Magnetic resonance (MR) enterography has an increasing role in the evaluation of the small bowel in patients with Crohn disease. MR enterography is accurate for disease assessment and can influence the choice of therapy. Functional sequences may increase the role of MR enterography in Crohn disease. Techniques such as high-resolution MR enterography, diffusion-weighted imaging, dynamic contrast-enhanced MR imaging, magnetization transfer, and MR motility imaging may allow better assessment of disease extent, activity, and severity. Quantitative analysis using these advanced techniques as well as the standard techniques may provide methods for evaluating and following the disease in the future.
Key points
- •
Three types of sequences constitute a basic magnetic resonance (MR) enterography protocol: single-shot fast spin echo, balanced refocused gradient echo, and fat-suppressed three-dimensional T1 gradient echo.
- •
High-resolution MR enterography, diffusion-weighted imaging, dynamic contrast-enhanced MR imaging, magnetization transfer, and MR motility imaging are newer techniques that may further enhance the accuracy of MR enterography.
- •
The goals of imaging in Crohn disease are accurate diagnosis and localization and assessing disease severity, activity, and extent. Assessing disease activity and the presence of penetrating disease have significant implications for the choice of therapy.
Introduction
Magnetic resonance (MR) enterography is playing an increasing role in the evaluation of the small bowel in patients with Crohn disease (CD). Conventional enteroclysis or small bowel follow-through have been used to evaluate the small bowel; however, with advances in technology, several cross-sectional imaging modalities including computed tomography (CT), MR imaging, positron emission tomography CT, and ultrasound are now available for small bowel imaging. The aim of imaging is to aid in the accurate assessment of the extent, severity, activity, and complications of CD. This role remains the subject of active research with numerous studies and publications in the past decade.
This review article discusses the conventional and emerging MR enterography sequences, and reviews the small bowel imaging findings of CD on MR imaging and the clinical role of MR imaging in the diagnosis of CD and monitoring therapy.
Pathology
CD is an idiopathic, chronic, transmural inflammatory disease that can affect any part of the gastrointestinal tract. It has a tendency toward segmental multifocal distribution. The causes and pathogenesis are not completely understood but are thought to be multifactorial. It is commonly complicated by fistulas, abscesses, bowel obstruction, and neoplasms. CD most commonly affects the small bowel, particularly the terminal ileum. About 70% of patients have small bowel involvement and about 30% have disease limited to the small bowel.
Clinical Presentation, Evaluation, and Management
The classic presentation of CD is abdominal pain, weight loss, and diarrhea; however, it can have various presentations and tends to have an unpredictable course marked by flares, remissions, and relapses. CD most commonly occurs in early adulthood with a second peak in the elderly. Its diagnosis is based on a combination of clinical findings, endoscopic appearance, biopsy, radiological studies, and/or biochemical markers. Most commonly, ileocolonoscopy and biopsies from the terminal ileum and colon are used to establish the diagnosis. In clinical practice, CD is stratified by disease severity (mild, moderate, or severe), disease location (upper gastrointestinal, ileal, ileocolonic, colonic, or perianal), extent of disease, and disease phenotype (penetrating, stricturing, or inflammatory). Clinical scoring systems have been developed to assess disease severity and activity, the most common of which is the Crohn Disease Activity Index (CDAI), which has been used in many clinical trials; however, it is inconclusive on the severity of the disease.
The management of CD is divided into medical therapies and surgical therapies. Therapeutic options are determined by an assessment of the disease location, severity, and extraintestinal complications. Surgery is typically used to treat the penetrating and stricturing complications of CD as well as neoplastic/preneoplastic lesions, suppurative complications, or medically intractable disease. Many patients require surgery during the course of the disease; however, surgery is not curative and the disease recurs in most patients within 5 years. Several medical therapies are used in treatment of CD, including corticosteroids, immunomodulators (such as azathioprine, mercaptopurine, and methotrexate), and biologic agents (such as tumor necrosis factor [TNF] alpha inhibitor). The goal of therapy is to induce and maintain symptomatic control, improve quality of life, and minimize short-term and long-term toxicity and complications. A newer goal of therapy is the induction and maintenance of mucosal healing.
MR Enterography Technique
The small bowel is first distended by oral contrast agents. In MR enterography, a large volume of enteric contrast is ingested by the patient over a period of 1 to 2 hours before the onset of the scan. MR enterography is generally preferred to MR enteroclysis because of its comparable performance and patient acceptance. The most commonly used type of oral contrast agents are biphasic agents that have low T1-weighted signal, which provides good visualization of wall enhancement, and have high T2-weighted signal, which allows assessment of the wall and fold thickness. A spasmolytic agent, such as glucagon, is usually administered before the scan to reduce bowel peristalsis.
Three types of sequences constitute a basic MR enterography protocol: half-Fourier acquisition single-shot turbo spin echo (HASTE), also known as single-shot fast spin echo (SSFSE); balanced refocused gradient echo, also known as balanced steady-state free precession (SSFP), fast imaging using steady-state acquisition (FIESTA), balanced fast field echo (FFE), fast imaging with steady-state precession (true FISP); and postcontrast fat-saturated three-dimensional (3D) T1-weighted ultrafast GRE. In the literature, SSFSE and true FISP sequences are considered complimentary and they are both commonly included in MR enterography protocols. We mostly rely on T2-weighted SSFSE images and, in most cases, have not found true FISP sequences as helpful. In addition to these standard sequences, T2-weighted turbo spin echo (TSE) sequence with fat suppression could be useful to improve the conspicuity of bowel wall edema, but their use is not standard.
Contrast-enhanced images are obtained using T1-weighted ultrafast GRE sequences with fat suppression, which are acquired in 2 or 3 dimensions. The postcontrast images are helpful in evaluating the extent and severity of the disease and are better at visualizing mural stenosis. There is no agreement on the optimal scan delay. Inflamed bowel segments enhance early compared with the normal small bowel segments, and enhancement keeps increasing into the delayed phases of 5 to 6 minutes after contrast enhancement. Normal jejunal segments enhance early and more intensely compared with ileal segments. In addition, nondistended small bowel segments may also mimic increased enhancement due to inflammation. One main advantage of MR enterography is that multiple phases of contrasts could be obtained and dynamic contrast enhancement techniques have the potential to provide quantitative parameters to assess wall enhancement and, therefore, disease activity.
Introduction
Magnetic resonance (MR) enterography is playing an increasing role in the evaluation of the small bowel in patients with Crohn disease (CD). Conventional enteroclysis or small bowel follow-through have been used to evaluate the small bowel; however, with advances in technology, several cross-sectional imaging modalities including computed tomography (CT), MR imaging, positron emission tomography CT, and ultrasound are now available for small bowel imaging. The aim of imaging is to aid in the accurate assessment of the extent, severity, activity, and complications of CD. This role remains the subject of active research with numerous studies and publications in the past decade.
This review article discusses the conventional and emerging MR enterography sequences, and reviews the small bowel imaging findings of CD on MR imaging and the clinical role of MR imaging in the diagnosis of CD and monitoring therapy.
Pathology
CD is an idiopathic, chronic, transmural inflammatory disease that can affect any part of the gastrointestinal tract. It has a tendency toward segmental multifocal distribution. The causes and pathogenesis are not completely understood but are thought to be multifactorial. It is commonly complicated by fistulas, abscesses, bowel obstruction, and neoplasms. CD most commonly affects the small bowel, particularly the terminal ileum. About 70% of patients have small bowel involvement and about 30% have disease limited to the small bowel.
Clinical Presentation, Evaluation, and Management
The classic presentation of CD is abdominal pain, weight loss, and diarrhea; however, it can have various presentations and tends to have an unpredictable course marked by flares, remissions, and relapses. CD most commonly occurs in early adulthood with a second peak in the elderly. Its diagnosis is based on a combination of clinical findings, endoscopic appearance, biopsy, radiological studies, and/or biochemical markers. Most commonly, ileocolonoscopy and biopsies from the terminal ileum and colon are used to establish the diagnosis. In clinical practice, CD is stratified by disease severity (mild, moderate, or severe), disease location (upper gastrointestinal, ileal, ileocolonic, colonic, or perianal), extent of disease, and disease phenotype (penetrating, stricturing, or inflammatory). Clinical scoring systems have been developed to assess disease severity and activity, the most common of which is the Crohn Disease Activity Index (CDAI), which has been used in many clinical trials; however, it is inconclusive on the severity of the disease.
The management of CD is divided into medical therapies and surgical therapies. Therapeutic options are determined by an assessment of the disease location, severity, and extraintestinal complications. Surgery is typically used to treat the penetrating and stricturing complications of CD as well as neoplastic/preneoplastic lesions, suppurative complications, or medically intractable disease. Many patients require surgery during the course of the disease; however, surgery is not curative and the disease recurs in most patients within 5 years. Several medical therapies are used in treatment of CD, including corticosteroids, immunomodulators (such as azathioprine, mercaptopurine, and methotrexate), and biologic agents (such as tumor necrosis factor [TNF] alpha inhibitor). The goal of therapy is to induce and maintain symptomatic control, improve quality of life, and minimize short-term and long-term toxicity and complications. A newer goal of therapy is the induction and maintenance of mucosal healing.
MR Enterography Technique
The small bowel is first distended by oral contrast agents. In MR enterography, a large volume of enteric contrast is ingested by the patient over a period of 1 to 2 hours before the onset of the scan. MR enterography is generally preferred to MR enteroclysis because of its comparable performance and patient acceptance. The most commonly used type of oral contrast agents are biphasic agents that have low T1-weighted signal, which provides good visualization of wall enhancement, and have high T2-weighted signal, which allows assessment of the wall and fold thickness. A spasmolytic agent, such as glucagon, is usually administered before the scan to reduce bowel peristalsis.
Three types of sequences constitute a basic MR enterography protocol: half-Fourier acquisition single-shot turbo spin echo (HASTE), also known as single-shot fast spin echo (SSFSE); balanced refocused gradient echo, also known as balanced steady-state free precession (SSFP), fast imaging using steady-state acquisition (FIESTA), balanced fast field echo (FFE), fast imaging with steady-state precession (true FISP); and postcontrast fat-saturated three-dimensional (3D) T1-weighted ultrafast GRE. In the literature, SSFSE and true FISP sequences are considered complimentary and they are both commonly included in MR enterography protocols. We mostly rely on T2-weighted SSFSE images and, in most cases, have not found true FISP sequences as helpful. In addition to these standard sequences, T2-weighted turbo spin echo (TSE) sequence with fat suppression could be useful to improve the conspicuity of bowel wall edema, but their use is not standard.
Contrast-enhanced images are obtained using T1-weighted ultrafast GRE sequences with fat suppression, which are acquired in 2 or 3 dimensions. The postcontrast images are helpful in evaluating the extent and severity of the disease and are better at visualizing mural stenosis. There is no agreement on the optimal scan delay. Inflamed bowel segments enhance early compared with the normal small bowel segments, and enhancement keeps increasing into the delayed phases of 5 to 6 minutes after contrast enhancement. Normal jejunal segments enhance early and more intensely compared with ileal segments. In addition, nondistended small bowel segments may also mimic increased enhancement due to inflammation. One main advantage of MR enterography is that multiple phases of contrasts could be obtained and dynamic contrast enhancement techniques have the potential to provide quantitative parameters to assess wall enhancement and, therefore, disease activity.
Advances in MR enterography technique
High-resolution Images
High-resolution sequences provide the potential for focused evaluation of a small segment of small bowel that is suspected of having disease. In an article by Sinha and colleagues, high-resolution images were obtained of bowel segments that were selected by the radiologist based on suspicion on reviewing the standard sequences. A combination of fat-suppressed true FISP and SSFSE sequences with a small field of view were then obtained. The images were aligned parallel and perpendicular to that bowel segment and were obtained with contiguous thin sections (2–3 mm thick), 160-mm to 250-mm field of view, and a matrix of 128 to 256 by 128 to 256, providing in-plane resolution of 1 to 2 mm. Multiplanar and endoluminal views can then be reconstructed. The investigators reported that this technique increases diagnostic confidence by depicting aphthous ulcers and transmural and mesenteric changes and allows more accurate characterization and classification of CD. In a subsequent study, Sinha and colleagues reported higher accuracy of the high-resolution MR enterography in the diagnosis of bowel ulceration, fistulae, and abscesses using surgical and histologic results as the gold standard. In that study, high-resolution true FISP sequences with smaller fields of view were obtained in each of the 4 abdominal quadrants, adding only 3 minutes to the length of the study. In addition, perpendicular or parallel high-resolution images of involved segments were acquired by the supervising radiologist, if required.
3T Imaging
Imaging with 3-T MR imaging compared with 1.5-T MR imaging increased the signal/noise ratio (SNR) by about 1.7-fold to 1.8-fold, which can translate into improving the special resolution or reducing the scan time, both of which would be of particular benefit in the evaluation of the bowel. The T1 shortening effect of gadolinium is more pronounced in 3-T imaging, yielding improved contrast/noise ratio, which translated into increased conspicuity of enhancing structure and lesions, as well as decrease in the amount of gadolinium needed. In addition, fat suppression in 3 T is more pronounced than in 1.5 T because of the wider difference between processional frequencies of fat and water in 3 T.
The advantages of 3-T imaging come at the cost of increasing artifacts and increasing energy deposition. Doubling the magnetic field strength quadruples the specific absorption rate (SAR), which becomes a limitation of 3-T imaging, particularly in radiofrequency (RF)-intensive sequences such as SSFSE and true FISP. This effect is particularly relevant in abdominal imaging because the SAR is proportional to the volume being imaged. The increase in SAR can be mitigated by increasing the recovery time and decreasing the flip angle at the expense of increasing the acquisition time and altering the contrast enhancement. The use of parallel imaging can reduce the number of RF pulses and hence the SAR. In addition, using torso coils instead of body coils can result in a more efficient RF deposition. B1-inhomogeneity artifacts (also referred to as standing wave artifacts or dielectric effects) are local variations in signal intensity across the image that reduce the quality of the image and can obscure certain portions of it by a dark band of low signal. This artifact is more pronounced in 3-T imaging, particularly in wider body habitus, and is even more pronounced in the presence of fluid in the abdomen. This artifact can be reduced by the use of parallel imaging. As an alternative, dielectric pads or RF cushions containing conductive gel could be placed anterior to the abdomen, which was shown to reduce or eliminate the dark B1-inhomogenity artifacts on TSE T2-weighted imaging such as HASTE, but not on true FISP. Chemical shift artifacts of the first type are amplified on 3-T imaging and can be mitigated by increasing the RF at the expense of reducing the SNR. It can also be reduced by applying fat-suppression techniques. Early studies have shown the feasibility of performing high-quality MR enterography on 3 T.
Dynamic Contrast-enhanced MR Imaging
Dynamic contrast-enhanced MR (DCE-MR) imaging is being increasingly investigated for quantitative assessment of tissue perfusion. It is most popular in oncologic imaging but increased tissue perfusion is also a hallmark of inflammation. Actively inflamed bowel segments show increased enhancement with an early hyperenhancement that increases over time until a plateau is reached. Bowel that is actively inflamed has also been observed to display a different enhancement pattern and dynamics than inactive disease. Studies have shown a correlation between dynamic enhancement parameters such as peak uptake and slope of enhancement curve and the clinical activity of the disease as well as inflammatory activity on biopsy. Such parameters that describe the enhancement curve (signal vs time) are referred to as semiquantitative analysis. Quantitative DCE-MR imaging analysis has also been applied to MR enterography more recently yielding promising results. In quantitative analysis, pharmacokinetic models are used to convert the signal intensity to the tissue concentration of gadolinium and calculate parameters that reflect the tissue perfusion. The inflamed bowel segments have shown faster values for the transfer constants of the contrast from the intravascular space to the extravascular extracellular space (K trans ) and larger volumes of the extravascular extracellular space per unit volume of tissue (V e ).
These semiquantitative and quantitative parameters derived from DCE-MR imaging can be used in distinguishing areas of active inflammation and also have the potential to monitor response following treatment. More efforts to standardize image acquisition and analysis and more studies to confirm the clinical usefulness of these parameters are needed.
Diffusion-weighted Imaging
Diffusion-weighted imaging (DWI) is increasingly becoming a standard part of MR imaging protocols in the abdomen and pelvis. Although most of the literature on DWI has investigated its role in various abdominal malignancies, DWI may likewise have a role as a quantifiable indicator of inflammation. Inflamed bowel segments have shown restricted diffusion compared with normal bowel. Oto and colleagues showed that actively inflamed small bowel segments in patients with CD can be differentiated from normal small bowel loops based on DWI and quantitative DCE-MR imaging parameters. The combination of DWI and DCE-MR imaging improved the specificity for detection of active inflammation. Kiryu and colleagues reported a sensitivity, specificity, and accuracy of 86.0%, 81.4%, and 82.4%, respectively, for DWI in the detection of disease-active segments. Restricted diffusion was also shown in inflamed portions of the colon in patients with ulcerative colitis and it could be of similar accuracy to contrast-enhanced sequences. In clinical practice, DWI is useful in highlighting bowel segments of active disease and hence it is becoming a standard component of the MR enterography protocol; however, more studies are needed to explore the full potential of this technique and guide its clinical applications. DWI also has the potential to provide quantitative information about the level of inflammation because the apparent diffusion coefficient (ADC) can be calculated from the bowel wall.
MR Motility Imaging Techniques
Real-time images with high temporal resolution (∼1–2 seconds) can be acquired by repeatedly acquiring ultrafast true FISP images through a coronal slab of the abdomen to evaluate the small bowel peristalsis. This technique has been referred to as MR motility imaging or cine MR enterography and can be used to identify areas of altered motility, specifically focal areas of paralysis or hypomotility. Froehlich and colleagues detected a larger number of CD-specific findings on cine MR enterography than on static MR enterography alone, and identified significantly more patients with CD than were identified on MR enterography alone. When a change in motility was identified on the cine MR imaging, the corresponding location on the static images was evaluated for CD-specific findings. The CD-specific findings that were analyzed were small bowel wall thickening, stenosis, prestenotic dilatation, layering of the bowel wall (thickened wall combined with alternating hyperintense and hypointense layers within the wall), ulcers, the comb sign, fistulas, and abscess. Kitazume and colleagues described the finding of asymmetric involvement or mesenteric rigidity with antimesenteric flexibility that correlated with longitudinal ulcer in small bowel CD. Software tools for automated evaluation and quantitative analysis of motility on cine MR enterography techniques are only available as research tools and are not yet commercially available. Menys and colleagues quantified the motility of the terminal ileum using a new parameter, the motility index (MI), in 28 patients with CD, showing a significant difference in motility between noninflamed and inflamed terminal ileum. The MI was negatively correlated with the acute inflammation score that was assigned to the biopsy, suggesting a role for quantified motility in assessing disease activity.
Magnetization Transfer
One of the most important questions in CD is differentiating between areas of active inflammation that may respond to medical therapy and areas of fibrosis that may require surgical intervention. Most of the current techniques provide, at best, an indirect evaluation of fibrosis, with the recognition that there is significant overlap between fibrosis and inflammation on imaging. Magnetization transfer (MT) is a promising MR technique that, in theory, can be used to directly image and quantify fibrosis, differentiating it from edema and inflammation. MT generates contrast that is primarily determined by the fraction of large macromolecules or immobilized phospholipid cell membranes in tissue; stiff body substances such as muscle or fibrotic tissue therefore have a high magnetization transfer effect. In an early study on rats, Adler and colleagues showed that MT ratio correlated with tissue collagen in the bowel but it remained unchanged in control rats that showed inflammation but no fibrosis. Pazahr and colleagues showed that MT imaging of the small bowel wall is feasible in humans if there is sufficient image quality and may help with the identification of fibrotic scarring in patients with CD. The ability to detect fibrosis in small bowel strictures can be useful in choosing between medical therapy in nonfibrotic stenosis and surgical intervention in fibrotic stricture. It can also evaluate response to new therapeutic agents targeted at treating fibrotic strictures.
MR imaging findings of small bowel in CD
Bowel Wall Findings
Bowel wall thickening ( Fig. 1 ) is the most reported imaging finding of CD. It is best assessed on SSFSE sequence or postcontrast T1-weighted images. The normal bowel wall thickening is 1 to 3 mm, provided the bowel is well distended for evaluation, whereas it typically ranges from 5 to 10 mm in bowel affected by CD. The thickening may decrease in remission, but it is likely to remain thicker than unaffected bowel.
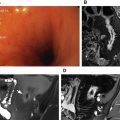
Edema in the bowel wall manifesting as increased intramural T2 signal ( Fig. 2 ) is one of the most useful finding to indicate active or sever inflammation. Evaluating edema is best done on fat-saturated T2 sequences to differentiate edema from the intramural fat that can be present in chronic CD.
Bowel wall enhancement may be one of the earliest signs of disease activity. It has been repeatedly reported to correlate with bowel inflammation and activity of the disease. The pattern of enhancement may be useful in assessing active disease. Homogeneous transmural enhancement ( Fig. 3 ) can be seen in active or chronic disease and is therefore nonspecific. A layered pattern of enhancement, also known as mural stratification ( Fig. 4 ), is more specific for active inflammation. Bowel wall enhancement has also been observed to decrease in patients transitioning into remission. Restricted diffusion of a bowel segment is another finding suggesting active inflammation in that segment ( Fig. 5 ). Hyperintensity on DWI was shown to highly correlate with disease activity evaluated on conventional MR enterography.