INFRAHYOID NECK AND CERVICO-THORACIC JUNCTION (THORACIC INLET)
CHAPTER 149  INFRAHYOID NECK, HEAD AND NECK LYMPHATIC SYSTEM, AND THORACIC INLET: INTRODUCTION AND GENERAL PRINCIPLES
INFRAHYOID NECK, HEAD AND NECK LYMPHATIC SYSTEM, AND THORACIC INLET: INTRODUCTION AND GENERAL PRINCIPLES
IMAGING APPROACH
Techniques and Relevant Aspects
Computed Tomography
The techniques for computed tomography (CT) studies of the head and neck for various clinical situations are outlined in Appendix A. The protocols vary somewhat mainly with regard to the beginning and ending point of the study depending on the indications. For example, a survey of the nodal groups may need to include the retropharyngeal or parotid groups in some situations but not in others. The knowledge of the particular drainage pattern for the primary site in question is essential for planning an efficient study that limits unnecessary dosage to the neck viscera; such awareness combined with the basic technical options produces the most accurate and efficient examination.
There are several technical details that can help produce neck studies of astonishingly good detail. First, contrast must be used effectively. This is now simple to accomplish on 16-slice or more multidetector computed tomography (MDCT) units while utilizing no more than 125 to 150 cc of contrast. Precontrast scans are almost never used.
Good positioning is absolutely essential. It is necessary to at least mildly extend the neck so that the body of the mandible should be perpendicular to the tabletop and preferably to get the shoulders relaxed and displaced as far inferiorly as possible. This simple attention to positioning can vastly improve image quality. This becomes most important in patients with short, thick necks and where pivotal decisions rely on the data collected in the low neck or thoracic inlet region and in young children and infants. Also, the gantry may be angled to help cut down on the troublesome “shoulder” artifacts. The angle may be parallel to the mandibular body, the vocal cords, or the plane of the thoracic inlet depending on the specific clinical context and related protocol. The exact angle is chosen according to the primary pathology under study. The default angle should be parallel to the true vocal cords or C4-5 disc space in a suitably extended neck for “neck only” studies, as this will also approximate the plane of the thoracic inlet. Sections should be parallel to the body of the mandible for those indications where the oral cavity and oropharyngeal structures are of significant interest. Such gantry angles allow more sections through the low neck before the full volume of the shoulders that cuts down on photon fluence is encountered. Ultimately, increased exposure may be necessary to maintain adequate image quality as the study progresses from the low neck to the cervical thoracic junction.
Scans of the neck should be made with 1- to 3-mm thick sections, depending on the indication. The field of view (FOV) should be tailored to the anatomy of interest and rarely exceed 16 to 18 cm.
Patients should always be instructed not to move or swallow during data acquisition. Respiration should also be suspended, if possible; this will markedly reduce motion artifacts. No special maneuvers, such as modified Valsalva, are routinely used in studying the neck.
Multiplanar and three-dimensional CT reconstructions are used when appropriate but do not need to be archived as a routine unless desired. Magnetic resonance (MR) is particularly useful for multiplanar depiction of pathologies affecting the low neck and thoracic inlet, especially if the neural elements are of most interest.
Magnetic Resonance Imaging
Basic MR protocols for the neck are presented in Appendix B. The most important factor to ensure good studies of the neck is the use of an appropriate coil that is well fitted to the neck.
Fat suppression techniques used both with and without an intravenous paramagnetic contrast agent should have assumed more importance by now. The frequency selective fat suppression techniques used in the extracranial head and neck still suffer unpredictably from susceptibility problems related to variable coil loading leading to inhomogeneous and unreliable suppression of the fat within the thoracic inlet (Fig. 1.3). Novel pulse sequences using “speeded-up” modifications of older subtraction techniques may improve on this troublesome problem (Chapter 1).
Axial sections of 3 to 4 mm are larger than optimal for study of the compact anatomy of the neck but are used most frequently because of coverage and signal-to-noise–related issues. FOV should be matched to the areas of interest. Coronal sections are particularly helpful for evaluating the root of the neck–thoracic inlet, supraclavicular area, and brachial plexus and for showing the relationship of laterally situated masses to the neural axis. Sagittal images are occasionally useful for the same purpose as well as for evaluating midline masses such as a thyroglossal duct cyst and showing the extent of a prevertebral or postvertebral process relative to the neural axis.
Ultrasound
Ultrasound examination of the neck is done with the patient supine with a pad placed under the neck to allow the maximum degree of hyperextension comfortable for the patient. This draws the structures of the root of the neck up from thoracic inlet and can make the difference between a diagnostic and an uninformative examination. This is especially useful positioning for the search for parathyroid adenomas. Water-soluble gel is used as a coupling agent. The examination of the neck should be done with high-resolution real-time units with color flow Doppler capability. Transducers of 7.5 to 10.0 MHz are standard. Ultrasound is so operator dependent that the examination of the neck should be supervised by a physician who thoroughly understands the clinical history, aim of the study, and technique. Abnormal masses or other abnormalities on ultrasound must always be confirmed in two planes; if this is not possible, another imaging study should be done before a critical management decision is based on the imaging finding.
Doppler techniques are very helpful for identifying normal and abnormal vascular structures, vascular masses, and the relationship of neck masses to the normal vessels. Swallowing may be used to help differentiate the esophagus from an abnormal retrotracheal or tracheoesophageal groove mass. Swallowing a small amount of liquid may also be used to move the esophagus out of the way when searching for parathyroid adenomas, especially at the left lower thyroid pole and infrathyroid region.
Nuclear Medicine
Fluorine-18 2-fluoro-2-deoxy-D-glucose positron emission tomography (FDG-PET) is usually done in conjunction with CT. Thyroid and parathyroid scintigraphy is used with great frequency, and studies with agents sensitive to neuroendocrine tumors are used infrequently. Standard sentinel node techniques are used in skin cancer and in some practices in mucosal-origin cancers. The technical points of these studies are reviewed in Chapter 5.
Pros and Cons
CT, ultrasound, and MRI are powerful and precise imaging tools that often contribute unique information vital to proper patient management when used in the proper clinical context. Over the last two or more decades, there has been an extraordinary increase in the use of CT, ultrasound, and MRI for the diagnosis and management of cervical metastatic disease, neck masses of uncertain etiology, and a variety of other lesions that involve the viscera and supporting structures of the neck. FDG-PET has brought about a more recent similar acceleration in the use of radionuclide studies.
The indications in Table 149.1 represent broad guidelines for the relative use of ultrasound, CT, and MRI in the neck. These relative roles vary in different practices. The neck is very often studied for regional nodal staging in a patient with head and neck cancer. FDG-PET is currently an adjunct that should be used with reasonable discernment for this purpose as well. For other indications, imaging is pivotal in medical decision making. The other major indication for neck imaging is the “neck mass of uncertain etiology.”
The very general indications in Table 149.1 do not include more traditional neuroradiology indications. However, disease in the neck more within the scope of general neuroradiology can result in symptoms that will present primarily to an otolaryngologist, such as carotid atherosclerosis, dissection of the carotid presenting as pulse synchronous tinnitus, or neck and throat pain that may be due to discitis.
The indications for imaging of the neck may be approached primarily with ultrasound, CT, or MRI. Policy should be based on problem-solving plans that determine the imaging pathway most likely to produce all of the information necessary for the least amount of cost and risk. Such plans should be reviewed and updated as technologic advances dictate. Physician acceptance and local availability, state of technology, and expertise often alter specific practice patterns. Imaging studies should integrate with the clinical approach to a particular diagnostic problem and facilitate the medical decision making as much as possible as discussed in Chapter 7.
The relative value of ultrasound, CT, and MRI can be considered in the context of four general problems consolidated from those listed in Table 149.1: (a) neck masses of known or uncertain etiology; (b) staging of cervical metastatic disease; (c) lesions of the thyroid and parathyroid glands; and (d) evaluation of the root of the neck, thoracic inlet, and brachial plexus.
TABLE 149.1 COMPARISON BETWEEN MAGNETIC RESONANCE IMAGING AND OTHER NONINVASIVE IMAGING TOOLS FOR HEAD AND NECK DISORDERS

Ultrasound, CT, or MRI might be the most appropriate primary study for a neck mass. MRI is most appropriate for disorders of the spine and neural axis. This includes patients with brachial plexopathy. Multiplanar MDCT may be more efficient for study of neurologic deficits related to the vagus, phrenic, and recurrent laryngeal nerves or cervical sympathetic plexus because of the large area of coverage, vascular detail, and bone detail required. Certainly, MRI can be held in close reserve for these indications or used primarily if preferred.
Many patients who present for imaging of the neck have a palpable and/or visible mass. Imaging studies in this clinical setting must be integrated with proper clinical triage as discussed in Chapter 7. This produces the most efficacious use of imaging evaluation. Such triage may first include study of pathologic results from needle sampling of the mass. Imaging guidance may sometimes be necessary for such sampling, especially if the mass may be vascular or a nearby vascular structure is at risk during biopsy as discussed in Chapter 6. If a biopsy is performed prior to imaging, the person performing the biopsy must be sure that the mass is not a vascular lesion and that the chosen method will not injure a major vessel. If a lesion is either pulsatile or potentially related to the carotid artery or jugular vein, then imaging, perhaps as simple as a localized ultrasound examination, should precede biopsy and perhaps the same examination can be used to direct such sampling. Ultrasound is excellent for prebiopsy screening and biopsy guidance in the hands of an experienced operator (Fig. 6.6). CT and MRI are less operator dependent and overall usually more informative although at increased cost. MR-guided procedures require costly specialized equipment and are generally less efficient than ultrasound and CT when done on most MRI units.
The clinical situation and biopsy results often dictate whether further imaging is necessary and will also direct a preference for CT or MRI. If the needle biopsy reveals presumably metastatic squamous cell carcinoma (SCCA) to a lymph node or is indeterminate, CT is likely a better primary choice. CT is more simple and likely preferable to MR for staging cervical adenopathy. In some practices, FDG-PET would be the next choice if SCCA is the pathologic result. Both contrast-enhanced computed tomography (CECT) and FDG-PET have value in the detection of clinically occult head and neck primary tumors responsible for cervical metastatic disease. MRI has the potential to improve on CT in this regard, but this has never been documented. All three studies may show entirely submucosal cancers in the nasopharynx, tonsil, and tongue base–glossotonsillar sulcus region. In these locations, CT relies on obliteration of deep and at least partially fatty tissue planes to suspect an infiltrating neoplasm. T2-weighted MRI may reveal more subtle findings such as infiltration of adjacent musculature as suspicious signs of a locally aggressive malignant tumor (Figs. 21.1 and 21.2). There are no studies to date confirming this added potential of MR.
Nondiagnostic biopsies are problematic. Because indeterminate biopsies of the neck often eventually turn out to be metastases from a head and neck primary, CT is suggested as the initial study based mainly on its better rendering of the extent of cervical metastatic disease. MR and/or FDG-PET could be the next step, if preferred. If the biopsy and/or clinical situation strongly suggest a diagnosis other than metastatic SCCA, then the choice of MRI or CT is determined mainly by clinical factors. For instance, in most cases, a known hemangioma is probably evaluated more simply and somewhat better studied by MR than CT.
The other two “general indications” for imaging the neck are the evaluation of cervical metastases discussed in Chapter 157 and the evaluation of thyroid and parathyroid disease discussed in Chapter 169. Specifics with regard to these anatomic sites are discussed in the chapters just indicated. In general, CT is recommended as the primary study together with FDG-PET, in selected cases, for evaluation of cervical metastatic disease. Thyroid mass triage begins with ultrasound. CT and MR are reserved for more advanced problem solving. Radionuclide studies are typically first line in parathyroid dysfunction.
Controversies
Cervical Metastatic Disease
An ongoing controversy in the infrahyoid neck centers on the preferred examination for diagnosing and staging cervical metastatic disease. The choices in various practices include ultrasound combined with fine needle aspiration, CT, MRI (perhaps using a ferromagnetic contrast agent), and FDG-PET usually in combination with a nondiagnostic CT. This controversy is discussed in detail with the specific topic of cervical metastatic adenopathy in Chapter 157.
Unknown Primary
The approach for finding a mucosally inapparent primary as a source of metastatic cancer to the neck stirs some debate. The debate centers on the adequacy of FDG-PET alone or together with definitive anatomic imaging by CT or MRI. All three tests are not justified. This author’s experience indicates that those that are going to be found without a substantial false-positive rate will be found with CT or MRI (Figs. 21.1 and 21.2). FDG-PET may find an occasional lesion not noted by an experienced observer on CT or MRI (Fig. 5.2); however, the false-positive rate due to normal activity in the lymphoid tissue in the Waldeyer ring on FDG-PET studies is so high as to not make this study routinely cost justified if an experienced observer uses an anatomic study with CT or MRI to its full advantage1 (Fig. 5.3). Perhaps FDG-PET could be used more rationally to find a primary after a definitive anatomic imaging study is found to not show the responsible primary. If observers are less experienced or less confident with anatomic imaging, then FDG-PET might be a better imaging starting point.
Thyroid Nodules
Another major controversy is how to approach the follow-up of thyroid nodules and other thyroid pathology detected as incidental findings. This is discussed in detail in conjunction with cancer and thyroid nodule evaluation in Chapter 172.
Pediatric Neck Imaging
There is some minor debate about the relative use of ultrasound, CT, and MRI in the pediatric age range. Ultrasound is often not definitive and adds cost. CT is often the most definitive at the cost of irradiation, but MDCT does not often require sedation. MRI may at times be preferable both for lack of ionizing radiation and the diagnostic problem at hand, but it often requires conscious or deep sedation, not infrequently general anesthesia, in the younger pediatric age ranges.
NORMAL ANATOMY
General Anatomic Description
The range of normal anatomy in the region, pathologic alteration of that anatomy, and basic diagnostic and therapeutic issues in treating neck disease is requisite knowledge for a diagnostic imaging specialist who wishes to make reasonable use of the imaging data. A systematic approach of some sort is the best way to organize this effort. It begins with anatomy.
The neck extends from its root at the thoracic inlet inferiorly to the mylohyoid muscle anteriorly and superiorly and the craniovertebral junction posteriorly and superiorly. The thoracic inlet is defined here as an oblique plane running from the sternal notch to the first thoracic vertebral body.
For descriptive and diagnostic ease in the interpretation of transverse images, it is useful to divide the neck into four compartments. These compartments may also be thought of as functional columns that in turn contain spaces. These spaces have been subdivided and given various names over the years. The important concept here is that one must have a working knowledge of the basic organization of the neck to be an effective imaging consultant with regard to how to study and interpret images of the neck in the many possible clinical contexts that arise. The basic scheme chosen can be reconciled with classical surgical–anatomic concepts of the neck, such as the various named triangles (Fig. 149.1). This organizational effort should not add ambiguity and new names to already existing and clinically long-accepted anatomic concepts, some of which have been used for a century or more.



FIGURE 149.1. A series of dissections and anatomic sections showing the relationship of the traditional surgical triangles of the neck to the deep structures of the neck as well as suggesting a general anatomic organization of the neck for diagnostic imaging purposes and effective communication with referring health care providers. A–C: The triangles of the neck as demonstrated on a cadaver dissection. The anterior triangle is classically composed of the submandibular triangle (A), the carotid triangle (B), and the muscular triangle (B). The anterior triangle of the neck in its most common usage actually excludes the submandibular triangle; that triangle is in the suprahyoid neck and is currently more commonly included in the submandibular space. In (B), the anterior triangle lower component is divided in its classic anatomic subdivisions. Such division is not necessary in working with images. This portion of the anterior triangle projects anteriorly and laterally in the neck. It is bounded by the sternocleidomastoid muscle (SCM) and the submandibular space as well as the infrahyoid strap muscles (IHS). Abnormal masses in the neck can be simply described to a surgeon as projecting toward the anterior triangle. This will facilitate communication. In (C), the classic anatomic posterior triangle is demonstrated. Its superior portion is sometimes called the occipital triangle and its inferior portion called the subclavian triangle. These are needless complications. The term posterior triangle can be used to describe the masses presenting posteriorly and laterally in the neck as projecting into this triangle. Almost all lateral compartment neck masses will project in either one or the other direction, and the use of this simple terminology will facilitate communication to the surgeons and speaks to them clearly in terms of their surgical approach and possible morbidity of surgery with respect to structures at risk during those procedures. D: An anatomic section with the investing layer of cervical fascia outlined in red. The anterior triangle projects superiorly into the area inferior to the mandible and beneath the layer of superficial fascia (arrows). At this level in the neck, the posterior triangle is relatively small, as shown by the arrowheads. E: A section through the mid portion of the neck, as in (D), shows the investing layer of surgical fascia outlined in red. The arrows demonstrate how anterior and lateral and posterior and lateral neck masses will project into the anterior and posterior triangles of the neck, respectively. F: Just above the thoracic inlet, the visceral fascia becomes very well defined as it surrounds the visceral compartment of the neck (white arrowheads). It is thick anteriorly and more flimsy (white arrows) posteriorly. In this anatomic section, the investing layer of cervical fascia (black arrowheads) can be seen surrounding the sternocleidomastoid muscle. The superficial fascia is seen as a separate layer of tissue deep to the subcutaneous fat (black arrows). G: Anatomic section demonstrating the confines of the visceral compartment of the neck outlined in yellow. H: A section lower in the neck showing the visceral compartment again outlined in yellow. At this level in the neck, there are significant transitions of the larynx to the trachea and the postcricoid hypopharynx (PC) to the esophageal verge (EV). I: The lateral compartments of the neck are outlined in green and include the sternocleidomastoid muscle, carotid sheath, and fatty envelopes related to those structures. Note that the lateral compartment frames the visceral compartment of the neck and related retrovisceral (retropharyngeal) space. J: Anatomic section showing the relationship of the carotid sheath and its internal structure relative to other structures in the neck. The carotid artery (C) and jugular vein (J) are loosely adherent to one another and are connected by relatively flimsy fascial layers. The vagus nerve (CN 10) is located between the vessels. The omohyoid muscle crosses the jugular vein at this position in the neck (OM) and can be seen distinctly from the sternocleidomastoid muscle (SCM). K: The posterior compartment of the neck is essentially musculoskeletal and neural. It is outlined in blue in this anatomic section. The arrows suggest that the compartment can be separated into prevertebral and paravertebral space subdivisions.
In general, basic organizational approaches to neck anatomy are based on the arrangement of its fascial layers. However, if the organizational focus is on the often variable, highly detailed discussions of the neck fasciae, needless confusion rather than a practical working knowledge of neck anatomy might be the result. It is enough for the diagnostic imager to take the general approach, such as the one presented here, that has been distilled from the classic literature on this topic.2–8 One can then relate this to the anatomy as it is understood by the surgeons, radiation oncologists, and other clinicians involved in treating neck disorders and who for the most part originally identified and named the fascial layers as well as their related spaces, triangles, and compartments (Fig. 142.3, Table 142.1, and Chapter 142). As one studies the natural history of diseases that affect the neck, it becomes readily apparent that a simplified approach is sufficient not only for understanding the spread patterns and nature of the disease in question but also for making important contributions to patient care. The key is to keep things uncomplicated.
The well-defined neck fasciae are most clearly identified as those membranes that can be incised, sutured, and/or affect the spread of pathology; those membranes include the superficial fascia, investing layer of deep cervical fascia, prevertebral fascia, pretracheal fascia, and carotid sheath (Fig. 149.2). More subdivisions such as the alar fascia and names given to relatively flimsy membranes such as the buccopharyngeal fascia are not very useful and are potentially confusing. Moreover, these are not terms generally considered useful by surgeons dealing with neck pathology, as they could create needless fog both in conversation and reports.






FIGURE 149.2. A series of diagrams demonstrating the fascial layers of the neck and the relationship of important structures in the neck to those fascial layers. A: Note that the alar fascia is shown in this diagram as it separates the retropharyngeal space into two potential compartments. This is a needless complication of this anatomy, and it is shown only for classic anatomic purposes rather than practical significance. Note how the fascial layers of the neck contribute to the carotid sheath and create continuity between the lateral compartments of the neck and the retropharyngeal space. B: Anatomic diagram through the mid neck emphasizing the relationship of the various layers of fascia to the compartments of the neck as demonstrated in Figure 149.1. C: Anatomic diagram relating the fascia to important nerves just above the level of the thoracic inlet. D: Anatomic diagram demonstrating the potential complex splitting of fascial layers. These are shown to emphasize how pathologic processes that involve the neck can spread between these unnamed subdivisions. E–G: Three sections from a contrast-enhanced computed tomography study (CT) showing that in some patients there is a prominent vascular plexus in the retropharyngeal space. This is usually at or above the level of the hyoid bone but may be present anywhere in the neck. H: CT section at the junction of the suprahyoid and infrahyoid neck demonstrating the relationship between the retropharyngeal space (RPS) and the visceral compartment of the neck. Note how the presence of air supports the concept of the neck being simply divided into visceral, lateral, and posterior compartments. The air in the retropharyngeal space communicates freely with that in the lateral compartments with air projecting into both the anterior and posterior triangles (arrows) from the lateral compartment. There is no need to further subdivide the lateral compartment since it is all truly in continuity. Note that the carotid sheath (CS) is not a separate space and its fascial attachments are flimsy. Note how the spread of air is contained by the investing layer of superficial fascia both anteriorly and posteriorly as it bridges the gap between the visceral, lateral, and posterior compartments. Also note how well defined the superficial fascia (arrowheads) is as a useful surgical landmark.
The superficial layer of cervical fascia underlies the dermis and encompasses the platysma; it contains nerves, vessels, and lymphatics (Fig. 149.1A,F). It is quite useful as a landmark for describing the extent of processes spreading in the neck to surgeon. In particular, it marks the depth of pathology or relationship of pathology to the subcutaneous fat and deeper fascial layers and related spaces and compartments.
The investing layer of the deep cervical fascia and prevertebral fascia separate the neck into its basic compartments or functional columns and related spaces that allow the neck to serve its various functions (Figs. 149.2 and 149.3). The investing layer of deep cervical fascia surrounds the neck like a collar. It splits to envelop the sternocleidomastoid and trapezius muscles from their origins on the skull to their insertions on the ribs, clavicle, and scapula. It also divides to cover the parotid gland on both its deep and superficial surface. Anteriorly, it is attached to the mandible and hyoid bone. Inferiorly, it has rather complex attachments, mainly related to the infrahyoid musculature it covers. The prevertebral fascia surrounds the deep neck and its paraspinous musculature (Fig. 149.2K). The carotid sheath, really not a fascia, is a condensation of loose connective tissue derived from multiple other fascial layers (Figs. 149.1J and 149.2B–D,H) that surrounds this major neurovascular bundle from the skull base to the aortic arch; it does not form a “space.” The functional compartments/columns and spaces of the neck are thus set off from one other.
The visceral compartment lies anterior to the cervical spine and is supported by an interwoven fascial sleeve that allows for the gross movements of the upper aerodigestive tract that occur during swallowing and speech. The visceral compartment contains the larynx, hypopharynx, trachea, esophagus, and thyroid and parathyroid glands (Fig. 149.1F–H).
The paired lateral compartments that have been referred to as the vascular and paravisceral spaces, compartments, or functional columns must remain free from constriction and maintain blood flow while the head twists and turns. To this end, the carotid sheath—the major neurovascular bundle of the neck including the carotid, jugular, and vagus nerves—lies within a fatty envelope suspended from condensations of loose connective tissue derived mainly from other fascial layers (Figs. 149.1J and 149.2B–D,H).
The prevertebral fascia surrounds the posterior compartment or functional column; this is the toughest fascia of all, especially anteriorly, because of the compartment’s supportive function (Figs. 149.1K and 142.3A). This compartment can be separated into prevertebral and paravertebral or paraspinous spaces. The various fasciae that separate these compartments are not normally visible on CT or MRI. The following more detailed discussion of the fasciae and the compartments they envelop can help one understand the anatomy, physiology, and patterns of disease seen in the neck.
Detailed Description of Specific Areas
Visceral Compartment and Retropharyngeal (Retrovisceral) Space
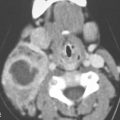
In the upper and mid neck, the larynx and hypopharynx are the sole occupants of the visceral compartment. That detailed anatomy is discussed in conjunction with those organs. In the lower neck, the anatomy of the upper aerodigestive tract is fairly simple. The cricoid cartilage is its most prominent landmark and is easy to recognize as it surrounds the airway posteriorly since it is the only complete ring in the airway (Fig. 149.1H). The postcricoid part of the hypopharynx forms a flattened, soft tissue ellipsoid structure that lies between the cricoid lamina and cervical spine. More inferiorly, the cervical esophagus creates an oval structure posterior to the trachea in the midline (Figs. 149.1F and 149.2D). Faint esophageal mucosal enhancement may be visible on CECT and contrast-enhanced magnetic resonance (CEMR). The trachea normally remains in the midline from the low neck to the thoracic inlet, while the esophagus will often deviate to the left just before it enters the superior mediastinum. The thyroid and parathyroid gland are within this compartment in the low neck at its junction with the thoracic inlet. Normal parathyroid glands are not visible on imaging studies. The more detailed anatomy and CT and MR appearance of these structures are described in Chapter 169.

FIGURE 149.3. A–F: A series of anatomic line diagrams demonstrating the anatomy of the neck and related lymph nodes and other structures to correlate with the computed tomography images in Figure 149.4.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree