Scrotum
Normal Anatomy
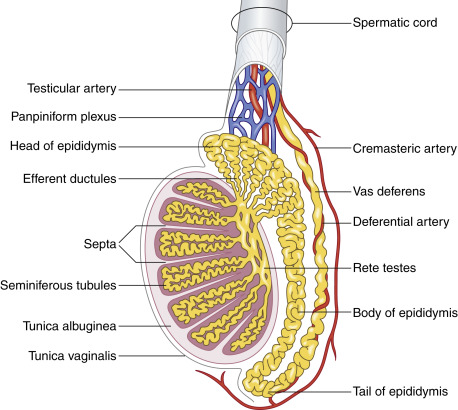
The surface of the testis is surrounded by the tunica vaginalis, which extends to the inner surface of the scrotal wall in continuity. A trace amount of fluid in between both layers allows a high mobility of the testis. This arrangement is similar to the pleura with the visceral and parietal layers that allow the lung excursion. Adult measurements of the testis are variable, in the range of 3 to 5 cm in length, 2.5 cm in anteroposterior diameter, and 3 cm in transverse diameter. The testis is surrounded by a fibrous capsule, the tunica albuginea. It cannot be recognized as such on scanning, but it is important because its smooth contour is disrupted with testicular trauma. The seminiferous tubules of the testes are separated by septa. They drain into the rete testes and then into the epididymis ( Fig. 22.1 ). The epididymis sits along the surface of the testis. It is classically divided in a head, body, and tail, from superior to inferior. Normally, only the head is visualized, measuring approximately 1 to 2 cm in diameter. The vascularity of the epididymis is less than that of the adjacent testis. The tail of the epididymis drains into the vas deferens, located in the spermatic cord. The testis is suspended by the spermatic cord that descends from the abdomen into the scrotum through the inguinal canal. The cord contains the vas deferens carrying the sperm to the ejaculatory ducts, the spermatic artery to the testis, other arteries, pampiniform plexus (venous), lymphatics, and nerves (see Fig. 22.1 ).
Ultrasound Technique/Findings
The patient should be in the recumbent position with the scrotum exposed over a towel. Starting with an axial view of both testes in grayscale and color Doppler, the procedure should be done with a high-resolution convex or linear transducer with convex settings. The grayscale comparison views of each testis are helpful to show symmetry between the two testes. The comparison color flow views of the testes are extremely helpful to detect potential abnormalities associated with either increased or decreased flow. With those views, the imager should get an immediate understanding of asymmetries in echogenicity, size, and perfusion between both sides ( Figs. 22.2 and 22.3 ).


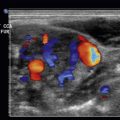
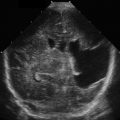
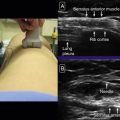
Then, with a high-resolution linear transducer, each testis, epididymis, tunica vaginalis, scrotal wall, and spermatic cord should be examined in grayscale and color Doppler. On ultrasound, the multiple histologic layers of the scrotal sac are seen as a single layer. Only with scrotal edema is the multilayered appearance of the scrotal sac seen, resembling an “onion skin.” The examination should include longitudinal views of each testis, including the epididymis, using both grayscale and color flow. Begin with the nonaffected side to set the technical parameters before examining the side of concern. Both testes are oval structures of homogeneous, midlevel echogenicity with their long axis in the superoinferior body direction ( Fig. 22.4 ). This is important in cases of torsion, where the long axis of the testis may rotate 90 degrees. A linear area of echogenicity in the periphery of the testis represents the mediastinum, where the seminiferous tubules and vascular structures exit and enter the testis (not to be confused with a testicular laceration) ( Fig. 22.5 ). Flow to the testes should be evaluated with color and pulsed wave Doppler, as the normal testis normally shows forward flow in systole and diastole, as do all other organ parenchyma. There is color flow in both the normal testis and the epididymis ( Fig. 22.6 ). Any reduction, absence, or reversal of diastolic flow is indicative of high resistance to flow such as seen with edema, ischemia, or venous compromise. The epididymis is isoechoic or slightly less echogenic than the testis. On ultrasound, the spermatic cord is observed as a laminar structure, not exceeding a caliber of 1 cm, without twists (important in cases of torsion). It is easier to identify with deep breaths, as the cord is observed moving up and down ( Fig. 22.7 , ![]() Video 22.1 ).
Video 22.1 ).




Acute Scrotum: General Considerations
Acute scrotum is defined as an acute painful swelling of the scrotum or its contents accompanied by local signs and possible general symptoms. Possible etiologies include epididymo-orchitis, testicular ischemia, trauma, torsion of testicular appendages, and complicated inguinal/femoral hernias. The incidence is variable, but their early correct diagnosis is extremely important because of a possible testis loss in cases of mismanagement.
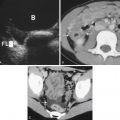
Since the advent of medical diagnostic ultrasound several decades ago, this tool has remained the best modality in the diagnosis of scrotal/inguinal pathology, despite the development of newer and more complex tools, such as computed tomography (CT), magnetic resonance imaging, and positron emission tomography. These modalities are of value when scrotal pathology extends beyond this region, such as with metastatic tumors.
As is well known, ultrasound has an exquisite image resolution in the range of millimeters, has the capability of real-time imaging (useful to observe changes with the Valsalva maneuver), and demonstrates blood flow (useful in cases of ischemia or inflammation). It is readily available at most medical institutions; it is less expensive than the other mentioned modalities and does not expose the patient to ionizing radiation.
Inflammation: Epididymitis and Epididymo-orchitis
Inflammatory changes of the scrotum are mostly bacterial in origin. It is one of the most common diagnoses in men from the teenage years to middle age. The most common presentation is in the form of acute epididymitis, which may extend to the testis, resulting in epididymo-orchitis. In sexually active patients, it results from bacteria ascending in the urethra through the vas deferens and reaching the epididymis. The main causal etiology in men younger than 35 years is Chlamydia , followed by Neisseria gonorrhoeae, Treponema pallidum, Trichomonas, and Gardnerella vaginalis. Urinary coliforms such as Escherichia coli, Pseudomonas species, Proteus species, and Klebsiella are seen in children and older men. In homosexual males, coliforms are prevalent.
In young children, infection may be due to congenital malformations. In older men, it may be due to bladder outlet obstruction or be secondary to urethral stricture or prostatic hyperplasia, resulting in urinary reflux into the ejaculatory ducts and vas deferens. Other causes of infection in the older group are urethral/prostatic instrumentation and bladder catheterization.
Infrequent isolated orchitis, without epididymitis, may be seen with mumps, with a preceding general malaise and parotiditis.
Clinical presentations favoring inflammatory disease rather than torsion are the following: gradual progression of unilateral scrotal pain and swelling, urinary symptoms, urethral discharge, possible fever/chills (more so in children), and absence of nausea/vomiting.
The ultrasound appearance of epididymitis includes edema and hypervascularity of the epididymis with enlargement, decreased/heterogeneous echogenicity, and increased Doppler flow ( Fig. 22.8 , ![]() Video 22.2 ). When the inflammation extends to the testis, similar findings to those in the epididymis are observed in the testis (
Video 22.2 ). When the inflammation extends to the testis, similar findings to those in the epididymis are observed in the testis ( ![]() Fig. 22.9 , Video 22.3 ). Frequent associated findings are reactive hydrocele, pyocele, and scrotal wall edema/hyperhemia ( Fig. 22.10 ). With reactive hydrocele, the fluid is clear. With pyocele, the fluid is echogenic with pseudomembranes (fibrin). Possible complications of inflammation are abscess ( Fig. 22.11 ) and testis ischemia, resulting in segmental or complete testis infarction.
Fig. 22.9 , Video 22.3 ). Frequent associated findings are reactive hydrocele, pyocele, and scrotal wall edema/hyperhemia ( Fig. 22.10 ). With reactive hydrocele, the fluid is clear. With pyocele, the fluid is echogenic with pseudomembranes (fibrin). Possible complications of inflammation are abscess ( Fig. 22.11 ) and testis ischemia, resulting in segmental or complete testis infarction.




Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree