) and displacing the diaphragm upward. In tension pneumothorax, a deep sulcus sign can be seen (see Figures 5-35 and 5-36 ), with the costophrenic angle on the affected side being deeper than on the unaffected side. In some extreme cases, the sulcus may be so deep that its inferior extent is not visible on the chest x-ray. Tension pneumothorax is discussed in detail in Chapter 6 . We discuss pneumothoraces in more detail later, but naturally a pneumothorax is distinguished by an absence of lung markings on the affected side. Purists argue that tension pneumothorax is a clinical diagnosis, marked by hypotension and shock, and never to be found on chest x-ray. Nonetheless, chest x-rays sometimes record these findings, and an emergency physician must immediately recognize chest x-ray findings suggestive of tension pneumothorax and respond appropriately.

Decreased Pressure and Volume
Decreased pressure and volume (see Figure 5-35 ) may result in the diaphragm appearing higher than usual—because of the absence of aerated lung pushing it down into its normal position. In addition, the normal position of the minor fissure may be changed on the frontal x-ray. The minor fissure is usually visible as a thin horizontal line roughly at the midpoint of the cephalad–caudad diameter of the thorax. When upper lobe collapse occurs, the minor fissure is elevated, whereas lower lobe collapse may result in the minor fissure assuming a lower-than-normal position. Think of lobar collapse as resulting in an accordion-like compression of the involved lung segment, with the minor fissure following in the direction of collapse and volume loss. In addition, the collapsed lobe may be visible as an increased density. If the collapsed lobe abuts the heart, abnormal loss of the silhouette sign may occur. Common scenarios leading to lobar collapse include basilar atelectasis ( Figure 5-37 ), bronchial mucus plugging ( Figure 5-38 ), iatrogenic right main bronchus intubation ( Figure 5-39 ), obstructing endobronchial lesion (e.g., carcinoma), and extrinsic compression of a bronchus from a mass lesion. Patients who have undergone pneumonectomy also have significant volume loss ( Figures 5-40 and 5-41 ).