Urinary tract infection (UTI) is among the most common of bacterial infections. Imaging studies are only required in cases of complicated UTIs. Ultrasound remains the first-line imaging method; however, CT is the gold standard for evaluation of UTIs. MR imaging’s improved contrast resolution and absence of ionizing radiation may recommend its use for assessment of lower UTIs. Furthermore, MR imaging performs with diagnostic accuracy at least similar to CT in complicated UTIs, except for the identification of calculi and emphysematous pyelonephritis. In this article, the role of MR imaging for the evaluation of infectious and inflammatory disease processes of the urinary tract is reviewed.
Key points
- •
Noncontrast MR imaging is the best imaging modality for the evaluation of complicated urinary tract infection in pregnant patients.
- •
MR imaging is particularly helpful for the evaluation of complicated lower urinary tract infections and inflammatory disorders, including prostatic and urethral infections and inflammation.
- •
A limitation of MR imaging compared with CT in the evaluation of urinary tract infections is low sensitivity for the detection of stones and air, impairing the ability to detect obstructing stones and emphysematous pyelonephritis and pyelitis.
Introduction
Urinary tract infection (UTI) is defined as the bacterial invasion of the urinary tract, which can occur anywhere between the urethra and the kidney and is among the most common of all bacterial infections. Bacteria infect the usually sterile urinary tract either by traveling from the blood stream to the kidneys (hematogenous spread) or, more commonly, by infecting the urethra and traveling upward toward the bladder and kidneys (ascending infection). Less frequently, infection can arise from direct extension, in cases of fistulas with the surrounding organs. Imaging studies are indicated for the evaluation of complicated UTIs, in patients who are septic or not responsive to initial antimicrobial therapy, and in patients with recurrent UTI episodes or atypical clinical presentation or atypical laboratory results, to look for complications (eg, obstruction, abscess, or perirenal fluid collection) and to identify rare forms of pyelonephritis.
In this article, the role of MR imaging and MR imaging semiology is reviewed. For systematization purposes, infectious and inflammatory diseases are divided into upper urinary tract, including kidneys and ureters, and lower urinary tract, including the bladder, urethra, and prostate.
MR imaging evaluation of the urinary tract
Ultrasound is routinely used as a first-line imaging method. It plays an important role in the detection of hydronephrosis, calculi, perinephric collections, congenital anomalies, or urinary tract wall thickening. This method has limited sensitivity, however, in the evaluation of urinary tract inflammation and infection.
CT and MR imaging provide excellent anatomic detail and image quality, which improve the diagnostic work-up of the pathologic conditions of the urinary tract and surrounding tissues.
Several investigators consider CT the gold standard for the imaging evaluation of UTIs because it allows the assessment of the renal parenchyma, urinary tract, and vasculature in a single examination and with extremely short acquisition times. CT has high accuracy in demonstrating changes in renal parenchymal perfusion, calculi, gas-forming infections, hemorrhage, obstruction, inflammatory masses, abscess, or retroperitoneal collections. The absence of ionizing radiation of MR imaging is also relevant considering the commonly young age of patients with UTIs. Also, MR imaging may improve the distinction between healing, fibrotic, and edematous (presumably active) infectious foci and improve imaging in kidney infections. Specific disadvantages of MR imaging encompass the lesser spatial resolution as well as the lower sensitivity at showing calcifications (contrary to CT, which has a great accuracy at depicting calcification) and gas.
One important aspect to consider is related to the use of intravenous (IV) contrast medium, because the high accuracy of CT depends greatly on the use of IV iodinated contrast agents, which allow the assessment of renal perfusion and the depiction and characterization of inflammatory lesions and abscesses. The administration of IV contrast is limited, however, in patients with renal impairment due to concerns of worsening the underlying renal impairment or of postcontrast acute kidney injury. Dynamic contrast enhancement (DCE)–MR imaging has been used as an alternative to contrast-enhanced CT, because it is much less nephrotoxic, especially when used in appropriate clinical doses. Iodinated contrast agents, however, may be used in patients underlying hemodialysis, where gadolinium-based contrast agents (GBCAs) are generally contraindicated due to potential triggering of nephrogenic systemic fibrosis or worsening gadolinium deposition in the body. Furthermore, unenhanced MR imaging, combining morphologic and functional sequences, such as diffusion-weighted imaging (DWI), show high diagnostic value compared with unenhanced CT, particularly in pregnant patients in whom the use of GBCAs is not recommended.
Sequences
Despite modern software development, the basic MR imaging examination is still based on a combination of T1-weighted images (WI) and T2-WI and functional techniques, including DCE and DWI.
High spatial resolution is achieved by a combination of external pelvic phased-array coils, thin-section thickness (≤3 mm), no interslice gaps, and large matrix size. Bowel peristalsis can be minimized with antiperistaltic agents.
To image the kidneys, axial 3-D T1-WI in-phase and out-of-phase gradient-recalled-echo (GRE) and fat-suppressed (FS) T1-WI GRE imaging is performed before and after the IV GBCA administration. Postcontrast sequences remain an integral part of most protocols. DCE images are acquired in a late arterial phase (35–40 seconds after contrast injection) and in nephrogenic phase (100 seconds after contrast injection), when all of the renal parenchyma, including the medulla, enhances. Interstitial phase and excretory phase are also usually acquired. Non-FS and FS single-shot (SS) echo-train spin-echo (ETSE) T2-WI in at least 2 planes are obtained. Damaged tissue tends to develop edema, which makes water-sensitive sequences sensitive for pathology and generally able to distinguish pathologic tissue from normal tissue.
For the lower urinary tract, in addition to FS T1-WI GRE, at least 2 planes of 2-D T2-WI turbo spin-echo are obtained.
MR imaging urography (MRU) can be performed with 2 different imaging strategies.
Unenhanced MR Imaging Urography
Unenhanced MRU, based on heavily (static-fluid) T2-weighted FSE or SS–fast spin-echo (FSE) sequences, is especially useful in the urinary tract, because water-containing structures, such as the collecting system and the bladder, are bright white, providing static hydrographic images of the urinary tract. Despite being useful and reliable to reveal hydronephrosis and perirenal high SI in acute urinary obstruction, its inability to fully visualize nondilated ureters is usually cited as a major disadvantage.
Contrast-Enhanced Excretory MR Imaging Urography
Contrast-enhanced excretory MRU (CE-MRU) is performed with 3-D–GRE T1-weighted sequences after IV GBCA administration. In routine practice, the excretory CE-MRU is the technique most commonly used because the administration of a paramagnetic contrast agent permits the evaluation of renal excretory function and better visualization of the nondilated urinary tract. Low-dose (5–20 mg) IV furosemide administration has been recommended for excretory CE-MRU, because the increased urine flow leads to a rapid and uniform distribution of gadolinium, improving the visualization of the nondilated upper urinary tract.
When IV contrast cannot be administered, DWI offers a viable alternative. DWI is usually performed in 2 blocks covering the abdomen and pelvis separately. At least 2 b values (<100 s/mm 2 and ≥600 s/mm 2 ) should be used to enable the calculation of the apparent diffusion coefficient (ADC) maps. DWI, with a low b value (<100 s/mm 2 ), can be used as a surrogate for FS T2-weighted MR imaging sequences to save examination time.
Upper urinary tract
Acute Pyelonephritis
Acute pyelonephritis (APN) is a nonspecific inflammatory process secondary to ascending (more frequently) or hematogenous bacterial infection. Escherichia coli causes more than 80% of APN. Patients with APN can present with all the symptoms of cystitis, flank pain, and fever, and they may be acutely ill with nausea, vomiting, and unstable vital signs.
Imaging studies are usually reserved for patients who do not improve to seek possible obstruction, urologic abnormalities, and perinephric and intrarenal abscesses.
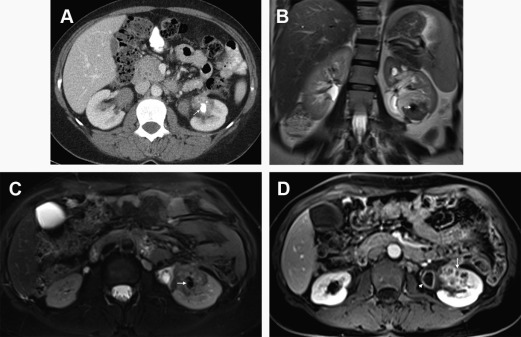
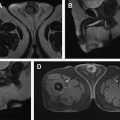
On MR imaging, T1 and T2 baseline acquisitions may reflect the distribution of edema within the renal parenchyma, resulting in typically increased renal volume and nonhomogeneous hypointensity in T1-WI and hyperintensity in T2-WI ( Fig. 1 ); however, these findings have low sensitivity to detect inflammatory foci. The decrease or loss of the corticomedullary differentiation is a nonspecific finding, which may be seen in a small percentage of cases.
DCE study harbors the greatest accuracy of MR imaging because it enables the identification of inflammatory foci that were not detected at the baseline and increases the diagnostic confidence. On postcontrast FS T1-WI, a reduction of parenchymal contrast enhancement is observed in the affected area, allegedly resultant of poor functioning parenchyma due to vasospasm, tubular obstruction, or interstitial edema. The distribution of inflammatory foci is typically patchy with wedge-shaped areas radiating from the papilla to the cortical surface, with the renal parenchyma demonstrating a typical striated appearance due to interspersed areas of parenchyma spared— striated nephrogram sign . The most sensitive phase for identifying these nonenhancing areas is the nephrographic phase. Differentiation of striated nephrogram sign from renal infarction is essential; however, it seldom represents a diagnostic challenge due to different clinical contexts.
The presence of perinephric heterogeneity and T2 hyperintensity immediately adjacent to the abnormal areas in the renal parenchyma, equivalent to CT perinephric fat-stranding sign, is also seen in a moderate amount of cases and is due to inflammatory infiltration that has spread beyond the renal capsule. This is better appreciated on FS T2-WI. The spread of these inflammatory changes also results in thickening of the Gerota fascia as well as of the urothelium of the renal sinuses.
The high blood flow and water transport functions of the kidney make it a particularly interesting organ to study with DWI. Some studies have shown good agreement between DWI and CE–MR imaging for the diagnosis of APN, with good sensitivity for DWI to detect the focal areas of pyelonephritis and to demonstrate the disappearance of inflammatory foci, providing evidence of complete healing. It still lacks specificity and clinical validation, however. Faletti and colleagues reported superiority of DCE–MR imaging over DWI in identifying complications of APN, including focal abscesses.
Isolated pyelitis and pyeloureteritis may rarely occur and are depicted as circumferential thickening of the walls of the collecting system, with T2 hyperintensity due to the presence of mural edema and increased contrast-enhancement and stranding of the periureteral fat due to the presence of inflammation.
Renal Abscess
Untreated or inadequately treated APN may progress to tissue necrosis and liquefaction, resulting in the coalescence of suppurative foci into a larger focal collection, especially in diabetics (75% of the cases), immunocompromised patients, and patients with obstruction.
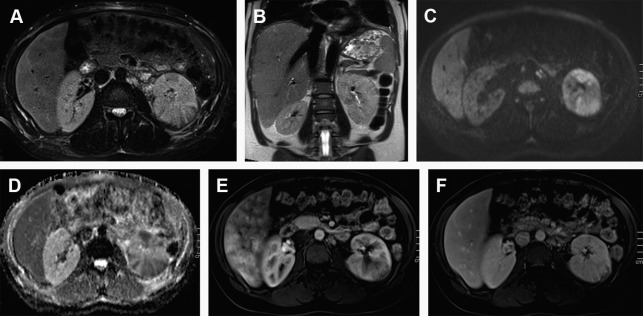
Although initially, abscesses appear as small wedge-shaped or rounded nonenhancing areas with irregular and poorly defined margins, the mature abscesses instead are sharply marginated. Abscesses typically show low, inhomogeneous signal intensity (SI) on T1-WI and high SI on T2-WI depending on the amount of protein, fluid, and cellular debris. When debris are present, a fluid-fluid level may be seen. Postcontrast images show a peripheral halo enhancement, which usually progresses through different phases ( Fig. 2 ). The delayed phase occasionally reveals itself critical to identify the abscess foci and differentiate them from nonabscessed foci and infarction scars. The external part of the capsule of the abscess can appear irregular due to associated inflammatory infiltration. Abscesses usually show high signal on DWI and low ADC values. One possible explanation for the decreased ADC values within the abscess cavity is the conglomeration of white blood cells with intact cell membranes, which leads to high viscosity and cellularity.
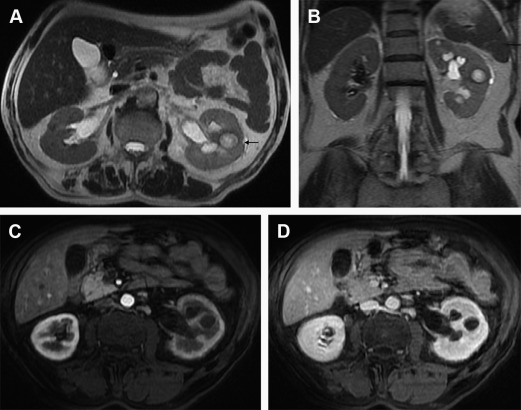
The presence of gas may be seen as areas of susceptibility artifact within an inflammatory mass, strongly implying abscess. It is an uncommon finding, however, and differentiation from calcific foci may be difficult or even impossible.
Abscesses may spread through the renal parenchyma and rupture its capsule into the perirenal space and remain contained by Gerota fascia or may diffuse into other retroperitoneal space and the abdominal wall, well evaluated by multiplanar MR imaging.
Emphysematous Pyelonephritis
Emphysematous pyelonephritis (EPN) is a potentially serious fulminant bacterial infection of the kidneys characterized by gas in the collecting system, renal parenchyma, and perinephric tissues. Most commonly, it occurs in diabetic patients with urinary tract obstruction and often carries adverse outcomes, with a mortality rate of 20%. Glucose-fermenting E coli is responsible for 70% of EPN cases, followed by Klebsiella pneumonia , and Proteus mirabilis . Nevertheless, nondiabetic patients also get EPN.
CT is very sensitive and allows an accurate staging of the disease and is considered the gold standard for diagnosis.
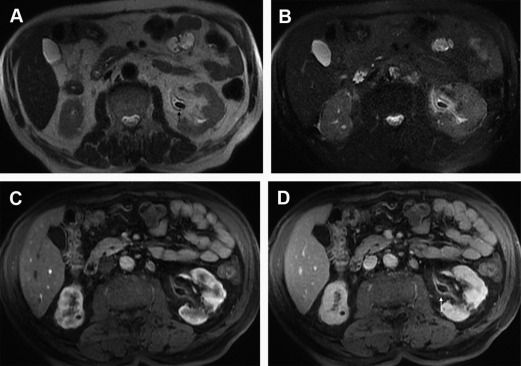
A diagnosis of EPN is challenging on MR imaging and is characterized by the presence of gas in the renal parenchyma, the urinary tract, or perirenal tissue. The free gas manifests as signal voids on both T1- and T2-WI, with blooming artifact recognized better on GRE sequences. Contrast injection may be useful to assess the extent of renal parenchyma destruction. The presence of gas limited to the collecting system may be related to emphysematous pyelitis, which has a less severe prognosis. A potential pitfall may be the presence of gas in the collecting system due to recent catheterization or a urointestinal fistula.
Hydronephrosis and Pyonephrosis
Hydronephrosis is important in the context of UTI because it makes the urinary tract vulnerable to infection and permanent dysfunction. Obstruction predisposes to and usually precedes the infection but the reverse can also occur. The differentiation of pyonephrosis and hydronephrosis is important because pyonephrosis needs immediate intervention due to the high risk of sepsis and loss of kidney function.
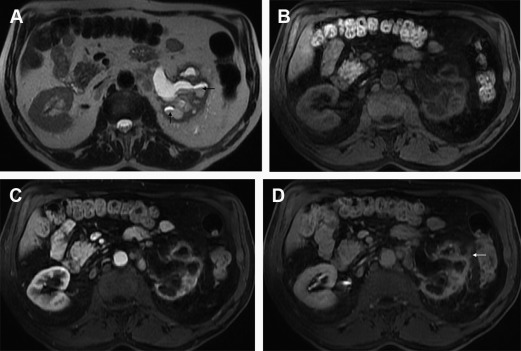
MR imaging is more sensitive than CT for the diagnosis of sources of urinary obstruction other than urolithiasis; nevertheless, MR imaging still has a high sensitivity (>90%) for the detection of obstructive ureteral calculi. The relative absence of protons within urinary calculi makes them incapable of generating signal on MR imaging; thus, calculi is only detected by the surrounding signal-generating urine on T2-WI ( Fig. 3 ). Having said that, it is understandable that the sensitivity for renal stones increases in relation to the size of the calculus and the use of thin sections. Balanced gradient-echo sequences are also particularly effective for the depiction of larger stones as hypointense filling defects surrounded by hyperintense urine, with the advantage of being insensitive to flow-related artifacts (present on SS-FSE T2-WI).
Other signs of urinary tract obstruction include swelling of the ipsilateral kidney, periureteric and perinephric edema, and urothelial wall thickening. Hyperenhancement after contrast injection renders the presence of inflammatory hyperemia. After contrast administration, there is a delayed nephrogram because of decreased renal perfusion. If delayed images are obtained, a persistent nephrogram can be seen on the obstructed side.
Pyonephrosis is defined as the presence of pus in a dilated renal collecting system. With pus under pressure, patients may deteriorate rapidly and become septic, prompting differential diagnosis between hydronephrosis and pyonephrosis, which has a vital clinical significance. Pyonephrosis may be caused by a broad spectrum of pathologic conditions involving either an ascending infection of the urinary tract or the hematogenous spread of a bacterial pathogen.
MR imaging findings in pyonephrosis include thickening of the renal pelvic wall (>2 mm) and dilatation and obstruction of the collecting system, which appears hypointense on T1-WI and hyperintense on T2-WI unless the dependent portion of the collecting system contains high-protein content material or debris (and fluid-debris level) ( Fig. 4 ). A striated nephrogram in the renal parenchyma similar to that seen in pyelonephritis can occur in both pyonephrosis and obstructive uropathy, although, in pyonephrosis, changes appear more severe. Some studies refer the potential of DWI in demonstrating changes in perfusion and diffusion during acute renal obstruction with significant lower ADC values of the pyonephrotic renal pelvis compared with the hydronephrotic renal pelvis ( Fig. 5 ).
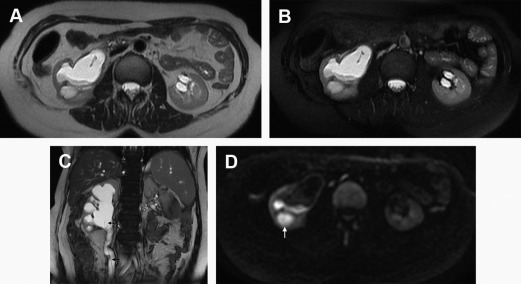
Pregnant women are a special subset of patients in whom MR imaging harbors substantial advantages. The incidence of ureteric stones is up to 1 in 1500 pregnancies, and 80% to 90% of these cases occur during the second and third trimesters. With a positive predictive value of 80%, MR imaging allows for the differentiation of the physiologic dilatation (seen in up to 90% of pregnant women in the third trimester of pregnancy) from the pathologic dilatation caused by an obstructive stone. DWI might be important in the diagnosis of eventual pyonephrosis, because there is a tendency to not administer GBCAs even in the third trimester.
Xanthogranulomatous Pyelonephritis
Xanthogranulomatous pyelonephritis (XGP) is a rare, chronic granulomatous inflammatory disorder of the kidney characterized by a destructive mass that invades the renal parenchyma. XGP is most commonly associated with P mirabilis or E coli infection, usually due to long-standing urinary tract obstruction, classically due to a staghorn calculus, seen in 80% of cases. The kidney is usually nonfunctional. Most cases of XGP involve a diffuse process; however, up to 20% are focal. Pathologically, there is parenchymal destruction and replacement with lipid-laden macrophages.
On MR imaging, it manifests most commonly as a diffuse form with nephromegaly, hydronephrosis, peripelvic fibrosis, and destruction of renal architecture often extending to the perirenal and pararenal space and adjacent structures. The renal cortical thickness is reduced. The cortex may contain multiple abscesses surrounded by the xantoid tissue. Reports on the signal characteristics of the XGP differ to some extent. Some investigators described a hyperintense appearance of the solid component of XGP on T1-WI compared with the renal parenchyma, whereas other investigators reported a more isointense appearance of the solid component on T1-WI. It is possible that the SI of the solid component of XGP on T1-WI depends on the amount of xanthoma cells involved in the granulomatous process. On T2-WI, the solid component of XGP tends to show isointensity with the normal kidney. The content of the locular spaces or cavities is variable and may reflect the heterogeneous composition of the content. Extensive reactive sinus replacement lipomatosis has been reported in XGP and is well-recognized and characterized with MR imaging. Perirenal inflammatory infiltration and enhancement of the perirenal fascia are usually present. Fistulization with the gastrointestinal tract or skin may be occasionally seen. Focal XGP may demonstrate findings similar to those seen in the diffuse form (with sparing of part of the kidney), but others may present as focal pseudotumor and mimic the features of bacterial abscess or renal cell carcinoma ( Fig. 6 ). Obstruction by lithiasis is usually present in both cases.