Proper performance of magnetic resonance neurography (MRN) is essential not only to make the examination easier to interpret but also for its accurate evaluation. This article outlines the technical considerations of MRN, various imaging pulse sequences available on current scanners, as well as their relative advantages and disadvantages. In addition, a guide to the optimal use of high-resolution and high-contrast MRN technique is provided, which will aid clinicians in attaining a good-quality examination.
Key points
- •
Magnetic resonance (MR) neurography (MRN) is a technically demanding examination requiring the knowledge of appropriate clinical questions, regional nerves, and perineural anatomy as well as the various available MR imaging pulse sequences.
- •
A good MRN examination should involve the referring clinician, protocoling radiologist, performing technologist, as well as the patient and interpreting radiologist as a team, and should follow the guidelines presented in this article.
Introduction
Magnetic resonance (MR) neurography (MRN) is a technique to enhance peripheral nerve visualization with a variety of available high-resolution and high-contrast nerve-nonselective and nerve-selective imaging pulse sequences. MRN has been performed for more than 2 decades with great emphasis being on two-dimensional (2D) imaging. Although 2D imaging remains the standard for the primary examination interpretation, high-quality three-dimensional (3D) imaging is essential for display and problem solving when current clinical and imaging evaluations show unclear or ambiguous results. A good MRN technique should enhance the visualization of peripheral nerves in various planes to provide assistance to the referring physician in understanding the disease process and its localization. This article discusses the technical considerations of MRN, various imaging pulse sequences available on current clinical scanners, as well as their relative advantages and disadvantages. In addition, a guide to the optimal use of high-resolution and high-contrast imaging technique is provided, which will aid clinicians in attaining a good-quality examination.
Introduction
Magnetic resonance (MR) neurography (MRN) is a technique to enhance peripheral nerve visualization with a variety of available high-resolution and high-contrast nerve-nonselective and nerve-selective imaging pulse sequences. MRN has been performed for more than 2 decades with great emphasis being on two-dimensional (2D) imaging. Although 2D imaging remains the standard for the primary examination interpretation, high-quality three-dimensional (3D) imaging is essential for display and problem solving when current clinical and imaging evaluations show unclear or ambiguous results. A good MRN technique should enhance the visualization of peripheral nerves in various planes to provide assistance to the referring physician in understanding the disease process and its localization. This article discusses the technical considerations of MRN, various imaging pulse sequences available on current clinical scanners, as well as their relative advantages and disadvantages. In addition, a guide to the optimal use of high-resolution and high-contrast imaging technique is provided, which will aid clinicians in attaining a good-quality examination.
Essentials of MRN technique
A good MRN examination should involve the referring clinician, protocoling radiologist, performing technologist, as well as the patient and interpreting radiologist as a team and should follow the guidelines presented in this article.
Referring Clinician
Close interaction between the referring clinician and radiologists is important for good interpretation of image findings. Clinicians should be informed about the technical limitation of MR imaging including the maximum size of field of view (FOV) as well as that the greater the anatomy to be covered, the longer the examination usually takes. Especially in older patients with peripheral neuropathies (eg, with tremor), reduced image quality must be expected. The use of gadolinium-based contrast agents should be discussed with regard to the various indications. In addition, preferred reconstructions should be discussed because some clinicians like maximum-intensity projections (MIPs) or 3D reconstructions. Although the latter may or may not useful for the radiologist’s interpretation, they might be an important preoperative guide for surgeons.
Patient Coaching
The technologist plays an important role in patient coaching for successful performance of the examination. The patient should fill out a form with most relevant acute/chronic complaints, any relevant electrodiagnostic information, history of diabetes mellitus/family history of neuropathy, and any prior regional nerve surgery, and so forth. A marker should ideally be placed at the most symptomatic site, and the patient should be asked to remain still during the image acquisition, as well as to breathe normally. The extremity should be well padded and coil(s) tightly wrapped around it to restrict motion during the examination. The urinary bladder should be voided in pelvic or lumbosacral (LS) plexus imaging because it interferes with the MIP images.
Scanner
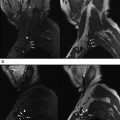
The imaging is ideally performed on a 3-T scanner to make use of the higher signal/noise ratio (SNR) available on these scanners. Higher SNR translates into higher contrast and potentially faster imaging, and the slice thickness is kept to a minimum ( Fig. 1 ). Minimal slice thickness results in excellent through-plane resolution for 2D sequences and thinner isotropic resolution for 3D imaging. If 3 T scanners are not available, 1.5 T can be used, with some drawbacks such as limits with respect to 3D imaging capability and prolonged examination times. Nevertheless, the authors encourage the use of MRN techniques for 1.5 T as well, because they show better performance than standard MR imaging techniques. Use of 1.5 T might even be advantageous, such as in patients with metal in the imaging FOV, for whom 3 T is expected to produce artifacts.
Coil Selection
Dedicated multichannel joint coils should be used in extremities for tunnel imaging (carpal, cubital, and tarsal tunnel) and proper use of parallel imaging should be used with acceleration factors of 2 to 3. For additional contiguous area imaging, multichannel flex (surface or body matrix) coils can be tightly wrapped around the larger extremity portion, such as upper arm, forearm, or thigh and can be combined with joint-specific coils, if needed. Clinicians should not try to wrap the joint and extremity together and image them both in the same FOV, because this produces low-quality images with a lot of blank (air) space around the region of interest. For pelvic, LS, or brachial plexus imaging, the spine array coils can be combined with body array coils on the front of the patient; in some systems, dedicated array designs may be used.
FOV
FOV should be determined based on the physician request and should be kept as small as possible, both in longitudinal and transverse planes, to obtain high-resolution imaging. Therefore, clinicians should describe the location of the lesion as precisely as possible (discussed earlier). However, lesion localization is often difficult clinically and, this is frequently the main indication for the imaging. Blank space around the extremity should be no more than approximately 20% of the diameter of the extremity for optimal assessment of small nerves. However, this method may result in wraparound artifacts, especially in off-center imaging. Phase oversampling or fold-over suppression techniques may be used to avoid such artifacts ( Fig. 2 ). In the case of nonspecific or nonlocalizing symptoms, the radiologist should make use of all available clinical and electrodiagnostic test information to best tailor the high-resolution examination across the known injury or entrapment sites of the extremity and perform the remaining examination as well-spaced images (axial and coronal thickness 5–6 mm) to keep the imaging time of the study short and within acceptable limits (less than 45–60 minutes).
2D Imaging
High-quality 2D imaging with both non–fat-suppressed T1-weighted (W) and fat-suppressed fluid-sensitive T2-W images is essential. These images should be sequential and ideally be obtained at similar table positions to allow the reader to evaluate the images with different tissue contrasts in tandem for various intraneural and perineural nerve anatomic and pathologic imaging characteristics. In case of patient motion, the imaging should be repeated and patient coaching may be required. The coronal and sagittal planes should be obtained in long axes, parallel and perpendicular to the extremity and not the body. For smaller nerves in the hindfoot, nerve-perpendicular, oblique axial images across the axis of calcaneus are useful for optimal evaluation of medial plantar, lateral plantar, and calcaneal nerves ( Fig. 3 ). It is usually helpful for the radiologist to control the correct image planes during the MR examination and to adjust or add planes and sequences, respectively.
2D Imaging Parameters
Axial images should have in-plane resolution of 0.3 to 0.4 mm and should not have slice thicknesses of more than 2 to 3 mm in distal extremities, and no more than 4 to 5 mm in proximal extremities with minimal or no interslice gap. For fluid-sensitive T2-W images, the fat suppression should be homogeneous and the effective echo time (TE) should be 60 to 70 milliseconds to minimize magic angle artifacts. Axial short tau inversion recovery (STIR) imaging is not routinely used in our practice, but if used in cases of failed fat suppression, should have a lower TE (30–50 milliseconds) to maintain good SNR. Dixon-type fat suppression is a good alternative in regions with inhomogeneous fat suppression using standard techniques (discussed later). The echo train length can vary from 3 to 8 for T1-W, 8 to 22 for T2-W, and 44 to 68 for 3D imaging. Parallel imaging with an acceleration factor of 2 to 3 is frequently used.
3D Imaging
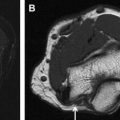
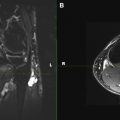
3D imaging is essential for good multiplanar nerve display and is currently available as high-contrast spin-echo type multislab acquisition on most vendor types. 3D imaging can be obtained with or without fat suppression. For non–fat-suppressed 3D T2-W imaging, the effective TE should be more than 90 milliseconds (ideally 100–105 milliseconds), and for 3-D fat-saturated (fs) T2 W imaging, effective TE can be lowered to 60 to 80 milliseconds. It can be obtained in acceptable imaging times of 5 to 6 minutes with isotropic resolution of 1 to 1.5 mm. MIP with or without curved planar reconstructions from 3D fsT2 W imaging allows excellent nerve visualization, and the abnormal nerve and the related lesion is easier to depict because the nerve gets brighter with abnormality ( Fig. 4 ). Gradient echo–type 3D imaging should be avoided because it is more prone to susceptibility artifacts and produces poorer contrast. However, it is frequently used with contrast imaging to obtain isotropic multiplanar depiction of contrast-enhancing lesions ( Fig. 5 ). With the addition of diffusion weighting, nerve-selective and functional images can be obtained with effective vascular signal suppression, as discussed later. Shimming is essential for any diffusion imaging to obtain the maximum field homogeneity and good fat suppression.

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree