Abstract
Spinal cord arteries and veins represent tiny, millimeter-to-submillimeter blood vessels for which in vivo depiction has only recently become possible using noninvasive imaging techniques. A brief overview of the complex and clinically relevant vascular anatomy of the spinal cord is provided, including the supplying and draining trajectories. A magnetic resonance angiography protocol is outlined that enables the visualization and differentiation of the spinal cord arteries and veins. Finally, it is discussed how the complex and tortuous venous anatomy may confound the interpretation of blood oxygen level–dependent (BOLD) activation responses obtained from the spinal cord.
Keywords
Adamkiewicz artery, Differentiating spinal cord arteries and veins, MR angiography, MR venography, Vascular spinal cord anatomy
4.3.1
Rationale
Spinal cord blood vessels are millimeter- to submillimeter-sized blood vessels that are hard to visualize in the human body. In vivo imaging of spinal cord arteries and veins was until recently only possible using invasive catheter-based angiography, which involves a radiation burden and needs to be performed by experienced specialists. Noninvasive imaging of spinal cord blood vessels had only recently become available in a robust way using computed tomography (CT) and magnetic resonance (MR) angiography. It was mainly from clinical research in aortic aneurysm surgery that the request came to develop noninvasive visualization of spinal cord arteries to avoid the risk of paraplegia induced by damaging the spinal cord blood supply. Traditional MR angiography techniques at that time (prior to 2000) were not able to depict normal intradural arteries and were restricted to the visualization of pathologically dilated arteries and veins. Today, techniques have strongly improved vessel-to-background contrast because they use relatively fast contrast-enhanced imaging techniques that are now able to depict and differentiate normal spinal cord arteries from veins.
In this chapter, the relevant vascular anatomy of the spinal cord is first briefly outlined. Then, a technique of fast contrast-enhanced MR angiography is described to image normal spinal arteries and veins. Finally, the possible confounding effect of the variable and unpredictable course of spinal cord veins in relation to the blood oxygen level–dependent (BOLD) effect of spinal cord functional MR imaging (fMRI) will be addressed.
4.3.2
Vascular Anatomy
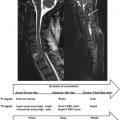
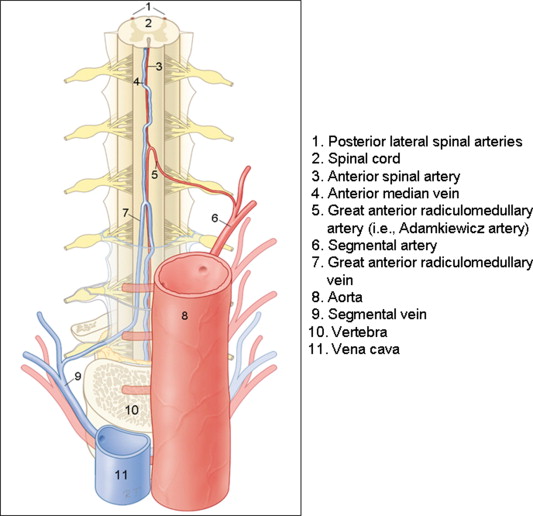
To understand the thoracolumbar spinal cord vasculature, it is useful to divide the spinal cord vasculature into the supplying arterial part (inlet) and draining venous part (outlet) (see Figure 4.3.1 ). In this section, a general outline of the spinal cord vasculature will be given as can be imaged with human invasive or noninvasive angiography techniques. In this context, it is important to notice that interindividual anatomic variation is the rule rather than the exception (also see Chapter 4.1 regarding the intersubject variability of BOLD responses).
- •
Anterior cord surface: one spinal artery and one median vein that runs in the anterior median sulcus
- •
Posterior cord surface: one or two very small posterolateral spinal arteries and one “median” vein. The course of the posterior median vein is more tortuous and is not strictly median as compared to the anterior median vein
4.3.2.1
Spinal Cord Arteries
The supplying pathway starts in the aorta and continues with a segmental artery that branches into a number of trunks. Its posterior trunk then forms the radicular artery, which supplies the nerve root. At specific, interindividually different levels, the radicular artery continues its route toward the midline of the anterior cord surface (i.e., the anterior radiculomedullary artery (ARA)) and there connects to the anterior spinal artery (ASA) with an ascending and descending branch. The downward branch has a hairpin-shaped configuration. The largest ARA is known as the Adamkiewicz artery (AKA; diameter 0.5–1.0 mm). Due to the opposing blood flow directions, watershed regions may manifest; these are regions between two entering ARAs where there will be very little or even no flow in either direction. The ASA varies strongly in diameter (0.2–0.8 mm) along its trajectory (the midthoracic part is thinnest, and the lumbar part is thickest) and distributes blood to the anterior two-thirds of the cord tissue by central and pial branches. On the posterior cord surface, the spinal arteries are usually paired and located posteriolaterally. They receive their supply also from posterior branches of the segmental arteries (i.e., the posterior radiculomedullary arteries). The diameter of these arteries is less than 0.5 mm and can be depicted only occasionally using catheter angiography.
4.3.2.2
Spinal Cord Veins
On the anterior cord surface, there is one collecting vein located medially, the anterior median vein (diameter 0.4–1.5 mm) that runs next to the ASA. Posteriorly, there is also one collecting vein (diameter ≤ 2 mm). Its course, however, is not strictly medial and is often more tortuous ( Figures 4.3.1 and 4.3.5 ). Both of these veins drain the cord by radiculomedullary veins, which have a spatial configuration comparable to that of the radiculomedullary arteries. Often, the radiculomedullary veins have a longer intradural trajectory than the radiculomedullary arteries.
- •
Intraarterial catheter-based (X-ray) angiography (arteries and veins)
- •
Contrast-enhanced MR angiography (arteries and veins)
- •
Susceptibility-weighted imaging (veins)
- •
CT angiography (arteries and veins)
4.3.3
MR Angiography
Currently, the best minimally invasive approach to depict the (normal) spinal cord vasculature using MRI is to employ contrast-enhanced MR angiography. Two contrast-enhanced three-dimensional (3D) MR angiography approaches appear successful for the localization of the AKA. The difference between these two approaches is that one technique uses a strong (i.e., temporarily high-concentration) bolus (gadolinium), of which mainly the first passage is captured during a period of 20–60 s. Table 4.3.1 lists the MR angiography protocol advised by Nijenhuis and coworkers. For the second approach, a long and slow contrast injection (≤1 ml/s) is used with lengthy acquisition times of 4–6 min (i.e., a decrease in pixel size and slice thickness). The main advantage of the strong-bolus technique is that it may provide separation of intradural arteries and veins, whereas the slow-bolus technique does not allow this temporal differentiation. However, the slow-bolus technique enables the acquisition of images with higher spatial resolution and thereby anatomical differentiation. Both techniques rely on intravenous contrast administration and spoiled gradient echo pulse sequences with ultrashort echo times and repetition times.