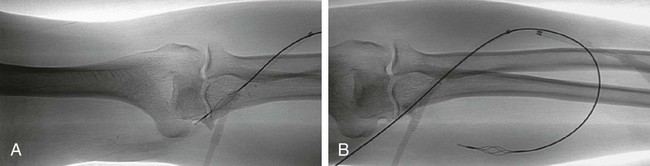
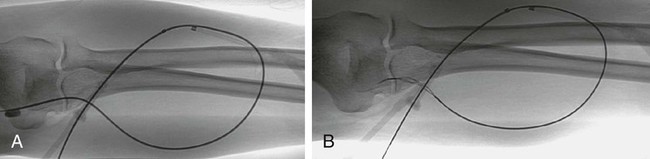
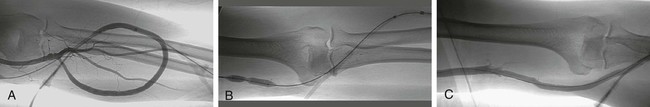
Approximately 20 million individuals in the United States have chronic kidney disease, and just as many are at increased risk for development of such disease.1 In 2004 in the United States alone, 320,000 patients required dialysis (peritoneal dialysis or hemodialysis) for end-stage renal disease.2 Because many of these patients require hemodialysis, a reliable long-term dialysis access is their lifeline. Although an increase in fistulas is the goal of the National Kidney Foundation Kidney Disease Outcomes Quality Initiative (NKF KDOQI) and the Fistula First Initiative, when a suitable vein is not available, use of an artificial graft is the only option. Regardless of whether it is a graft or fistula, thrombosis is inevitable in time, although evidence suggests that the rate may be lower with fistulas. Clotted access may be managed by open surgery (increasingly rarely) or by various endovascular techniques (including mechanical thrombectomy, pharmacologic thrombolysis, and combination techniques, all with associated angioplasty of the culprit lesions). In the past, surgical thrombectomy was the norm. However, in the last decade, endovascular management has replaced open surgery as the primary method of treatment of a thrombosed dialysis access.3 In this chapter only the endovascular management of clotted hemodialysis access grafts is discussed. • Hemodialysis access graft infection or overlying cellulitis. Unlike fistulas, in which thrombophlebitis is common, redness, warmth, and swelling at the access site should be considered infection until proven otherwise. Treatment should begin with intravenous antibiotics. Because the infected material in the graft cannot be cleared with antibiotic therapy alone, subsequent surgical intervention is needed, even if the overlying tissue appears to have healed. • Severe allergy to contrast material. Because of the current U.S. Food and Drug Administration (FDA) warning on the use of gadolinium in patients with end-stage renal disease (concern over a possible relationship to nephrogenic fibrosing dermopathy), it should not be used until more evidence is gathered and the issue is resolved.4,5 Pretreatment with prednisone, 40 mg orally 12 and 2 hours before intervention, is usually sufficient for most allergic reactions; some add H1 and possibly H2 blockers, but there is little evidence for this.6 • Abnormal but correctable coagulation parameters • Ischemia of the ipsilateral extremity, because restoring flow will worsen the ischemia by exacerbating the steal phenomenon. • History of a significant right-to-left shunt because of the risk for paradoxical emboli with rare reported strokes from percutaneous declotting.7,8 However, successful and complication-free thrombectomy in patients with a known patent foramen ovale, even with right-to-left shunting, has been reported.9 • Significantly reduced pulmonary reserve (pulmonary hypertension, severe lung disease, right heart failure) because of an inability to tolerate the small pulmonary emboli resulting from virtually any form of declotting. Fatal pulmonary emboli have been reported in this population.10,11 • Access needle/set (angiocatheter, 19-gauge needle, or a micropuncture set) • Two infusion catheters or a mechanical percutaneous thrombectomy device (e.g., Arrow-Trerotola PTD) • Appropriately sized percutaneous transluminal angioplasty (PTA) balloons • An inflation device or a 1-mL polycarbonate syringe (preferred), 10-mL syringe, and a flow switch The importance of clinical evaluation (history, physical examination, and laboratory tests) in the management of a clotted hemodialysis access graft should not be underestimated. It can help the operator prevent futile efforts and avoid complications. The preoperative workup includes a directed history of the dialysis access and previous interventions and a subsequent physical examination.3,12 Determining when the access was created, the type of access in place, and when the graft thrombosis occurred is important. If more than two previous interventions have been performed in less than 1 month, the access may require surgical management. It is critical to determine whether there are signs of local (hemodialysis access graft site) infection (warmth, erythema, tenderness) or systemic infection (fever, chills). If along with physical examination it is determined that graft infection is present, access intervention is absolutely contraindicated.3,12–14 The history should also focus on possible contraindications such as right-to-left shunts and severe pulmonary disease. Physical examination is just as important. The flow circuit in the extremity should be assessed by palpating and documenting the ipsilateral radial, ulnar, and brachial pulses. Blood supply to the distal part of the extremity must be assessed as well (capillary refill and warmth of the hand/arm). To assess for central venous stenosis or occlusion, chest wall collaterals should be noted with care. If present, it should be determined whether arm swelling was present when the graft was functioning, because swelling may dictate treatment of a central lesion discovered after restoring graft patency.15 Cardiac and pulmonary examinations are also important to assess for signs of significant fluid overload.3,12–14 In addition, each graft should be evaluated by physical examination before every intervention. The site should be assessed for warmth, tenderness, and erythema, because these symptoms may indicate graft infection or cellulitis. To choose optimal puncture sites for intervention, one must determine the type of access (straight or loop), direction of inflow and outflow, presence of aneurysmal graft degeneration, and type of anastomosis.3,12 A few cases of postprocedure septic shock have been reported, and colonization of clotted grafts is very common.16 Therefore (though not evidence based), most interventionalists give preprocedural prophylactic antibiotics (cefazolin [Ancef], 1 g intravenously). To prevent rethrombosis during the procedure, as well as to blunt any pulmonary vasospastic or bronchospastic response to small pulmonary emboli occurring during declotting, 3000 to 5000 units of heparin should be administered before intervention. Both open surgical and endovascular approaches can be used to declot a thrombosed hemodialysis access graft. Only endovascular approaches (mechanical thrombolysis, pharmacologic thrombolysis, and combination approaches) are addressed in this chapter. Thrombolysis of a hemodialysis access graft has two major steps: clot removal (Fig. 116-1) (including the arterial plug [Fig. 116-2]) and treatment of the underlying anatomic lesion (Fig. 116-3). In the absence of infection, there are usually underlying causes (e.g., stenoses at the venous or arterial anastomoses, outflow veins, or rarely, arteries proximal to the arterial anastomosis) that have to be addressed with PTA. Some common techniques, but not all techniques, are described in this chapter; it is not meant to be a comprehensive review. Many excellent review articles are listed in the Suggested Readings at the end of this chapter. Access to the graft depends on the configuration of the graft but is the same regardless of declotting technique used. Although there are many individual variations and preferences, the basic crossed-catheter technique has stood the test of time. It should be noted that despite the name, the access sheaths should not actually cross, or flow will be partially obstructed and efforts at declotting will be impeded. In a loop graft, the first puncture is made near the apex, directed toward the venous anastomosis, and the second puncture is made approximately 1 to 2 cm from the venous anastomosis, directed toward the arterial anastomosis. Similarly, for straight grafts, the first puncture in made near the arterial anastomosis and the second near the venous anastomosis, with each directed toward the other anastomosis. The apex technique17 deserves mention only because we believe this technique is not suitable for declotting because of the need to repeatedly access both ends of the graft at different times during the procedure.3,12 Mechanical devices for clot fragmentation and removal include those that fragment the clot and those that fragment the clot and aspirate the fragments. Commonly used devices are the Arrow-Trerotola PTD (Teleflex Inc., Limerick, Penn.), AngioJet (Possis Medical Inc., Minneapolis, Minn.), and Helix Clot Buster Thrombectomy devices (ev3/Covidien, Plymouth, Minn.). Although the AngioJet fragmentation/aspiration device is commonly used, published evidence from a randomized trial does not support its use in this setting.18
Management of Clotted Hemodialysis Access Grafts
Contraindications
Absolute
Relative
Equipment
Technique
Anatomy and Approach
Patient Evaluation and Preparation
Technical Aspects
Graft Access
Clot Removal
Mechanical Methods
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree