Abstract
Immune hydrops is fetal hydrops resulting from the passage of maternal antibodies into the fetal compartment causing fetal anemia and subsequent hydrops. This disease process can affect both the fetus and neonate and is better labeled as hemolytic disease of the fetus and newborn (HDFN). Although the incidence of rhesus (Rh) D alloimmunization has decreased with the advent of rhesus immunoglobulin to the D antigen, HDFN resulting from Rh(D) and non-Rh(D) antibodies still poses a significant risk to the fetus and neonate. This chapter will outline the definition, epidemiology, pathophysiology, laboratory and ultrasound evaluation, and clinical management of women affected by alloimmunization.
Keywords
hydrops, immune, fetal, alloimmunization, rhesus
Introduction
The fetal-maternal interface in pregnancy does not act as a barrier to the passage of fetal cells into the maternal circulation, and the passage of fetal deoxyribonucleic acid into the maternal compartment is one proposed mechanism for preventing immunologic rejection of the fetus with its paternally derived foreign antigen. The transfer of fetal red blood cells into the maternal circulation, however, has the ability to initiate a maternal immunologic response that can be deleterious to the health of the fetus. If red blood cell antigens foreign to the maternal immune system cross into the maternal compartment, a process may be initiated leading to the immune destruction of fetal red blood cells. This can lead to severe fetal anemia, and if untreated, eventually to immune-mediated hydrops fetalis. This process can affect both the fetus and the neonate, and is thus known as hemolytic disease of the fetus and newborn (HDFN).
HDFN is an example of modern medicine both treating and preventing a disease process with significant morbidity and mortality. In the 1950s, 15% of all “isoimmunized” (the currently accepted term is the more appropriate “alloimmunization” ) gestations ended in stillbirth. Today, survival in severely affected neonates is well over 90%. Advancements in ultrasound and intrauterine transfusion technique have certainly played a role in the improved outcome, but the prevention of rhesus (Rh) alloimmunization by the introduction of rhesus immunoglobulin (RhIG) has significantly decreased the disease burden of Rh(D) disease in particular. As stated in the Benjamin Franklin axiom, “an ounce of prevention is worth a pound of cure.”
Disease
Definition
Immune hydrops is defined as the diagnosis of fetal hydrops in the presence of a maternal immunologic response to a paternally derived red blood cell antigen in the fetus. Fetal hydrops is further defined as the accumulation of fluid in at least two of the following fetal serous compartments: abdomen, pleural cavity, pericardium, and skin. Polyhydramnios and placental thickening are also commonly included in the diagnostic criteria.
Prevalence and Epidemiology
The incidence of Rh alloimmunization in the United States is reported to be 6.8 : 1000 live births. The adoption of prenatal and postpartum RhIG has resulted in a marked reduction in the incidence of disease caused by the presence of the paternal Rh(D) antigen, however, 1%–3% of Rh(D)-negative women will still become alloimmunized despite proper prophylaxis. There has been a corresponding increase in the incidence of other Rh antigens and non-Rh antigens that can cause HDFN, with a reported incidence for these antigens of 0.2% of pregnancies. A recent Canadian study identified 559 cases of alloimmunization among 155,153 pregnancies for an incidence of 0.36%. Only 34 (0.06%) of the affected pregnancies had adverse outcomes defined by the need for prenatal therapy (maternal plasmapheresis or intrauterine transfusions), an intrauterine death attributed to alloimmunization, preterm delivery secondary to HDFN, or neonatal treatment (intravenous immunoglobulin [IVIg] administration, transfusion, or exchange transfusion). A prospective analysis of over 300,000 women in The Netherlands showed that the antibodies most commonly associated with HDFN were anti-E, followed in decreasing frequency by anti-D, anti-Kell, and anti-c antibodies. The decreasing prevalence of anti-D antibodies was also shown in the aforementioned Canadian study with anti-E (43%), anti-c (13%), anti-Jk a (9%), and anti-C (5%) antibodies making up a majority of affected women, with anti-D accounting for only 0.4% of women with a single antibody identified. Currently in the United States, HDFN causes approximately 200 perinatal deaths yearly, most from alloimmunization to the Rh(D), Rh(c), and Kell antigens.
Etiology and Pathogenesis
Fetal red blood cells express antigens by 30 days of gestation, half of which are paternally derived. The transfer of fetal antigens across the fetal-maternal interface occurs in almost all gestations with increasing frequency and volume as the pregnancy progresses. Fetomaternal hemorrhage before or during birth is thought to be the primary factor in maternal alloimmunization. A list of potential spontaneous and iatrogenic events associated, with maternal sensitization are listed in Table 123.1 .
| Unprovoked | Provoked |
|---|---|
| Idiopathic-spontaneous | Termination of pregnancy |
| Delivery | Amniocentesis |
| Spontaneous abortion | Chorionic villus sampling |
| Ectopic pregnancy | Cordocentesis |
| Abruption | External cephalic version |
| Antepartum hemorrhage | Fetal intervention: fetoscopy, shunt, drainage |
| Trauma | |
| Manual removal of placenta |
The maternal response to foreign antigen is well documented. Cells of the innate immune system identify and destroy foreign cells and present the antigens to the humoral immune system, where specialized B lymphocytes can recognize the antigen in future encounters and respond rapidly with immunoglobulin (Ig) G antibodies. The initial development of IgG antibodies is a slow process taking 5–15 weeks for a human antiglobulin titer to be detected after a sensitizing event. With few exceptions, the maternal response to the paternal antigen is not sufficient to affect the sensitizing pregnancy. Exposure to a subsequent gestation with the precipitating antigen present will trigger the rapid production of IgG antibodies. These IgG antibodies freely cross the placenta and bind to fetal red blood cells with the offending antigen. These marked cells are then sequestered by the fetal spleen and destroyed by macrophages resulting in fetal anemia if the response is significant enough.
The maternal response to paternal antigens on fetal red blood cells that cross the fetomaternal barrier is variable. Studies in Rh(D)-negative men have demonstrated that some individuals can be sensitized by as little as 0.1 mL of Rh(D)-positive blood, whereas 30% of individuals are still not sensitized after subsequent exposure to 10 mL of Rh(D)-positive blood. Other factors thought to play a role in sensitization are the frequency of the exposure, the ABO compatibility between the fetus and the mother, the antibody class, placental transport of the antibody, the maturity of the fetal red blood cell antigen, and the status of the maternal immune system as immunodeficiency may prevent alloimmunization.
Hydrops occurs when the fetal hemoglobin is greater than 7 g/dL below the mean for the estimated gestational age. This is consistent with a fetal hematocrit and hemoglobin of less than 15% and 5 g/dL, respectively. Erythropoiesis is elevated in the fetus with the exception of alloimmunization caused by anti-Kell antibodies that destroy erythroid precursor cells and result in an earlier onset and more severe anemia than that found with other alloantibodies.
The exact mechanism for the development of hydrops is not understood. Lower serum albumin has been hypothesized to lead to decreased colloid osmotic pressure, the third-spacing of fetal fluid, and finally the development of hydrops. This theory, however, is not supported by animal models or in human fetuses with hypoproteinemia. Other proposed theories for hydrops are: (1) iron overload from hemolyzed red blood cells leading to increased free radical formation and endothelial cell dysfunction, (2) tissue hypoxia from anemia leading to increased capillary permeability, and (3) elevated central venous pressure resulting in lymphatic blockage. The development of hydrops is also gestational-age dependent. Hydrops under 22 weeks’ gestation is rare despite the presence of severe anemia.
Clinical Presentation
The American College of Obstetrics and Gynecology recommends the screening of all women for red blood cell antibodies at their first prenatal visit. A positive maternal antibody screen should identify all women at risk for HDFN, and this should prompt a referral to a maternal-fetal medicine unit with expertise in handling alloimmunization and the appropriate monitoring of the pregnancy. Patients who do not, however, have access to medical care or fetuses with late-presenting developing antibodies may first present with maternal symptoms suggestive of hydrops fetalis. Polyhydramnios can lead to size greater than dates, preterm contractions, and preterm labor. Mothers of hydropic fetuses are at risk of developing Mirror syndrome. This is characterized by a preeclampsia-like picture with generalized edema, hypertension, proteinuria, and often pulmonary edema. It differs from preeclampsia in that the hematocrit is low, as opposed to the hemoconcentration seen in preeclampsia, and it is reversible if the cause of the fetal hydrops, in this case anemia, can be treated and resolves.
Imaging Techniques and Findings
Ultrasound.
The diagnosis of hydrops is dependent on identifying any two of the following:
- •
Abdominal ascites: is initially seen as a rim of echolucent fluid on the periphery of the abdominal cavity, and as it progresses the bowel and liver will become more prominent secondary to the surrounding fluid ( Fig. 123.1 ). Ascites must not be confused with pseudoascites, which is the hypoechoic rim normally seen around the anterior abdominal wall caused by the abdominal muscles. Care must also be taken to differentiate isolated urinary ascites, as seen with a cloacal abnormality or a decompressed lower urinary tract obstruction, from early HDFN.
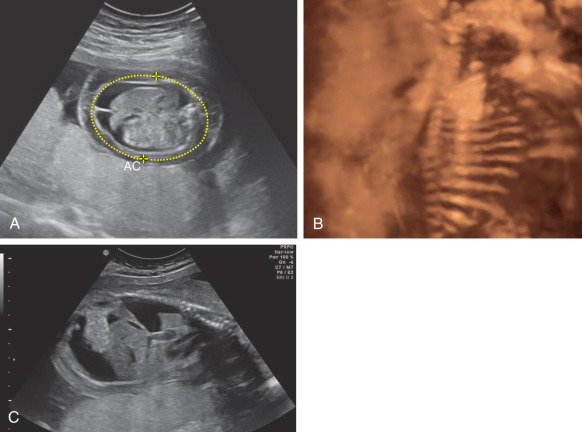
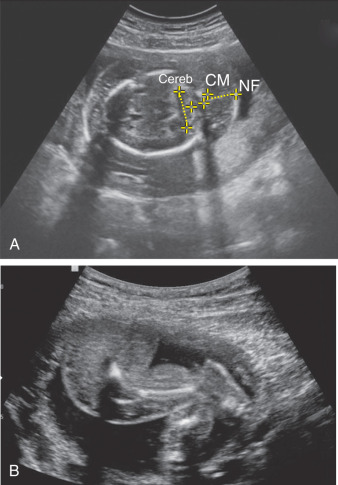
Fig. 123.1
(A) Fetal ascites and body wall edema. (B) Three-dimensional image of fetal abdominal ascites. (C) Sagittal view of pleural effusion and ascites.
AC, Abdominal circumference.
- •
Pleural effusion: similar to ascites, presents as a rim of echolucent fluid, but within the thoracic cavity surrounding the lungs ( Fig. 123.2 ). The effusions may be unilateral or bilateral and if persistent and severe, may result in pulmonary hypoplasia.
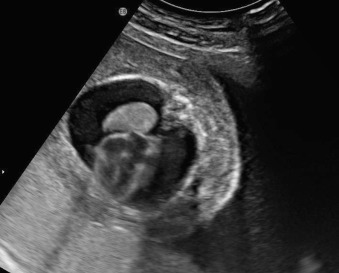
Fig. 123.2
Pleural effusion shown with the lung and fetal heart (there is also body wall edema).
- •
Pericardial effusion: presents as a rim of echolucent fluid surrounding the heart ( Fig. 123.3 ). Up to 2 mm of fluid around the heart is considered normal, and care must be taken to measure the fluid during cardiac diastole to avoid artificially increasing the thickness of the pericardial space. True pericardial effusions generally extend all the way around the heart, extending above the atrioventricular groove, and do not move with adjacent cardiac activity.
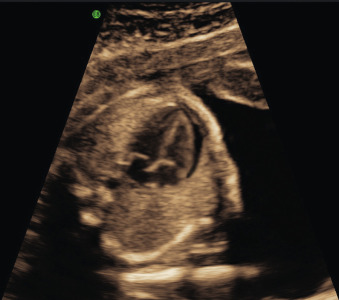
Fig. 123.3
Pericardial effusion at the level of the fetal four-chamber view.
- •
Skin edema: is defined as skin thickness greater than 5 mm and must be differentiated from underlying subcutaneous fat ( Fig. 123.4 ).