Chapter 3 Mammographic Analysis of Breast Calcifications
Technique for Finding Calcifications
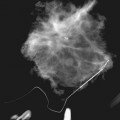
On the other hand, air-gap magnification mammography with a 0.1-mm focal spot actually increases the resolution power of the imaging system by about 1.8 times normal and shows more calcifications than were present on the original image. Air-gap magnification separates closely grouped calcifications into their individual forms, displays faint calcifications not detected at screening, and sharpens the image (see Fig. 1-2). Thus, air-gap magnification mammography is an integral part of calcification analysis and should be obtained on all calcifications requiring further analysis.
Anatomy
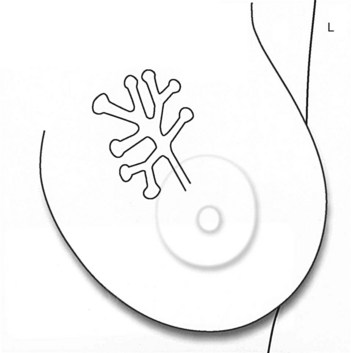
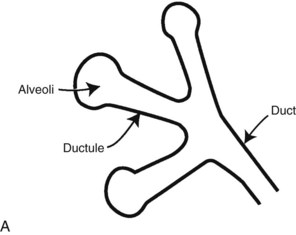
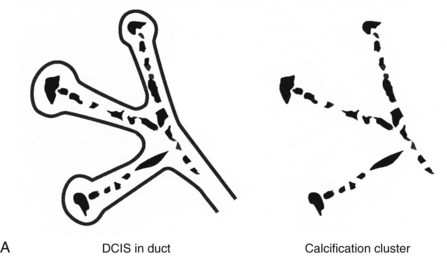
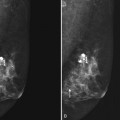
Calcifications form in breast ducts (Fig. 3-1), in lobules (Fig. 3-2A), or within breast tumors. Calcifications forming in the interlobular stroma, in periductal locations, or in blood vessels, fat, or skin are usually benign. Recognizing the location is important because calcifications within the skin, muscle, or nipple are almost invariably benign. Skin calcifications are especially important to recognize because they can easily be mistaken for intraparenchymal calcifications, leading to unnecessary biopsy. Skin calcifications are usually tiny, about the size of the skin pore on the mammogram, and often occur in skin folds where skin touches skin (e.g., axilla, inframammary fold, or in between the breasts). They are classically eggshell-type or contain a lucent center.
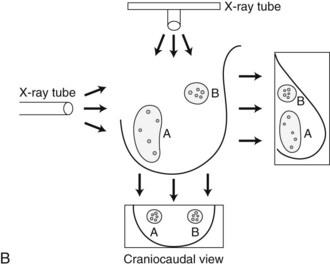
In general, clustered intraparenchymal calcifications are more suspicious for cancer than scattered calcifications. To be considered a suspicious intraparenchymal cluster, the finding must represent a true cluster and not simply be scattered calcifications superimposing on one another. The cluster must be tightly grouped on two orthogonal views to prove it is not a fake cluster (see Fig. 3-2B). To prove that clustered calcifications are truly grouped together, the radiologist looks for similar-appearing clustered calcifications over the same volume of tissue on orthogonal views. If the cluster is tightly packed on orthogonal views, it is a true cluster and should be assessed further. If the cluster is tightly packed on one view and scattered on the other view, it represents a superimposition of calcifications, is a fake cluster, and can be dismissed.
BI-RADS® Lexicon for Calcifications and Individual Calcification Shapes
The American College of Radiology (ACR) Breast Imaging Reporting and Data System (BI-RADS®) lexicon has a good section on description and assessment of calcifications. In the mammography report, radiologists use BI-RADS® terms to describe calcification forms, distribution, location, associated findings, and whether any change has occurred since the previous study. BI-RADS® terms are powerful descriptors that help the clinician understand the seriousness of the finding, such as fine linear branching in cancers. The BI-RADS® terms also help the radiologist classify calcifications into BI-RADS® assessment categories, which prompt patient management (Boxes 3-1 and 3-2). For example, the BI-RADS® term pleomorphic, which is suspicious for cancer, would prompt the radiologist to classify the calcifications into a BI-RADS® category 4, which calls for biopsy to be performed, whereas the term large rodlike, which indicates benign secretory disease, would be classified as a BI-RADS® category 2 and would be dismissed.
Box 3-1
Calcification Report
Size of the cluster or calcific group
Location (right or left breast, quadrant or clock position, centimeters from the nipple)
Overall characteristic of the worst-looking individual calcifications in the group
Change, if previous films are compared
BI-RADS®, Breast Imaging Reporting and Data System.
From American College of Radiology: ACR BI-RADS®—mammography, ed 4, In ACR Breast Imaging and Reporting and Data System, breast imaging atlas, Reston, VA, 2003, American College of Radiology.
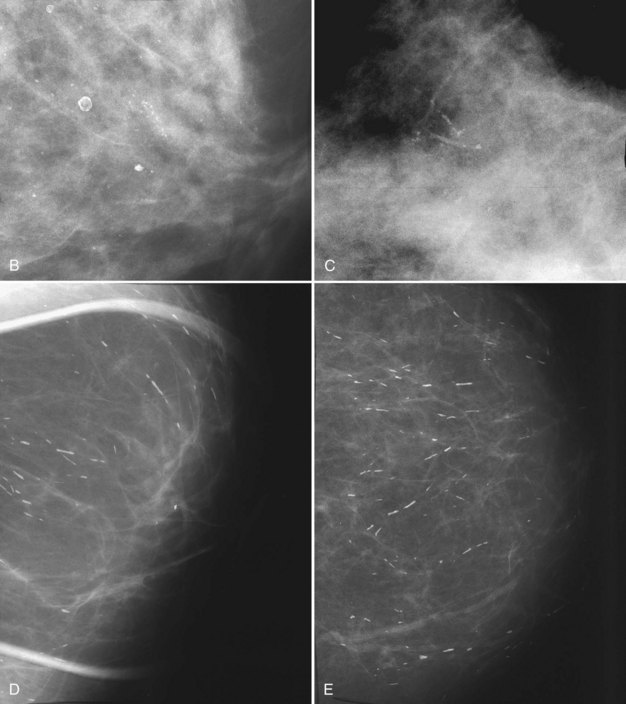
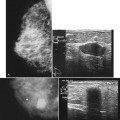
BI-RADS® terms are based on calcification morphology. Knowing the underlying anatomic structure in which calcific particles form helps the radiologist to understand why some calcific shapes have specific morphologies and why they suggest benign or malignant disease. An example of how anatomic structures influence calcification shapes are the round calcifications that form in round benign terminal breast acini or lobules. These benign calcifications take on the round shape of the acini in which they form, and hence, are round, regular in shape, densely calcified, and sharply marginated (Fig. 3-3A to C). The BI-RADS® term for these calcifications is punctate; they are usually benign.
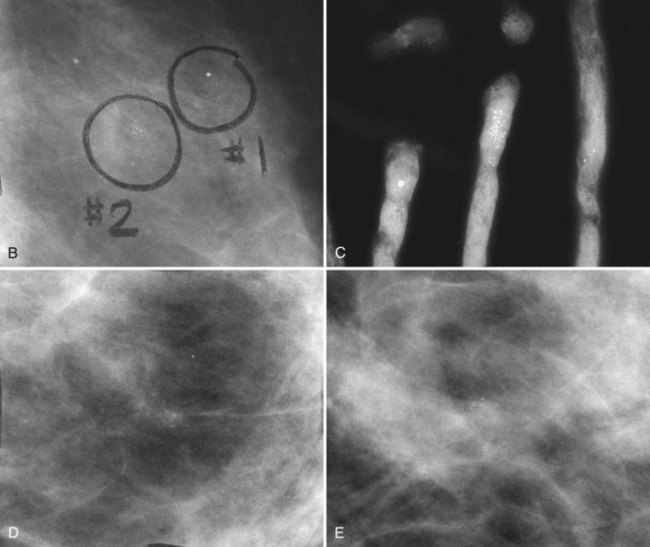
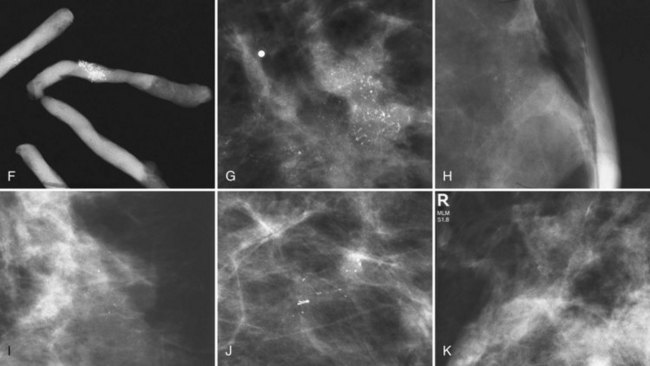
The BI-RADS® term amorphous or indistinct describes indeterminate calcifications that are tiny, roundish, flake-shaped particles that are too small and vague to allow further characterization. Both benign and malignant processes produce this type of calcification (Box 3-3). Benign fibrocystic disease and sclerosing adenosis produce blunt duct extension and ductal dilatation that result in indeterminate amorphous or indistinct calcifications (see Fig. 3-3D to F). However, some amorphous or indistinct calcifications can also form in ductal carcinoma in situ (DCIS) (see Fig. 3-3G to J). This overlap between benign- and malignant-appearing calcifications results in “false-positive” biopsies and accounts for up to 75% of benign biopsy results from procedures prompted by calcifications.
Box 3-3
Terms for Suspicious Calcifications
From American College of Radiology: ACR BI-RADS®—mammography, ed 4, In ACR Breast Imaging and Reporting and Data System, breast imaging atlas, Reston, VA, 2003, American College of Radiology.
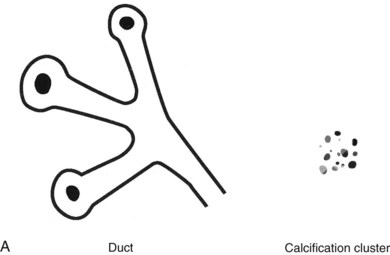
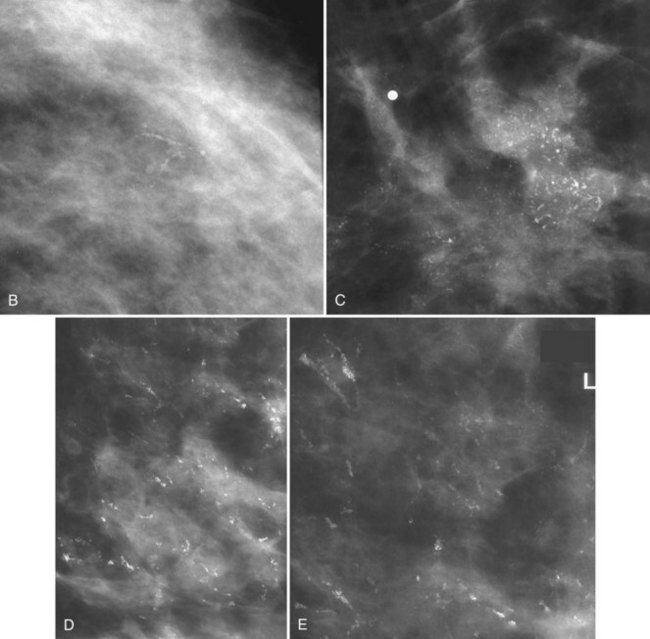
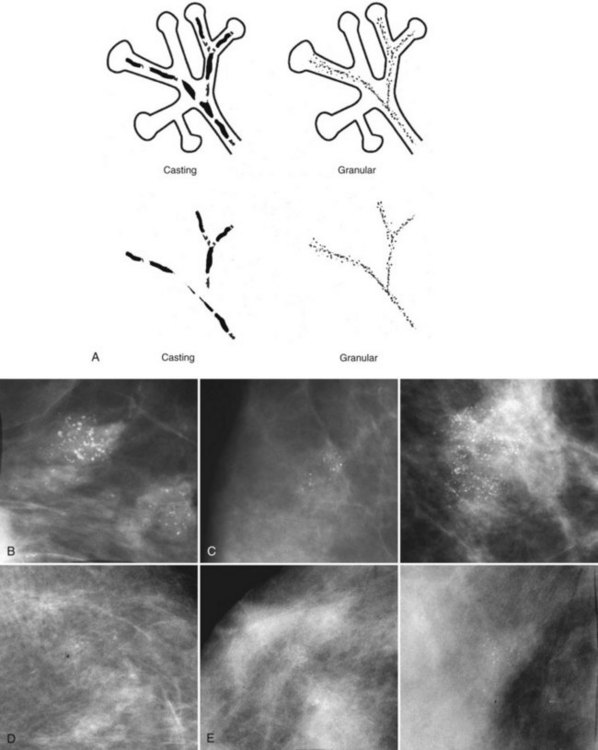
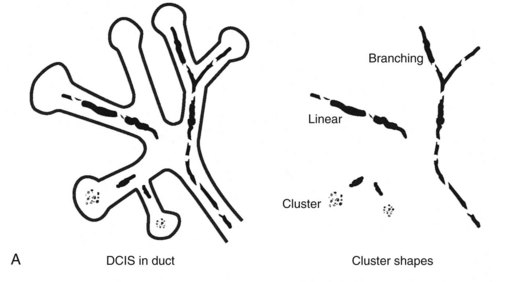
Calcifications that develop in DCIS or invasive ductal cancer grow in breast ducts and have classic appearances (Fig. 3-4A and B). The ACR BI-RADS® term for these calcifications is fine linear or fine linear branching (casting) calcifications. These calcifications have linear forms because DCIS grows in branching ducts and the calcifications form within the DCIS, making tiny irregular casts of the duct. These calcifications may look like little broken needles with pointy ends or may have a “dot-dash” appearance with both round and linear shapes. Calcific casts of tumors growing in duct branches form X-, Y– or Z-shaped calcifications. Radiologists describe these classic calcifications as fine linear, fine linear branching, casting, or pleomorphic in the report to reflect their concern for cancer. This is in contradistinction to benign-appearing round, punctate calcifications (see Fig. 3-4C to E).
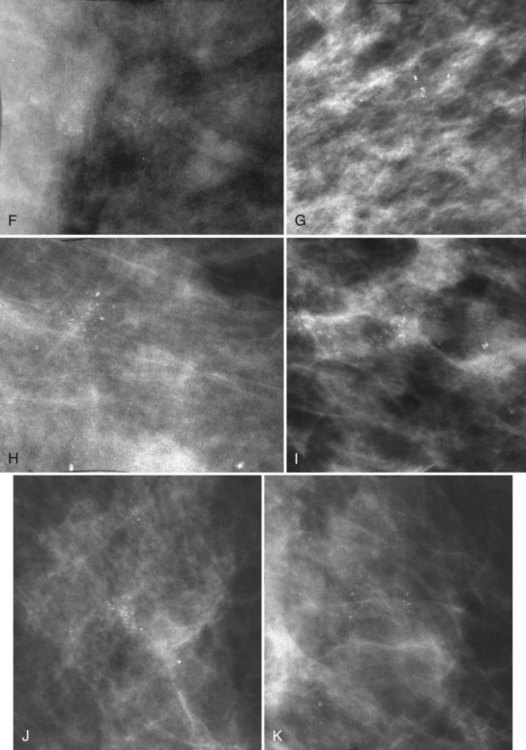
Another suspicious calcification form described by the ACR BI-RADS® lexicon is pleomorphic or heterogeneous (granular). This term reflects very tiny, irregularly shaped calcific particles that look like bizarre broken glass shards forming inside pockets of necrotic tumors, such as the micropapillary or cribriform forms of DCIS (Fig. 3-5). The individual calcifications are roughly round in shape but have irregular borders, are faint, smaller than 0.5 mm, and vary in size and density. A cluster containing granular calcifications may not exhibit casting or linear forms but should still be considered suspicious even in their absence. Occasionally, granular calcifications form in a duct and look like sand stuffed in a plastic straw. Unfortunately, benign disease occasionally mimics DCIS and also forms granular calcifications (see Fig. 3-5B to G).
Calcification Group Shape or Distribution within the Breasts
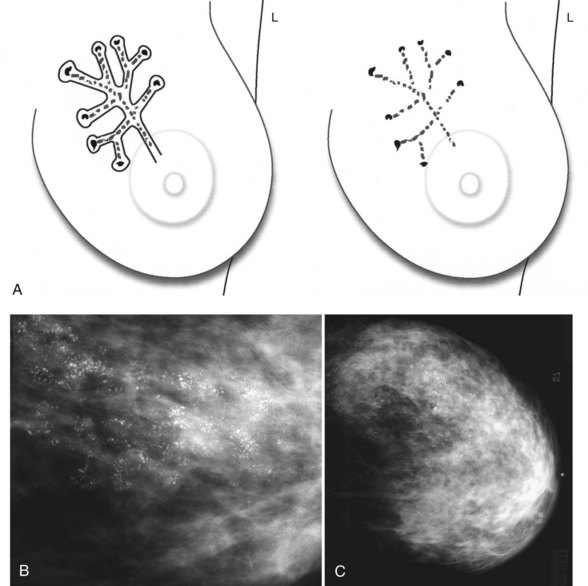
Calcification distributions that may represent cancer in the ACR BI-RADS® lexicon are those described as clustered or grouped, linear, branching (calcifications in a line that may show branching) (Fig. 3-6), and segmental (Fig. 3-7). Cancer forms in diseased ducts within a breast lobe, or the so-called “sick lobe,” as described by Tot and Gere.
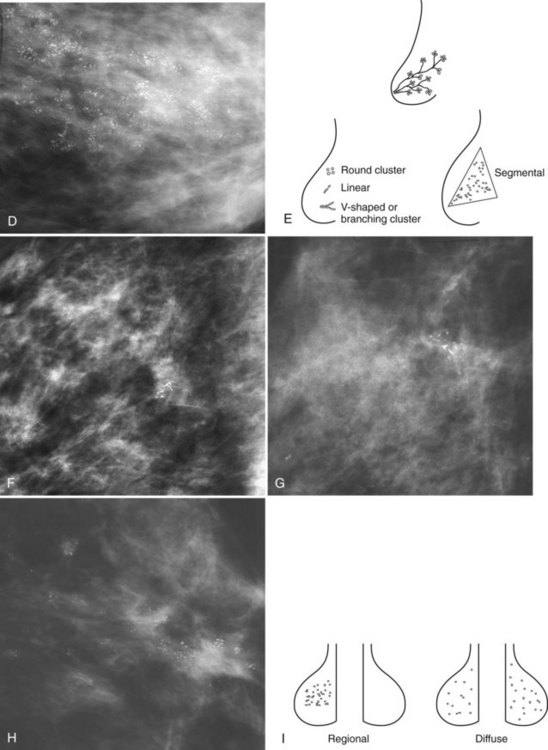
Isolated calcification clusters suggest an isolated disease process in a small volume of tissue. This may represent DCIS, invasive cancer, fibrocystic change, papilloma, or sclerosing adenosis. For clusters, one analyzes both the individual calcification forms and the overall cluster shape, which may be a clue to whether the cluster is benign or malignant. Lanyi suggested that the overall cluster shape is especially suspicious if it has a swallowtail, or V, shape, because it suggests cancer in tumor-packed branching ducts. On the other hand, calcifications forming in round clusters may be forming in acini and be benign, especially if the individual calcifications within the cluster are also benign-appearing (see Fig. 3-7E).
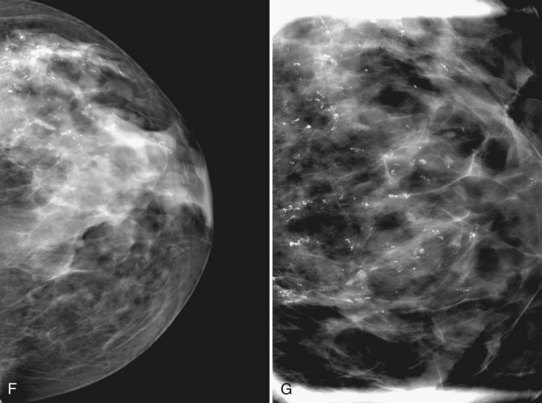
The ACR BI-RADS® terms linear branching and segmental describe suspicious findings because they suggest a process within a duct and its branches. The term linear describes calcifications in a line and can represent tumor in a duct or a focal benign process. A segmental distribution is suspicious for cancer because it suggests a process within a branch and its ducts. Segmental calcifications cover slightly less than a quadrant and form in a triangle with its apex pointing at the nipple (see Fig. 3-7F to H).
BI-RADS® terms suggesting benign calcification distributions include regional and diffuse/scattered (see Fig. 3-7I). This pattern suggests innumerable scattered and occasionally clustered calcifications widely dispersed over the breasts and often reflects benign processes, which are also often spread widely throughout both breasts. Calcifications widely distributed in both breasts are usually due to fibrocystic change. Regional calcifications extend over more than one ductal distribution (Box 3-4). Diffuse extensive benign-appearing calcifications in both breasts rarely represent breast cancer. The decision to biopsy calcifications is based on their distribution within the breasts, the worst features of the individual calcification clusters, change over time, the clinical scenario, and common sense.
Benign Calcifications
The specific benign calcifying entities covered in the following subsections require no further workup and should prompt no further action (Box 3-5).
Box 3-5 Typically Benign Calcifications
Artifacts (deodorant, hair, fingerprints)
Skin artifacts: antiperspirant, material in moles
Eggshell-type calcifications, radiolucent centers
Fat necrosis (postbiopsy, post-trauma)
Vascular calcifications (tram-track appearance)
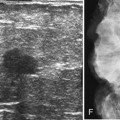
Fibroadenoma (mass with round, coarse peripheral calcifications)
Plasma cell mastitis or secretory disease (needle-like or sausage-shaped calcifications pointing toward the nipple; found in middle-aged women; benign entity, usually asymptomatic)
Milk of calcium (linear on the mediolateral view, smudgy on the craniocaudal view)
Dystrophic calcifications (be alert for such calcifications in women after biopsy for cancer)
Suture calcifications (cat gut, postradiation)
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree