The management of neck nodes in head and neck cancer is critical, given a markedly increased poor prognosis in patients with nodal metastasis. The surgical management of neck nodes has undergone radical changes secondary to a paradigm shift from curative surgery to nonsurgical organ and function-preserving options, such as radiation therapy. In the neck after treatment, radiologists should be familiar with imaging findings in various types of neck dissections and post-chemoradiation changes, along with signs of residual or recurrent disease. A multidisciplinary approach is essential with well-designed evidence-based surveillance imaging protocols and standardized reporting.
Key points
- •
The management of metastatic nodal disease is a key part of treatment of head and neck cancers.
- •
The neck can be managed with a variety of surgical methods, as well as by radiation with or without chemotherapy.
- •
Knowledge of the typical methods and extent of nodal dissections (as well as the related understanding of the typical nodal basins involved for different primary tumor sites) aids interpretation of post-treatment imaging and further treatment planning.
- •
To decrease variability, reporting methodology from the Neck Imaging and Reporting Data System should be used for post-treatment surveillance imaging.
Introduction and history
Metastatic lymph node spread of squamous cell carcinomas of head and neck portends poor prognosis and survival. Neck dissection in its different forms comprises the bulwark of surgical treatment for clinical and subclinical metastatic neck cancer. Although neck dissections have been performed for the past 200 years, the major underlying principles of neck dissection surgeries are based on seminal works of George Washington Crile and Hayes Martin in the early and mid-20th century. Martin in his landmark article titled “Neck Dissection” in 1951 emphasized that the spinal accessory nerve, internal jugular vein, and the sternocleidomastoid muscle should be removed in the presence of cervical lymph node metastasis. This approach was the norm for head and neck surgeons until the latter part of the twentieth century, when modifications of the radical dissection technique started being accepted.
The morbidity associated with radical neck dissection (RND) especially related to shoulder function resulted in modifications, resulting in various conservative surgeries also referred to as functional neck dissections. The concept of selective nodal dissection was introduced by Byers in 1985. The present classification of neck nodal surgeries is based on Shah’s landmark article from 1990, where he classified the deep lymph nodes of the neck into 5 different levels assigned Roman numerals from I through V. In 2002, the current standard classification of neck dissections was proposed by the American Head and Neck Society and the American Academy of Otolaryngology–Head and Neck Surgery, which classifies and describes radical neck dissection, extended neck dissection, modified radical neck dissection and selective neck dissection.
Four major changes over the last 40 years have led to a continued refinement and evolution of the neck dissections. The first of these was the advent of cross-sectional imaging, including computed tomography (CT), MR imaging, and PET studies, which have led to more accurate preoperative assessment of neck disease and subsequent increase in selective nodal dissections. Second, the shift in thinking from the resection of tumors and wound closure to an increased emphasis on establishing normal function and appearance has led to newer techniques and advances in head and neck reconstructive surgery. A third major change has involved the remarkable improvements in radiotherapy (RT) over the past few decades, which has now led to a large subset of patients with head and neck cancer undergoing RT as their primary treatment or as adjuvant therapy after surgery, which has led to a gradual shift toward nonsurgical management. Last, the exponential increase in human papillomavirus (HPV)–associated oropharyngeal cancers involving a younger subset of patients, who also have better outcomes, has led to a fundamental rethinking of the role of surgery. As a consequence of all these improvements, a paradigm shift has taken place from curative primary surgery to function–preservation RT and other nonsurgical options in many patients.
Imaging in the post-treatment head neck
Imaging in the post-treatment neck depends on the early versus late stage presentation of the head and neck cancer, which dictates management as well as the extent of the disease. Typically, early stage head and neck cancers are treated with either RT or surgery. Locally advanced head and neck cancers without distant metastasis require multimodal treatment, which usually consists of a combination of curative surgery followed by adjuvant RT, with or without chemotherapy. The overall imaging appearance in the treated neck depends on effects of RT, surgery for the primary site (with or without reconstruction) and the type of neck dissection (radical neck dissection, selective neck dissection, etc).
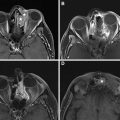
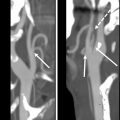
Early post-RT changes include reticulation of the subcutaneous fatty tissues; thickening of the skin, fascial tissues, and platysma; thickening with increased enhancement of the pharyngeal walls and mucosal surfaces; and hyperenhancement of the major salivary glands ( Fig. 1 ). Later findings include loss of neck fat, atrophy of the salivary glands, and thickening of fascia, platysma, and constrictor muscles (see Fig. 1 ).
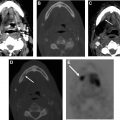
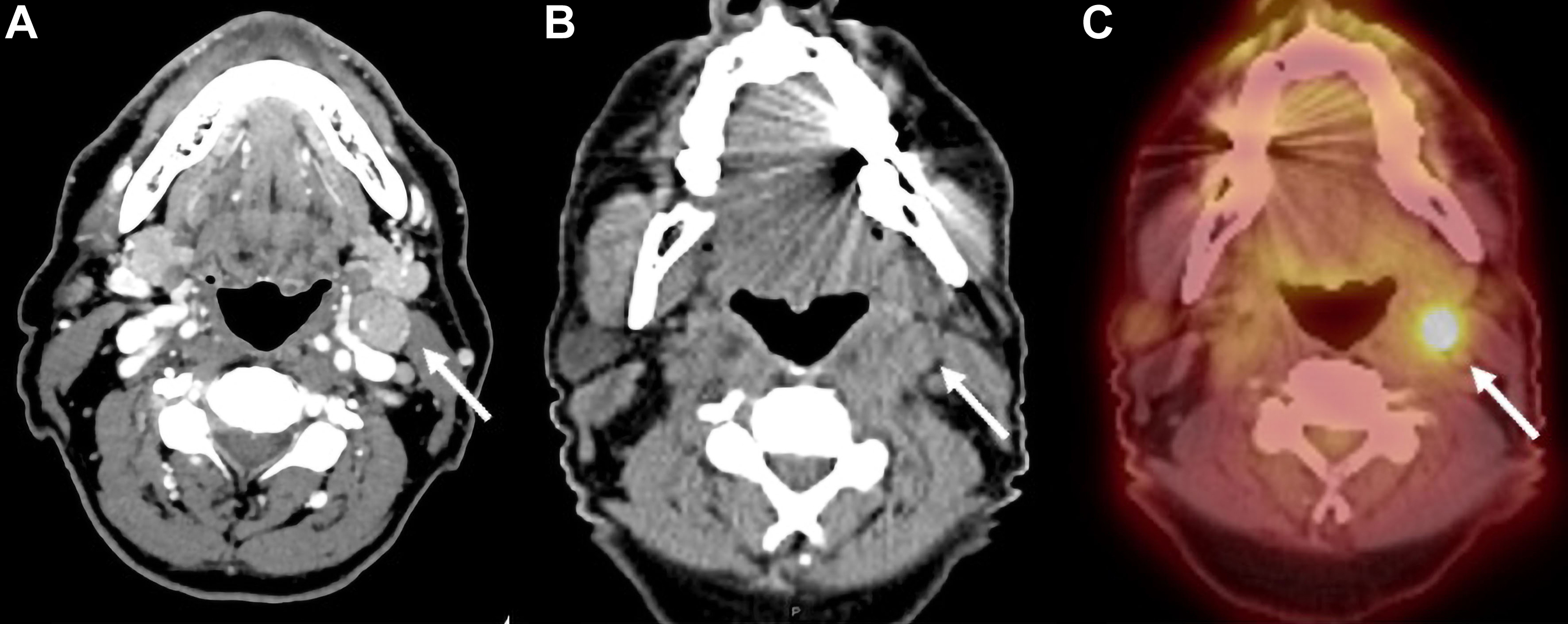
Expected changes after RT in the neck nodes include decreased size of pathologic nodes, development of irregular nodal margins, increased perinodal fat reticulation, and edema in the adjacent superficial and deep neck spaces along, with thickening of the adjacent fasciae ( Fig. 2 ). The decreased size of the larger pathologic neck nodes on a contrast-enhanced CT scan alone is not a reliable predictor of residual disease, and an accompanying PET scan with fluorine-18-2-fluoro-2-deoxy- d -glucose (FDG-PET) scan may be essential in excluding residual nodal disease (see Fig. 2 ).
The post-treatment appearance at the primary tumor location after surgery depends on the site and size of the primary defect, as well as the type of reconstruction performed. The changes after RT and complex neck reconstructions are covered in further details in separate articles elsewhere in this issue. The major types of neck dissection, the underlying principles and their imaging appearance are discussed elsewhere in this article.
In the post-treatment setting, both contrast-enhanced CT scans and contrast-enhanced MR imaging are relatively insensitive in identifying residual or recurrent nodal disease, with PET/CT scans having much greater sensitivity and specificity. Conventional imaging criteria for recurrent or residual nodal disease in a post-treatment neck include an increase in size (typically using similar size thresholds as those on staging scans), necrosis, abnormal morphology, or evidence of extranodal extension (ENE). A combined FDG-PET/contrast-enhanced CT scan represents the best imaging modality to exclude residual/recurrent cancers, especially in the early post-RT setting, and has the highest negative predictive value.
If the diagnosis is unclear on a CT scan or MR imaging, or tissue confirmation is necessary, most cervical nodes are readily accessible via fine needle aspiration. Ultrasound guidance is sufficient for most nodal groups, although some deeper nodes (ie, retropharyngeal) may require CT guidance.
Principles of neck dissection surgery
Squamous cell carcinomas of the head and neck commonly metastasize to cervical lymph nodes; even in patients without clinical or imaging evidence of nodal disease, occult metastases are often present. The management of the lymph nodes is a key part of improving survival, and can be accomplished by surgical resection or RT. For non–HPV-related disease, stages I and II suggest a small primary lesion without nodal disease, whereas stages III and IV include larger primary lesions with invasion of adjacent structures and/or lymph node metastasis.
Owing to the prevalence of occult nodal disease, elective neck dissections are often performed in patients who clinically have N0 disease and are undergoing surgical resection of the primary tumor. Early nodal disease (N1/2a) is also managed with selective neck dissection when the primary is treated surgically. In patients who present with locoregionally (N2b/2c/3) advanced disease who are being treated primarily with surgery, neck dissections (possibly radical or modified radical) are performed for treatment of the nodal disease as well as for staging.
Alternatively, in patients for whom the primary tumor is being treated with RT with or without chemotherapy, the neck may also be managed initially via (chemo)radiation with neck dissection deferred for use as salvage therapy if there is evidence of residual or recurrent nodal disease on clinical examination or imaging.
Nodal staging and changes in the American Joint Committee on Cancer/Union for International Cancer Control Cancer Staging Manual, Eighth Edition
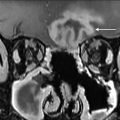
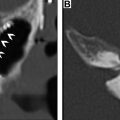
From an imaging perspective, a thorough understanding of the various radiologic landmarks, which are used as boundaries, is essential in accurate staging of lymph node groups ( Fig. 3 ). These boundaries have been summarized in Table 1 . Although a complete review of nodal staging for head and neck cancer is beyond the scope of this article, it is worthwhile to review some of the important changes made to the nodal staging in the eighth edition of the American Joint Committee on Cancer’s Cancer Staging Manual. ,
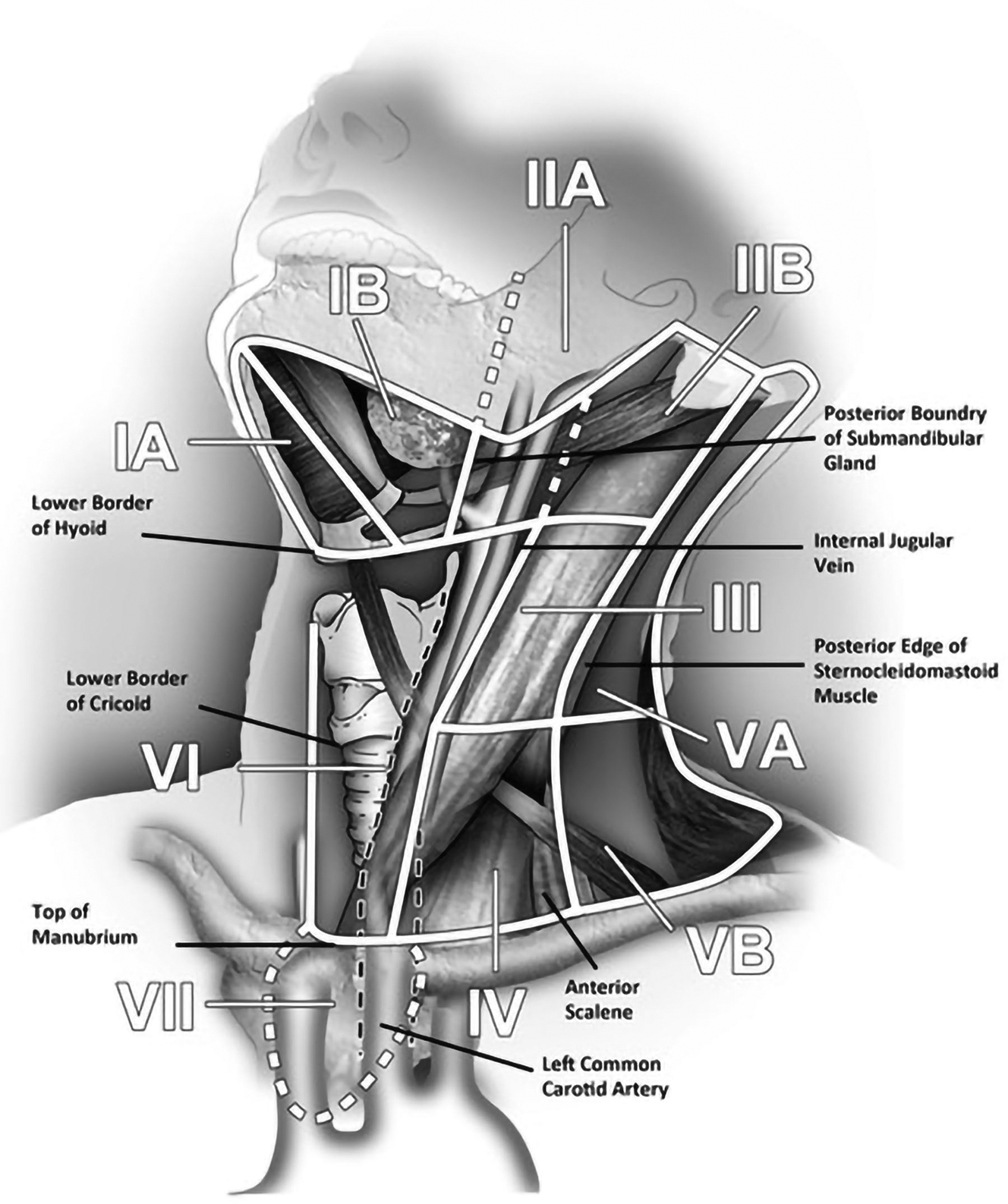
| Levels | Boundaries | |||
|---|---|---|---|---|
| Superior | Inferior | Anterior (Medial) | Posterior (Lateral) | |
| IA Submental group | Symphysis of mandible | Body of hyoid | Anterior belly of contralateral digastric muscle | Anterior belly of ipsilateral digastric muscle |
| IB Submandibular group | Body of mandible | Posterior belly of digastric muscle | Anterior belly of digastric muscle | Stylohyoid muscle |
| IIA Upper jugular group nodes | Skull base | Horizontal plane defined by the inferior border of hyoid bone | Stylohyoid muscle | Vertical plane defined by the spinal accessory nerve |
| IIB Upper jugular group nodes | Skull base | Horizontal plane defined by the inferior border of hyoid bone | Vertical plane defined by the spinal accessory nerve | Lateral border of the sternocleidomastoid muscle |
| III Mid jugular group | Horizontal plane defined by the inferior border of hyoid bone | Horizontal plane defined by the inferior border of cricoid cartilage | Lateral border of the sternohyoid muscle | Lateral border of the sternocleidomastoid muscle or sensory branches of the cervical plexus |
| IV Lower jugular group | Horizontal plane defined by the inferior border of cricoid cartilage | Clavicle | Lateral border of the sternohyoid muscle | Lateral border of the sternocleidomastoid muscle or sensory branches of the cervical plexus |
| VA Posterior triangle group | Apex of the convergence of sternocleidomastoid and trapezius muscles | Horizontal plane defined by the inferior border of the cricoid cartilage | Lateral border of the sternocleidomastoid muscle or sensory branches of the cervical plexus | Anterior border of the trapezius muscle |
| VB Posterior triangle group | Horizontal plane defined by the inferior border of cricoid cartilage | Clavicle | Lateral border of the sternocleidomastoid muscle or sensory branches of the cervical plexus | Anterior border of the trapezius muscle |
| VI Central or anterior group | Hyoid bone | Top of manubrium | Common carotid artery | Common carotid artery |
| VII Superior mediastinal group | Top of manubrium | Inominate vein | Common carotid artery | Common carotid artery |
One of the major changes involves introduction of different clinical and pathologic N classifications for staging regional lymph node metastasis. Both clinical TNM (cTNM) and pathologic (pTNM) classifications have different purposes. The cTNM classification applies to all patients whether they undergo surgery or nonsurgical therapy. The pTNM classifications provides additional information (high-risk histopathological features of the primary site, ENE, volume of nodal disease, etc), which is only available after surgery and allows for additional adjuvant treatment.
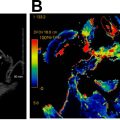
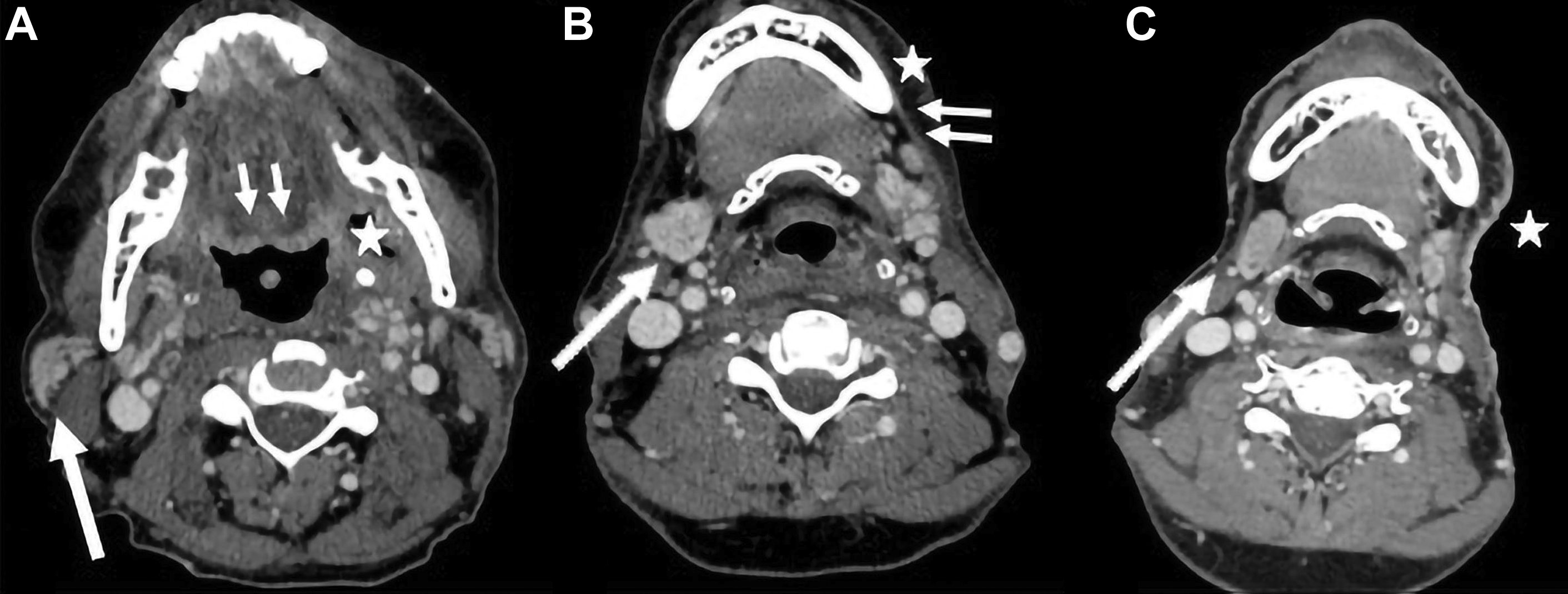
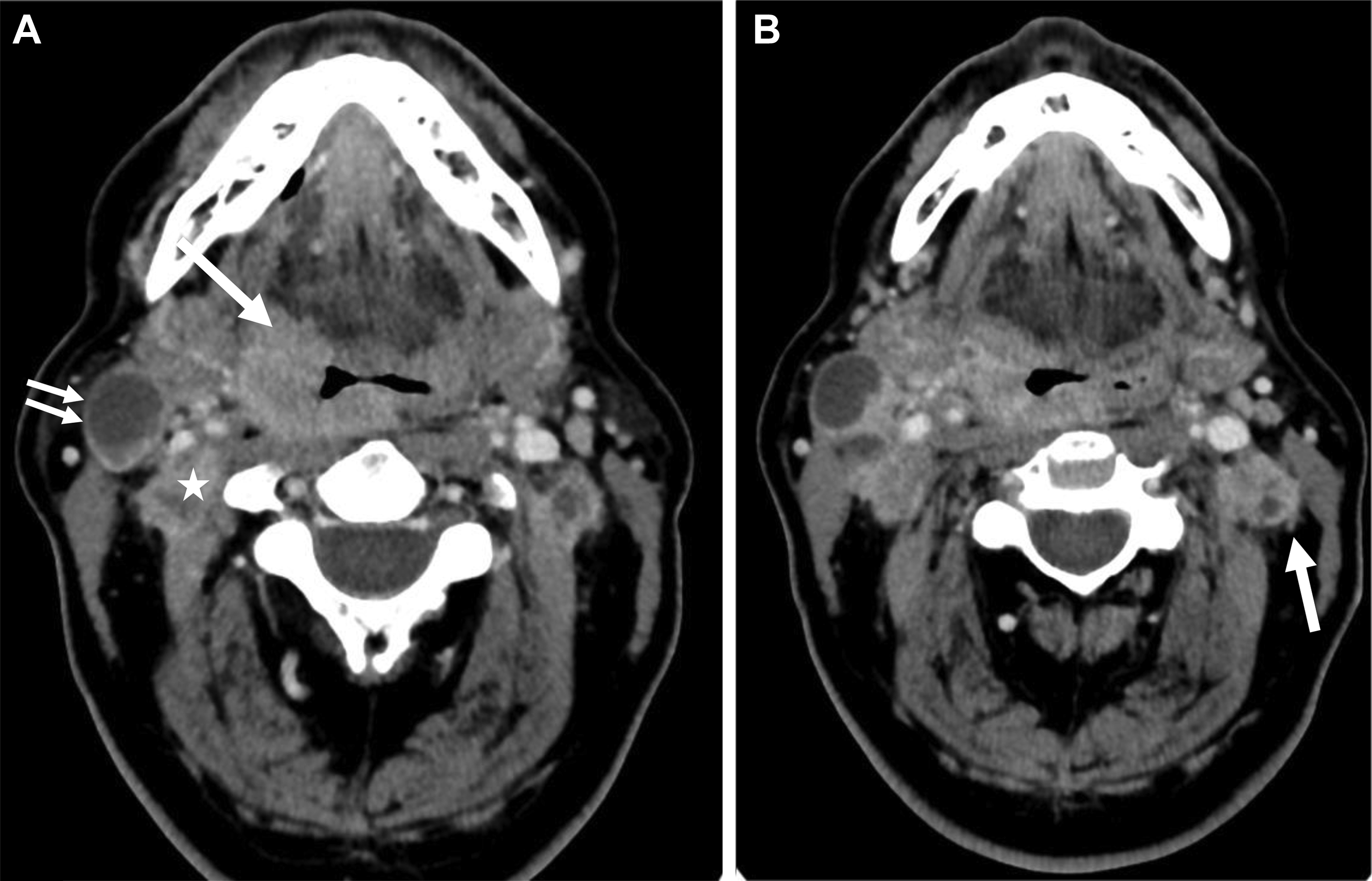
Another major change is the introduction of ENE, which is now a part of N classification for all head and neck sites other than nasopharyngeal and HPV-associated oropharyngeal cancers. Although imaging findings may suggest or support ENE (irregular margin, infiltration of adjacent fat planes or other soft tissue structures), imaging findings alone are not sufficient for the designation as ENE positive ( Fig. 4 ). Clinical ENE is defined as invasion of skin, muscle infiltration, tethering or fixation to adjacent structures, or cranial nerve, brachial plexus, sympathetic trunk, or phrenic nerve invasion with dysfunction ; alternatively, ENE can be diagnosed histologically after neck dissection.
A change in the prognostic significance of T and N stages of oropharyngeal cancer over time in part led to the adoption of a new staging system for HPV-associated (p16-positive) oropharyngeal cancers. The clinical nodal staging for HPV-associated oropharyngeal cancer in the eighth edition is based on node laterality and size, as summarized in Table 2 . Pathologic staging for HPV-associated oropharyngeal cancer, in contrast, is based on the number of nodes involved, as detailed in Table 3 . The clinical nodal staging for all non-HPV, non–Epstein-Barr virus squamous cell cancers is summarized in Table 4 . Last, nasopharyngeal carcinomas, which are usually Epstein-Barr virus related, and are often nonkeratinizing carcinomas, have a modified nodal staging, which is summarized in Table 5 .
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree