LARYNX: INTRODUCTION, NORMAL ANATOMY, AND FUNCTION
IMAGING APPROACH
Techniques and Relevant Aspects
Computed Tomography
A discussion of the use of computed tomography with regard to the larynx is presented in Appendix A.
Positioning and Respiration
The patient should be positioned supine and the neck slightly hyperextended. The body of the mandible long axis should be about perpendicular to the tabletop. Too much hyperextension of the neck may induce coughing or aggravate symptoms of airway obstruction frequently present in these patients and should be avoided. The head should be secured. A digital radiograph is obtained, and a plane of section parallel to the true vocal cords (TVCs) should be selected. If an acquisition plane parallel to the TVC plane is not possible, then it is mandatory that a volume data set suitable for reconstruction parallel to the TVC level be done.
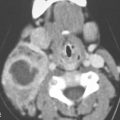
Acquisitions may be performed in quiet respiration or suspended inspiration. Motion artifact on initial acquisitions may require repeated sectioning during suspended inspiration. Suspended inspiration often produces better images; however, during breath-holding, many patients do a partial or modified Valsalva, which may alter the laryngeal anatomy significantly (Fig. 201.1). Such “strained” breath-holding may cause adduction of the TVCs, making visualization of the anterior and posterior commissure difficult with the arytenoids rotating and sliding medially. The hypopharynx may be distended with air down to the apices of the pyriform sinus if there is a coincidental modified Valsalva. In this case, the base of the aryepiglottic folds and the posterior pharyngeal wall will be easier to evaluate. However, a true Valsalva during breath-holding will collapse the pyriform sinuses, possibly making it more difficult to evaluate the hypopharynx and lower aryepiglottic folds.
The added value of acquiring images during a dynamic maneuver in evaluating laryngeal cancer is, on average, very limited and something that is not expected by or asked for by our clinical colleagues. The success rate of these dynamic maneuvers is variable and strongly dependent on the cooperation of the patient. Dynamic maneuvers are mainly helpful in showing superficial tumor spread, whereas the main purpose of imaging is to delineate deep tumor extent. Our clinical colleagues do much better than imaging at assessing the mucosa even in potentially hidden areas such as the laryngeal ventricle. Also, abnormal mobility of the vocal cord is seen more accurately during clinical examination than on such dynamic imaging studies. Few practices use the dynamic techniques. A planned modified Valsalva may be used to evaluate the apex of the pyriform sinus or posterior pharyngeal wall. This is accomplished by patients holding their breath and puffing out their cheeks. During prolonged phonation of [e], arytenoid mobility can be judged and the laryngeal ventricle may be seen, and a slight distention of the pyriform sinuses may also allow better delineation of the aryepiglottic folds.1 The added radiation dose for such information is not justified as a routine protocol.
Standard data acquisition is from the base of the skull through the thoracic inlet to about the T2 vertebral level. For nonneoplastic conditions, the upper extent of the acquisition may be limited to the angle of the mandible. Targeted higher-resolution images from the hyoid to the cricotracheal junction may be reconstructed from a properly acquired initial data acquisition.
Technical Factors
Volume acquisition of data is the now the most common practice. The general neck examination should be reconstructed from the data set at 2 to 3 mm slice thickness (SLT) and 14 to 16 cm field of view (FOV) to include node-bearing areas. Targeted smaller FOV images of the larynx and hypopharynx should be reconstructed at an FOV of about 10 cm and an SLT of 1.0 to 1.5 mm. In noncancer cases, sections through the larynx should be reconstructed at 1 to 2 mm SLT and 9 to 11 cm FOV. This reconstructed data set facilitates excellent-quality reformatted images when necessary (Fig. 201.2).
If only one acquisition through the neck is done for any indication, it should be reconstructed at no more than 1.5 to 2 mm SLT so that adequate resolution is possible in multiplanar reformations.
Contrast Infusion
The standard injection rate for contrast infusion is 1.0 to 1.5 mL per second. A total of 100 to 125 mL of contrast is all that is normally necessary; this can be tailored to patient size and weight and less contrast used in smaller individuals. There should be enough delay to allow about 70 to 75 mL of contrast flow before the start of scanning assuming the initial acquisition will take about 10 seconds. Another 30 to 50 mL can run in during the second, more focused acquisition.

FIGURE 201.1. Contrast-enhanced computed tomography study of the larynx in two patients done with the patients breath-holding. A, B: Patient 1. In (A), at the true vocal cord level, the glottis is closed and only a slitlike airway remains. The arytenoids (arrowheads) are rotated medially. The patient did a slight Valsalva during breath-holding so that the apex of the pyriform sinus would be visible adjacent to the cricoarytenoid joint (arrow). In B, the false vocal cord level again shows the now false cords apposed in the midline and arytenoids (arrowheads) rotated medially and apex of the pyriform sinus open (arrow). C–E: Patient 2. In (C), physiologic anterior (white arrow) and posterior (arrowheads) commissure thickening is shown. Also note the only small amount of paraglottic fat (black arrows). In (D) and (E), the results are shown of a substantial modified Valsalva with distension of the pyriform sinuses (arrows) and the aryepiglottic folds compressed to thin bands (arrowhead).

FIGURE 201.2. Sagittal reformatted computed tomography image of the larynx in the midline. The quality of reformation is excellent. This view provides fairly strategic information about the relationship of the pre-epiglottic space (arrow) to the tongue base (BOT), with delineation of the free margin of the epiglottis (white arrowhead) and hyoid bone (black arrow) as prominent landmarks.
Indications for intravenous contrast laryngeal studies include determining the extent of primary cancers; detecting and determining the extranodal extent of cervical metastatic disease; and evaluating infiltrating, inflammatory, and mass lesions of uncertain etiology. Patients suffering laryngeal trauma and those with subglottic and/or tracheal stenosis are generally studied without intravenous contrast.
Magnetic Resonance Imaging
A discussion of the use of magnetic resonance imaging with regard to the larynx is presented in Appendix B.
Detailed imaging of the larynx requires anterior and posterior element receiver coils that are well matched to the size of the neck. Sections should be no thicker than 3 to 4 mm and have an FOV no larger than 14 to 18 cm. Axial images are the mainstay. This SLT is obviously not optimal for study of the larynx, but currently it remains the best compromise. Three-dimensional Fourier transform acquisitions are not practical because of the respiratory motion and swallowing that are virtually unavoidable in magnetic resonance imaging (MRI) of the larynx and hypopharynx.
Sagittal images in supraglottic tumors should show the proximity of tumor to the anterior commissure and the proximity of cancer of the tongue base. Coronal magnetic resonance (MR) images are used to assess the relationship of tumors to the true cord, false cord, laryngeal ventricle, and paraglottic space (PGS) that may not be clear on axial images.
Ultrasound
There is really no use for ultrasound in the evaluation of laryngeal lesions. For a brief period in the 1980s, it was suggested that it might be useful to look for cartilage invasion in laryngeal cancer. Ultrasound is used in some practices as an adjunct to evaluate nodes in patients with cancer of the larynx. However, ultrasound cannot be used to evaluate the important retropharyngeal lymph node group definitively.
Radionuclide Studies
Standard fluorine-18 2-fluoro-2-deoxy-D-glucose positron emission tomography (FDG-PET) and positron emission tomography (PET)/computed tomography (CT) techniques are used in selected patients for specific indications. The primary use is for suspected posttreatment recurrence. In this population of patients, its use may be justified in screening for lung cancer, especially in patients with supraglottic cancer in whom the rate of second primary lung cancer is particularly high. It may also reasonably be used in advanced-stage cancer to screen for distant metastases. The use in staging laryngeal cancer must have a justified and reasonable role in affecting medical decision making. Indiscriminant use is not reasonable and wastes medical resources.
Nondiagnostic CT done for PET localization cannot be substituted for proper anatomic studies of laryngeal cancer intended for use in clinical decision making. It is currently not possible to do adequate-quality, diagnostic anatomic CT of the larynx in conjunction with PET.
Pros and Cons
Indications for Study
Evaluating the Extent of Known Laryngeal Cancer and Related Regional Lymph Node Metastases
Both CT and MR can supply information needed by the clinician for optimal treatment planning (Chapter 206). CT has a number of important advantages. It is more widely available and costs less. The CT studies are much easier to execute and are much more reproducible than MRI because of the short data acquisition time that results in less image motion degradation. Multidetector computed tomography (MDCT) systems also now offer high-quality multiplanar imaging, with the image quality equal to or exceeding that of multiplanar MR images. These same qualities make CT preferable for surveillance imaging, including early detection of tumor recurrence and/or treatment complications such as osteoradionecrosis of the larynx.
Evaluation of Submucosal or Perilaryngeal Masses of Uncertain Etiology
Both CT and MR can be used to demonstrate the presence, nature, and extent of a submucosal mass lesion (Chapters 203, 205, and 206). In most cases, a CT study is adequate for all of these tasks. A variety of sometimes unusual lesions, either benign or malignant, can be encountered in this clinical context. As the initial biopsy results may be inconclusive or negative, MR may be used in an attempt to further characterize the lesion. In young patients, MR might be preferred because of the absence of ionizing radiation.
Dysphonia and Vocal Cord Dysfunction and Palsy
TVC dysfunction believed clinically to be due to recurrent laryngeal or vagal neuropathy will occasionally be caused by an entirely submucosal laryngeal pathology; the most frequent cause is cancer (Fig. 206.3). CT is the preferred screening tool in these patients.
CT and/or MRI are otherwise very useful in patients with vocal cord paralysis and no obvious endolaryngeal lesion. In case of isolated vocal cord paralysis, the lesion is more likely on the course of the recurrent laryngeal nerve, and CT is the imaging modality of choice. The entire course of the recurrent laryngeal nerve must be studied, meaning that in a case of left-sided vocal cord paralysis, the study should extend as low as the aortopulmonary window.
If the vocal cord paralysis is accompanied by symptoms related to dysfunction of other lower cranial nerves, then the offending lesion is very likely situated above the level of the hyoid bone. In such circumstances, MR is preferred and should be directed at searching for a lesion along the proximal part of the vagus nerve, from its origin in the brain stem to the level of the hyoid bone as discussed in Chapter 138.
Laryngeal Trauma
CT is the preferred modality for laryngeal trauma, as it can be performed more simply than MRI in the acute or immediate subacute clinical setting and provides superior cartilage detail and multiplanar images. It simultaneously allows a thorough evaluation of the other neck structures, including soft tissue injury that might be associated with pharyngeal tears, vascular injury, and cervical spine injuries, among others, discussed in Chapter 207.
Subglottic and/or Tracheal Stenosis
CT is the preferred modality for subglottic and/or tracheal stenosis, as imaging usually needs to determine only the degree and extent of airway narrowing. With MR, motion artifacts are a problem especially in patients with dyspnea. However, functional airway studies of the trachea are possible with dynamic MR techniques and now with 320 MDCT or equivalent units (Chapters 208 and 214).
Controversies
There is some debate with regard to whether CT or MR should be the primary imaging tool for evaluating patients with laryngeal cancer. This discussion often emphasizes which study is best at showing cartilage involvement. MRI seems to be a more sensitive technique than CT to detect cartilage abnormalities.2 However, MR also results in a considerable number of false-positive readings of cartilage invasion. In some practices, cartilage invasion is an absolute indication for total laryngectomy, thus eliminating as an option a treatment plan aimed at conserving a functional larynx. By combining several diagnostic CT criteria into a risk profile model, a reasonable accuracy for predicting outcome in patients who may have minimal cartilage invasion can be developed. Recent studies challenge the belief that laryngeal cartilage invasion alone dictates an unacceptably low probability of cure by irradiation and/or an increased risk of late complications. This is discussed in much more detail in conjunction with laryngeal cancer evaluation in Chapter 206.
MRI is often degraded by motion and lacks sections <3 mm; both of these factors are not part of a satisfactory profile for a first-choice imaging study of the larynx.
NORMAL ANATOMY
General Description
Laryngeal Skeleton
The laryngeal skeleton supports the functional soft tissue structures of the larynx and includes the cricoid, arytenoids, and thyroid cartilages and the hyoid bone. The hyoid and cartilages and their connecting membranes are the logical starting point for organizing this anatomy. The laryngeal skeleton has a unique appearance on any section through the larynx, allowing rapid orientation on any given laryngeal CT or MR image (Figs. 201.3 and 201.4).
Deep Tissue Planes
The PGS is that part of the larynx containing all of the paired structures of the larynx normally not seen by the clinician. Extending this concept to include the paired deep spaces of the subglottis and the pre-epiglottic space (PES) brings to light the major contributions of CT and MRI in laryngeal diagnosis (Figs. 201.5 and 201.6). These sectional imaging techniques show the deep planes. The clinical examination, including direct and indirect laryngoscopy and biopsy, evaluates the mucosa. The imaging and clinical evaluations are therefore inherently complementary exercises, and each yield important diagnostic information not directly available from the other.


FIGURE 201.3. Three drawings (A–C) from various perspectives illustrating the important relationships between the laryngeal cartilages and connecting membranes and some of the related musculature.
The conus elasticus and, less importantly, the quadrangular membrane (Fig. 201.7A,B) are elastic tissue structures that separate the various deep compartments. These structures are not seen as distinct structures. However, the conus elasticus directs the spread of pathologic processes and by this is “seen” indirectly.
The conus elasticus anchors to the upper margin of the cricoid arches, with its free upper border thickening to become the vocal ligament (Figs. 201.3. 201.7, and 201.8). It is also known as the cricothyroid membrane. It separates the deep planes of the subglottis into an anterior wedge of tissue bound anteriorly by the cricothyroid ligament and two lateral compartments that are in continuity with the TVCs and PGS (Figs. 201.3 and 201.7A).
The supraglottic larynx also has an anterior and two lateral compartments by the quadrangular membrane. The membrane attaches to the lateral borders of the epiglottic cartilage and continues back to the arytenoid cartilages within the substance of the aryepiglottic folds (Fig. 201.7). The PES in the anterior midline separates the two lateral PGS (Figs. 201.2, 201.5, and 201.6).


FIGURE 201.4. Diagram illustrating axial orientation of the larynx based on primary recognition of laryngeal skeletal landmarks. (AEF, aryepiglottic fold; PYS, pyriform sinus; LV, laryngeal ventricle; PES, pre-epiglottic space; E, epiglottis; SC, superior cornu thyroid cartilage; SCM, sternocleidomastoid muscle; CCA, common carotid artery; IJV, internal jugular vein; HB, hyoid bone; BT, base of tongue; GEF, glossoepiglottic fold; PEF, pharyngoepiglottic fold; TVC, true vocal cord; IS, infrahyoid strap muscles; PS, paraglottic space.)
Detailed Description of Specific Areas
Laryngeal Skeleton
Cricoid Cartilage
The cricoid cartilage is the foundation of the larynx. It is the only complete ring in the airway. It has a broad portion posteriorly that is known as the lamina; the anterior part of the ring is the cricoid arch (Figs. 201.3, 201.4, 201.8, and 201.9–201.12). The inferior cornua of thyroid cartilage articulate with the cricoid near the cricotracheal junction posteriorly to form the cricothyroid joints.

FIGURE 201.5. Diagram showing the relationship of various regions of the larynx to its spaces.
The lamina of the cricoid measures about 2 to 3 cm from superior to inferior. The cricoid slopes from the margins of the lamina posterior and superiorly to the arch anteriorly and inferiorly. At the mid subglottis, the inferior margins of the thyroid laminae lie lateral to the cricoid arches (Figs. 201.4 and 201.9–201.12).
The upper subglottis begins just below the undersurface of the TVCs and extends to the bottom of the cricoid cartilage (Figs. 201.13 and 201.14). The cricoid arches are less noticeable at this level, and the cricoid lamina mainly bridges the space between the paired thyroid cartilage laminae posteriorly (Figs. 201.4 and 201.9–201.12).
There is a depression of variable depth between the facets for the cricoarytenoid joint atop the cricoid lamina (Figs. 201.3 and 201.4). The cricoarytenoid joint facets and vocal process of the arytenoid identifies the mid plane of the TVCs.
In children and teenagers, the cricoid has a uniform density, similar or slightly higher than the surrounding soft tissues (Fig. 201.15). In adults, the cricoid very often has a calcified outer margin and low-density center similar to the cortex and medullary space of bones. A low-density medullary space often is not present within an only partially mineralized cartilage. Once the cartilages ossify, a true medullary space may develop partially or extensively within ossified segments of the cartilage (Fig. 201.16). Occasionally, a marrow space will be present only in areas of focal ossification, creating an islandlike focal nodular appearance of such an ossified area (Fig. 201.17).
Sometimes, a very dense calcification is seen along the posterior margin of the cricoid arch that in the appropriate clinical setting might be misinterpreted as a penetrating foreign object3 (Fig. 201.18). Such a dense calcific margin of the posterior cricoid rim may be asymmetric (Fig. 201.19). The median posterior part of the cricoid often remains nonossified (Fig. 201.20). A depression of variable depth may be present on the posterior side of the cricoid arch along the attachment of the posterior cricoarytenoid muscles (Figs. 201.18 and 201.20).

FIGURE 201.6. Sagittal diagrams of the larynx relating the regions of the larynx to critical anatomy and deep spaces.
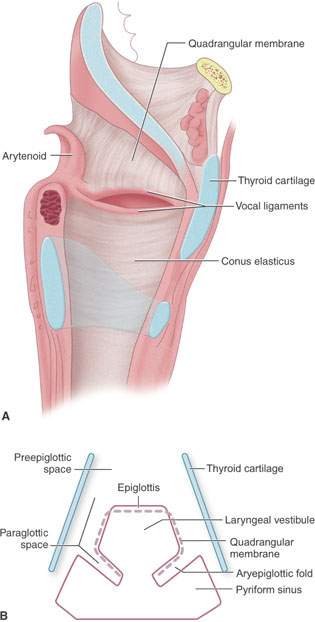
FIGURE 201.7. Diagrams demonstrating the relationship of deeper membranes and spaces within the larynx. A: Whole origin diagram showing the relationship of the quadrangular membrane to the supraglottis and conus elasticus to the subglottis. B: Schematic diagram showing the relationship of the deep spaces of the larynx within the supraglottic larynx and more superficial landmarks.

FIGURE 201.8. Diagram showing the relationship of the true vocal cords to the laryngeal skeleton and various connecting membranes. In (A), the cords are abducted. In (B), the vocal cords are adducted.
MRI shows a relative lack of signal from the ossified edges of the cricoid and a relatively high signal on non–fat-suppressed sequences from the more central fatty marrow (Fig. 201.21). The MR signal from nonossified portions of the cricoid varies with its extent of ossification.
Thyroid Cartilage
The thyroid laminae fuse anteriorly, forming the thyroid angle and creating a shield-shaped structure that protects the soft tissues of the larynx. The typical inverted V-shaped orientation of the laminae is a useful landmark at the level of the TVC. The fused anterior contour of the laminae appears more rounded in the upper to mid subglottis (Figs. 201.3. 201.4, 201.8, and 201.22–201.25). As the inferior cornua articulate at the cricothyroid joint, the joint allows the cricoid and thyroid to be tilted; thus, the distance between their anterior margins may vary. The thyroid cartilage appearance changes about 1 cm above the glottis, where the thyroid notch separates the two laminae. The superior cornua of the thyroid laminae are posterior structures that attach to the hyoid by the lateral thyrohyoid ligament (Figs. 201.3, 201.4, and 201.26–201.29). This lateral part of the thyrohyoid ligament contains a small sesamoid structure located between the tip of the superior horn of the thyroid cartilage and the greater horn of the hyoid bone (Fig. 201.13). These triticeal cartilages may calcify and become visible on CT. This thyrohyoid ligament is merely a thickening of the posterior free margin of the thyrohyoid membrane. The thyrohyoid membrane is not normally seen as separate from the infrahyoid strap muscles. This muscle group creates a tissue-density band lying along the thyroid laminae maintaining this relationship between the superior margins of the thyroid cartilage and the hyoid (Fig. 201.1).
Thyroid cartilage mineralization and ossification vary tremendously. The ossification typically starts from a primary ossification center at the posteroinferior border of both thyroid laminae and/or one of several secondary ossification centers. During this slowly progressive process, islands of nonossified cartilage are often formed (Figs. 201.30 and 201.31). The thyroid laminae tend to be more calcified (ossified) in males and with increasing age (Fig. 201.13). In teenage patients, mineralization is normally sufficient for diagnostic purposes. Minor to moderate asymmetry of thyroid cartilage calcification pattern is the rule in adults (Figs. 201.32 and 201.33).
In adults, thyroid laminae most often show peripheral dense calcification with a more hypodense and sometimes fatty central space. When the laminae are ossified, this becomes a true medullary cavity. Focal areas of ossification or expansion are common. The laminae are often thin in the paramedian region, especially at the glottic level. At areas of maximal thinning, the internal and external surfaces may almost merge and obliterate the intervening space.
On MR images, the margins of an ossified thyroid cartilage appear as signal voids. Fat within the ossified portions of the laminae will have a high signal on non–fat-suppressed images. Focal or broader areas of ossification and mineralized cartilage have variable but generally lower signal intensities (Fig. 201.34). The intensity of the infrahyoid strap muscles contrasts well with the adjacent fat and laryngeal skeleton.

FIGURE 201.9. Anatomic diagrams to correlate with anatomy demonstrated on computed tomography and magnetic resonance images seen in Figures 201.10 through 201.12.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree