Fig. 6.1
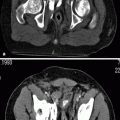
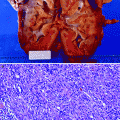
Renal amyloidosis. Most of the glomeruli in this renal biopsy exhibited extensive mesangial deposits of pink-staining amyloid (Case courtesy of Gretta Jacobs, M.D.)
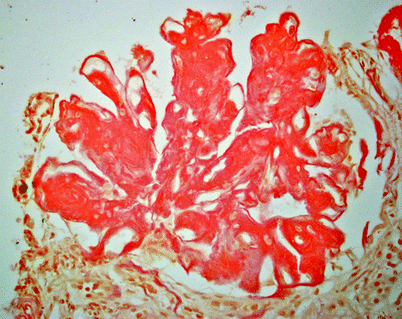
Fig. 6.2
Renal amyloidosis. Same case as Fig. 6.1. The reddish-orange material is amyloid that is highlighted by Congo red histochemical stain
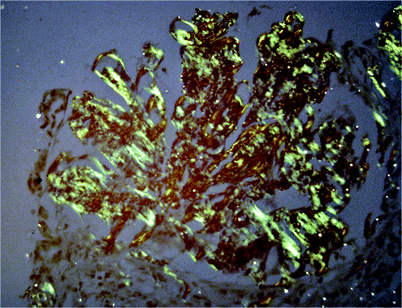
Fig. 6.3
Renal amyloidosis. Under polarized light, amyloid is apple green. Because of the unique beta-pleated structure of amyloid, not all the amyloid present in the section is visualized by polarized light
In extraglomerular amyloidosis, amyloid accumulates in the interstitium and renal vasculature.
Vascular amyloidosis is rare and results in hypertension and occlusive vascular disease. Microscopically, amyloid is highlighted by Congo red histochemical stain and exhibits apple-green birefringence when polarized (Figs. 6.2 and 6.3).
Differential Diagnosis
Calcified focal lesions may mimic calculus.
Focal parenchymal masses must be differentiated from neoplasms.
Pearls and Pitfalls
Amyloid deposition may mimic focal renal parenchymal or pelvic masses.
Pelvic filling defects due to focal amyloid or blood clots of spontaneous hemorrhage.
The most common radiological finding in renal amyloidosis is normal-sized kidneys.
Renal vein thrombosis can occur with amyloidosis.
Diabetic Nephropathy
General Information
As the main cause of end-stage renal disease, diabetic nephropathy develops in up to 35 % of all type 1 or type 2 diabetic patients after 15–20 years of diabetes.
Nephropathy has a traditional cumulative incidence of 30–40 % in type 1 diabetics and 25 % in type 2 diabetics at more than 20 years after diagnosis.
In the United Kingdom, the overall prevalence of nephropathy in general diabetic clinics is 4–8 %. Meanwhile, it must be remembered that up to 20 % of diabetics can develop renal failure due to nondiabetic kidney disease, with classical findings of other chronic renal diseases like decrease in renal size and parenchymal thickness.
Imaging
Ultrasonography
Early stage
Normal or enlarged kidneys in early stage. Nephromegaly in early stage is typical and may remain until overt nephropathy. This may be decisive, and renal size cannot be used as a reliable criterion to decide the irreversibility of nephropathology unlike other renal pathologies.
Maintains renal shape, parenchymal thickness, and architecture due to uniform involvement of all parenchymal components.
Normal Doppler indices in intrarenal arteries.
Late stage
Normal or small kidneys
Parenchymal hyperechogenicity with gradual loss of corticomedullary differentiation (Fig. 6.4)
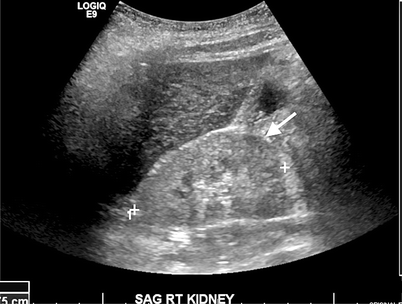
Fig. 6.4
Diabetic nephropathy. Gray-scale ultrasound demonstrates increased echogenicity of renal parenchyma and absence of corticomedullary differentiation (arrow)
Increased RI and PI values in Doppler scanning with correlation to serum creatinine concentration
Angiography
Increased incidence of renal artery stenosis
Differential Diagnosis
Early phase may show similar imaging findings as small-vessel vasculitides.
Pearls and Pitfalls
Nephromegaly in early stage is typical and may remain until overt nephropathy.
Renal size cannot be used as a reliable criterion to decide the irreversibility of nephropathology unlike other renal pathologies.
Up to 20 % of diabetics can develop renal failure due to nondiabetic kidney disease, with classical findings of other chronic renal diseases like decrease in renal size and parenchymal thickness.
Small-Vessel Vasculitis in Kidney
General Information
Large vessel vasculitides (Takayasu arteritis and giant cell arteritis) cause renovascular disease; the medium-sized vessel vasculitides (polyarteritis nodosa, Kawasaki’s disease) result in renal infarcts and hemorrhage.
Vasculitides of more peripheral vessels, namely, capillaries, venules, and arterioles, and in some instances, medium-sized arteries, may clinically manifest as glomerulonephritis.
Small-vessel vasculitides comprise a variety of diseases. Wegener’s granulomatosis, microscopic polyangiitis, Churg-Strauss syndrome, and drug-induced ANCA-associated vasculitis are associated with positive antineutrophil cytoplasmic antibodies (ANCA). Cryoglobulinemic vasculitis, Henoch-Schönlein purpura, lupus vasculitis, rheumatoid vasculitis, and drug-induced immune-complex vasculitis are associated with immune complexes. Other vasculitides include paraneoplastic or inflammatory bowel disease vasculitis.
Wegener’s granulomatosis, microscopic polyangiitis, and Churg-Strauss syndrome usually start after the fifth decade and have an approximate annual incidence of 1–2 cases per 100,000 population. On the other hand, 60 % of adult patients suffering lupus may develop renal abnormalities late in the course of the disease, which is especially prevalent in Afro-American females (approximately, 1/400).
Imaging
Ultrasonography
Normal, increased or decreased (late in the course) renal size
Increased cortical echogenicity, decreased corticomedullary differentiation
Cortical hypoechoic areas with cortical distortion (due to edema)
Normal or increased RI and PI values on Doppler imaging
Computed Tomography
Affected kidney may show reduced nephrogram after contrast administration.
Rarely ill-defined mass-like lesions may be seen.
Magnetic Resonance Imaging
Infiltrative lesions may be isointense on T1 and show heterogeneous appearance on T2 sequence.
Lesions may be seen as pseudotumors.
Pathology
Inflammation of the vessels, or vasculitis, is encountered in a variety of clinical settings.
The most common histologic pattern is crescentic glomerulonephritis. In this pattern, there is necrosis of the glomerular capillary loop wall and subsequent scar formation (fibrosis), the size of which depends on the severity of the initial event. Crescents can form from other causes; direct immunofluorescence is needed to classify the underlying process.
Most detectable immune reactants are antineutrophil cytoplasmic antibodies. Necrotizing vasculitis is rare; in this pattern, there is necrosis with fibrin deposition, neutrophils, and karyorrhexis (Fig. 6.5 and 6.6). This pattern of vasculitis is common in entities such as classic polyarteritis nodosa and Wegener’s granulomatosis. Serologic studies and clinical correlation are needed to classify the underlying process.
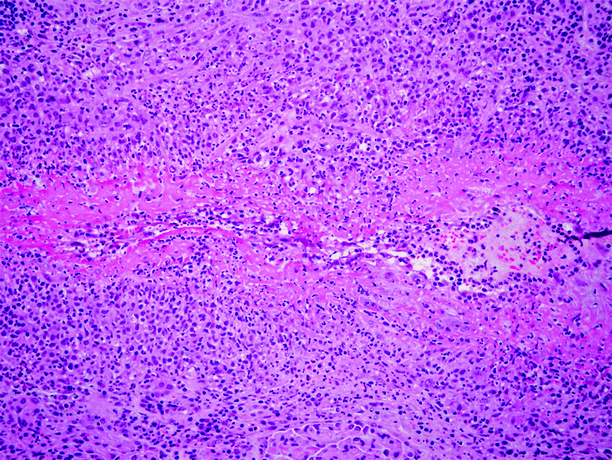
Fig. 6.5
Necrotizing vasculitis in kidney. This male patient had polyarteritis nodosa clinically. In addition to the renal involvement illustrated in this image, he also experienced loss of vision in both eyes. This small renal artery is diffusely infiltrated by inflammatory cells, including abundant neutrophils, and is undergoing fibrinoid necrosis (Case courtesy of Gretta Jacobs, M.D.)
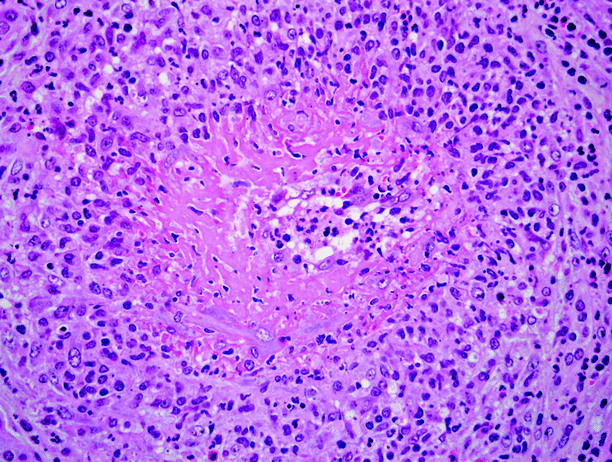
Fig. 6.6
Necrotizing vasculitis in kidney. Same case as shown in Fig. 6.5. Higher power view of a small renal artery undergoing fibrinoid necrosis
Differential Diagnosis
No specific imaging finding in differentiation from other medical renal disease
Pearls and Pitfalls
Medium-sized vessel vasculitides (polyarteritis nodosa, Kawasaki’s disease) result in renal infarcts and hemorrhage.
Thrombotic Microangiopathies
General Information
Thrombotic microangiopathies comprise a group of disorders characterized by an acute syndrome of microangiopathic hemolytic anemia, thrombocytopenia, and manifestations of organ injuries resulting from platelet thrombosis in microcirculation. Diarrhea may accompany other symptoms in some cases.
Although the cause may be an infection, medication, or vaccination, a systemic disease is the most common. Among various systemic diseases, hemolytic-uremic syndrome (HUS), thrombotic thrombocytopenic purpura (TTP), the antiphospholipid syndrome, and scleroderma are the mostly diagnosed ones as the underlying factor in many cases.
HUS affects predominantly children, especially the ones under 3 years of age, and results in lesions causing acute renal failure. Neurologic involvement is absent or minimal. On the contrary, TTP mostly is diagnosed in adults and is characterized by severe neurologic and variable renal involvement in many cases. These two entities may coexist as well, which is called HUS/TTP. As the most common cause of acute renal failure in children, the annual incidence of HUS among pediatric population is quite low, which is about 1–3 cases per 100 000 per year in different countries.
Imaging
Ultrasonography
Normal or slightly increased renal size
Increased cortical echogenicity
Severely increased Doppler index values (RI, up to 0.90–1.00) found in the intrarenal arteries in the oliguric/anuric phase. Normalization of the Doppler indices is early sign of the recovery of renal function.
Decreased renal parenchymal perfusion on color/power Doppler imaging.
Nuclear Scintigraphy
Extreme reduction of renal perfusion in 99mTc-DTPA images.
99mTc-MAG3 uptake is a good prognostic sign.
Pathology
Thrombotic microangiopathy is encountered in a variety of clinical settings, the commonest of which are hemolytic-uremic syndrome and thrombotic thrombocytopenic purpura, but the use of the term has been expanded to include such entities as malignant hypertension, disseminated intravascular coagulation, scleroderma renal crisis, postpartum acute renal failure, irradiation, and other entities, all of which are histologically similar.
In the acute phase, notable features include glomerular capillary loop and arterial thrombus formation, and in the chronic phase, duplication of the basement membrane and mesangiolysis (Fig. 6.7).
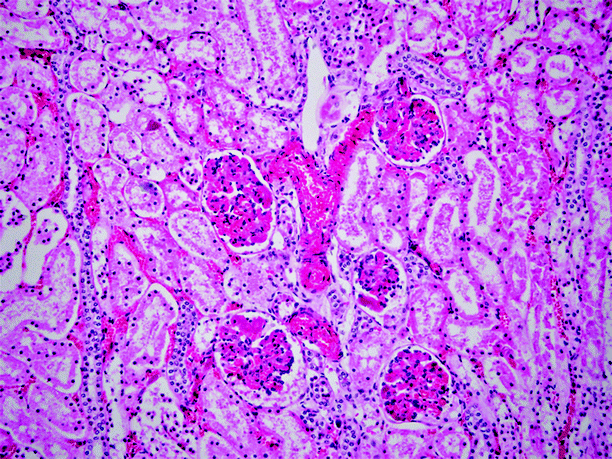
Fig. 6.7
Disseminated intravascular coagulation. This is one of the many entities classified as examples of thrombotic microangiopathy. This section is from a kidney of a child who died of sepsis, having shown clinical features of disseminated intravascular coagulation premortem. Thrombus formation was widely present in arterioles and glomeruli, as exemplified in this image (Case courtesy of Beverly Dahms, M.D.)
Differential Diagnosis
Increased renal size may mimic diabetic nephropathy, and Doppler indices are not increased in early diabetic nephropathy.
Differentiation of HUS and acute tubular necrosis not be possible using imaging.
Pearls and Pitfalls
HUS affects predominantly children, whereas TTP affects adults.
Normalization of the Doppler indices is early sign of recovery of renal function.
99mTc-MAG3 uptake is a good prognostic sign.
Tubulointerstitial Disease
General Information
Primary interstitial nephropathies consist of diseases which cause interstitial inflammation and renal tubular damage.
Interstitial nephritis has been classified into acute and chronic forms. The acute form is generally characterized with rapid deterioration in renal function. The glomerular and vascular components of the kidney are typically spared.
Current scientific data supports the hypothesis that acute interstitial nephritis (AIN) is an immunologically induced hypersensitivity reaction to an antigen associated with a drug or infection. Although it is identified in only about 2–3 % of all renal biopsies, this type of interstitial nephritis is estimated to be responsible in about 10–15 % of cases suffering acute renal failure. Currently, drugs like antimicrobial agents and nonsteroidal anti-inflammatory drugs are the main triggering factors of the pathology, contrary to previous era when antibiotics were not available and infections were responsible in most of the cases.
Chronic interstitial nephritis (CIN) causes a more gradual deterioration in renal function due to progressive scarring of the tubulointerstitium. Among primary causes of CIN, analgesic nephropathy, vesicoureteral reflux, chronic obstruction, sickle cell nephropathy, radiation, and sarcoidosis are included. Secondarily, glomerulonephritis, diabetes, hypertension, polycystic kidneys, aging, and all progressive renal diseases that cause tubulointerstitial scarring may lead to CIN.
The characteristic tubulointerstitial fibrosis and atrophy, associated with interstitial mononuclear cell infiltration, are followed by the involvement of glomerular and vascular structures in late phase. About 15 % of end-stage renal disease cases are accepted to be the result of CIN.
The imaging features of the tubulointerstitial nephritis, either in acute or chronic form, are nonspecific in nature, and a definite diagnosis is usually reached only by means of renal biopsy.
Acute Interstitial Nephritis
Imaging
Ultrasonography
Normal or increased renal size (due to interstitial edema)
Often increased cortical echogenicity
Decreased renal perfusion on color and power Doppler ultrasound
Increased resistive index values in intrarenal arterial branches
Computed Tomography
Kidneys are enlarged in the acute phase.
Delayed postcontrast CT reveals corticomedullary contrast enhancement due to vascular and tubular stasis.
Nuclear Scintigraphy
Increased uptake of gallium (67Ga) (nonspecific; increased also in pyelonephritis, glomerular diseases, and malignancy)
Chronic Interstitial Nephritis
Imaging
Plain Film Radiography
Bilateral and asymmetrically diminished renal size
Papillary calcifications
Intravenous Pyelography
Early-stage papillary necrosis (“claw” sign)
Complete papillary necrosis (“ring” sign)
Partial papillary necrosis with central cavitation of calyces
Filling defects caused by sloughed papillae
Ultrasonography
Bilateral atrophic kidneys
Irregular renal contours
Increased parenchymal echogenicity mainly in medulla and loss of corticomedullary differentiation
Papillary calcifications
Calyceal cavitations
Intra-calyceal free material due to sloughed papillae
Renal cysts
Diminished renal perfusion in Doppler scanning
Computed Tomography
Papillary calcifications in non-contrast scanning
Shrunken kidneys
Changes in calyceal contour associated with papillary necrosis
Renal cysts
Differential Diagnosis
In case of bilateral small kidneys, prerenal (e.g., generalized atherosclerosis, atheroembolism, renal artery stenosis, radiation nephritis), intrarenal (hereditary nephropathies, amyloidosis in late phase, chronic glomerulonephritis, post-inflammatory atrophy), or postrenal (reflux nephropathy, post-obstructive atrophy) affecting both kidneys should be considered in the differential diagnosis of tubulointerstitial disease. It should also include papillary necrosis, diabetes nephropathy, and sickle cell nephropathy.
Papillary necrosis due to analgesic nephropathy is almost always a bilateral disease with wavy contours of kidneys contrary to those in reflux nephropathy with blunted calyces and deep cortical scars.
Acute Renal Failure
General Information
Acute renal failure (ARF) is defined as a syndrome with acute decrease in glomerular filtration rate. It is often reversible and may occur through three different mechanisms: (a) as an adaptive process in case of reduced renal perfusion in the presence of intact nephrons, (b) when the nephrons lose their structural and functional integrity as a result of cytotoxic insult, and (c) as a consequence of obstructed drainage of urine. Thus, according to the causative mechanism, ARF cases can be classified in one of three different groups, respectively: prerenal, intrinsic (renal), and postrenal.
ARF complicates about 5 % of all hospital admissions and 30 % of cases referred to intensive care units.
Most of the cases of ARF are related to prerenal and intrinsic renal causes. More than 90 % of intrinsic renal ARF cases are attributed to ischemia and/or nephrotoxic insults with associated acute tubular necrosis.
The main indication for imaging in ARF is to exclude possible obstruction like urinary stone, retroperitoneal fibrosis, or a tumor affecting the excretion of both kidneys or a solitary kidney.
The evaluation of renal size, parenchymal structure and related arterial and venous flow may supply valuable data on the nephrologic status of the patient.
Imaging
Plain Film Radiography
A kidney-ureter-bladder (KUB) radiography may demonstrate vascular calcifications of aorta and renal arteries.
Furthermore, urinary stones in kidney, bladder, and ureters can be delineated. The relatively low sensitivity of ultrasound in detecting ureteral stones can be compensated using KUB radiograms.
Intravenous Pyelography
This modality is not suggested in the diagnostic work-up of ARF due to the nephrotoxic effects of intravenous iodized contrast material.
Antegrade Pyelography
The exact level of obstruction can be demonstrated in postrenal ARF patients when other noninvasive techniques are not helpful.
Retrograde Pyelography
Similar to antegrade pyelography, the obstruction can be diagnosed and has additional advantage of treating the cause in the same session in selected patients.
Ultrasonography
It is the modality of choice to start evaluating patients with ARF. As a modality which is noninvasive and widely available, US provides valuable information on renal structure and hemodynamics.
Renal size: Normal in many cases. Increased size of kidneys may be found in acute tubular necrosis, acute glomerulonephritis, amyloidosis, and renal vein thrombosis. Decreased renal size may indicate underlying chronic renal pathology.
Cortical thickness: Increased in ATN, renal venous thrombosis, glomerulonephritis, and interstitial nephritis, while decreased in chronic renal dysfunctions.
Cortical echogenicity: Increased echogenicity and corticomedullary differentiation may accompany increased cortical thickness (Fig. 6.8). Meanwhile, hyperechoic cortex may be associated with decreased renal size and decreased cortical thickness, indicating chronic renal failure.
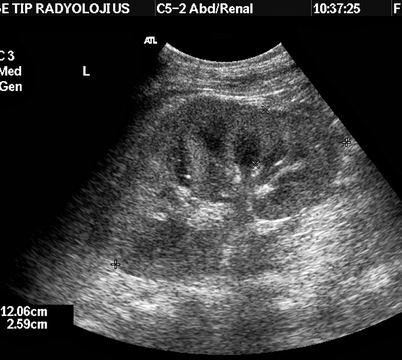
Fig. 6.8
Acute renal failure. Increased parenchymal thickness (26 mm) and cortical echogenicity in a case of rhabdomyolysis
Collecting system: May be bilaterally dilated in cases of postrenal ARF. Hydroureters may accompany bilateral hydronephrosis in cases due to bilateral lower urinary tract obstruction. In prerenal and intrinsic renal ARF cases, there is no sign of dilatation of urinary tract.
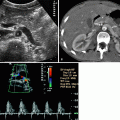
Spectral Doppler demonstrates normal RI values (less than 0.70) in most of the prerenal ARF patients, while RI indices are usually above 0.70 in intrinsic renal ARF cases. Clinical crush syndrome as a consequence of rhabdomyolysis is a typical example of renal disease with significant and reversible increase of RI and PI values (Fig. 6.9).
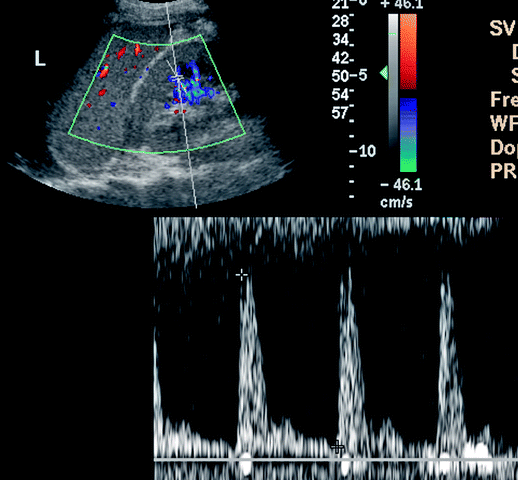
Fig. 6.9
Significantly increased RI in interlobar artery in a patient suffering from crush syndrome. RI: 0.93
Hepatorenal syndrome is a functional renal disorder without significant glomerular and tubular pathology, secondary to hepatic failure. Kidneys usually show high Doppler indices (Fig. 6.10). Renal function returns to normal when liver function normalizes.
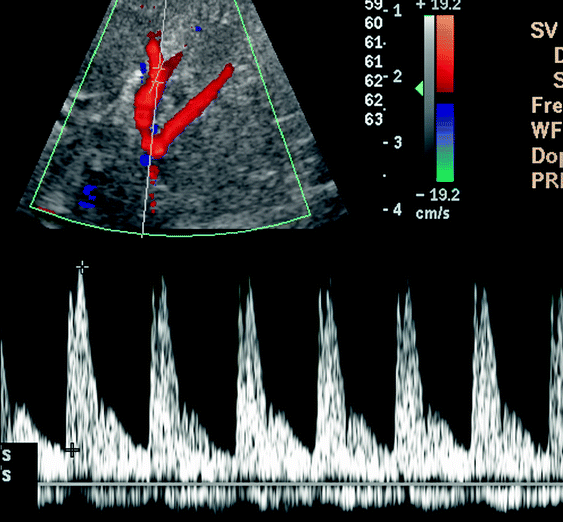
Fig. 6.10
Renal Doppler US demonstrates high Doppler indices (RI 0.84) in a case of hepatorenal syndrome
Computed Tomography
Non-contrast computerized tomography has the highest sensitivity in detecting urinary stones.
It also offers superior diagnostic information about retroperitoneal pathologies, namely, retroperitoneal fibrosis and malignancies.
Magnetic Resonance Imaging
This modality may be used as an adjunct to US when another cross-sectional imaging modality without ionizing radiation is needed. It provides structural information regarding the kidneys (size, parenchyma, collecting system) and retroperitoneum.
Magnetic resonance urography techniques may demonstrate dilatation of the urinary tract and the level of obstruction. While T2-weighted imaging delineates dilatation of the system in a static fashion, dynamic imaging using T1-weighted images and contrast media may supply information nearly equivalent to that provided with intravenous urography.
However, nephrogenic systemic fibrosis, a clinical entity demonstrated to be associated with exposure to gadolinium-containing MRI contrast media mostly diagnosed in patients with impaired renal function (GFR < 30), should be borne in mind.
Angiography
In ARF, arteriography is indicated to confirm and/or guide interventional therapy in cases of significant renal artery stenosis which can be diagnosed using Doppler ultrasound.
Renal arteriography can demonstrate multiple microaneurysms typical of polyarteritis nodosa.
Nuclear Scintigraphy
In ATN, the concentration of 99mTc-DTPA is poor with preserved renal perfusion. The progressive parenchymal accumulation of 131I-Hippuran or 99mTc-MAG3 and the absence of excretion are typical.
Pathology
Two patterns of injury are seen in acute tubular necrosis.
The predominant histologic finding in the first pattern is diffuse extensive necrosis of proximal tubular epithelium; this pattern is seen largely in toxic injury to the kidney (Fig. 6.11).
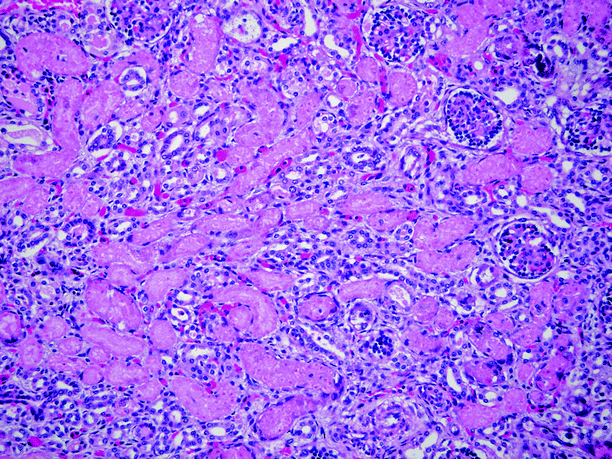
Fig. 6.11
Acute tubular necrosis. Glomeruli show no pathologic changes. Tubular epithelial cells show extensive coagulative necrosis, with loss of nuclei. No interstitial inflammation is present (Case courtesy of Beverly Dahms, M.D.)
In the second pattern, there is flattening of proximal tubular cells, resulting in luminal enlargement, and apoptosis (single cell necrosis). The apoptotic cells slough, becoming granular casts.
In both patterns, there is interstitial edema and a paucity of inflammatory cells. Grossly, the kidneys are enlarged due to interstitial edema. In an older individual with preexisting tubular atrophy, the kidneys may be of normal size.
Differential Diagnosis
Increased size of kidneys may be found in acute tubular necrosis, acute glomerulonephritis, amyloidosis, and renal vein thrombosis.
Cortical thickness: Increased in ATN, renal venous thrombosis, glomerulonephritis, interstitial nephritis, while decreased in chronic renal dysfunctions.
Acute renal failure should be considered in the differential diagnosis of thrombotic microangiopathies. Postrenal (obstructive) failure can be diagnosed with the demonstration of hydronephrosis using gray-scale ultrasound. The intrarenal arterial RI is often normal in acute prerenal failure.
Pearls and Pitfalls
Increased echogenicity and corticomedullary differentiation may accompany increased cortical thickness.
Normal RI values (less than 0.70) are present in most prerenal ARF patients, while RI > 0.70 are seen in patients with intrinsic renal ARF.
HIV Nephropathy
General Information
Renal disease is not an uncommon complication in patients who have HIV (human immunodeficiency virus). HIV-related nephropathy (HIVAN) is reported to be cause of 10 % of new end-stage renal disease cases in the United States. In autopsy series, the prevalence of HIV-associated nephropathy in the United States has been reported to be 3–7 %.
HIV nephropathy accounts for 40 % of patients of HIV-related renal disease and may be caused primarily from direct kidney infection and secondarily from adverse affects of medications used in HIV treatment.
HIVAN manifests histologically as focal glomerulosclerosis and membranous, membranoproliferative, and mesangial proliferative glomerulonephritis.
Opportunistic infections (fungal infections, Pneumocystis carinii, tuberculosis) in patients with HIV may also manifest with kidney disease.
Highly active antiretroviral therapy (HAART) with indinavir may result in calculus disease.
Patients with HIV may present with renal neoplasia, including Kaposi sarcoma and lymphoma.
HIV-associated nephropathy usually manifests as deterioration in renal function, nephrotic-range proteinuria, and azotemia in the absence of edema and hypertension.
Mortality and morbidity rates are affected by the early recognition of renal disease in HIV-infected patients.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree