Solitary Lung Nodule or Mass
A pulmonary nodule is defined as any pulmonary lesion that is well-defined, discrete, approximately circular, and 3 cm or less in diameter. The term mass is used for lesions greater than 3 cm in diameter. The differential diagnosis of a nodule or mass detected on a radiograph or CT is very broad ( Table 4.1 ). However, more than 95% fall into one of three groups: malignant neoplasms (primary or metastatic), infectious granulomas (tuberculosis or endemic fungi), or less commonly, benign tumors (neoplastic or nonneoplastic). Approximately 40% of solitary lung nodules detected on a chest radiograph represent primary lung cancer.
| Etiology | Features |
|---|---|
| CONGENITAL | |
| Bronchogenic cyst | Water density on CT (50%). Almost all have high signal intensity on T2-weighted MRI |
| Pulmonary arteriovenous malformation | Round, oval, or slightly lobulated. CT shows a feeding artery and draining vein |
| INFECTIOUS/INFLAMMATORY | |
| Tuberculosis | Common. Mainly upper lobes. Frequently calcifies |
| Histoplasmosis | Common in endemic areas. Frequently calcifies |
| Coccidioidomycosis | Common in endemic areas. Frequently cavitates. Rarely calcifies |
| Echinococcus | Water density on CT |
| Lung abscess | Cavitation or low-attenuation center on CT |
| Focal (round) pneumonia | Less common in adults than in children. Tends to have ill-defined margins |
| Granulomatosis with polyangiitis | A solitary nodule is uncommon |
| Rheumatoid nodules | Necrobiotic rheumatoid nodules most commonly multiple |
| Organizing pneumonia | Tends to have ill-defined margins |
| Lipoid pneumonia | Irregular margins. Foci of fat density on CT (80%) |
| Conglomerate fibrosis in sarcoidosis | Uncommon. Typically bilateral, perihilar, and upper lobes. Air bronchograms |
| Conglomerate fibrosis in talcosis | Intravenous drug user. Typically upper lobes. Characteristic high attenuation because of talc deposition |
| PNEUMOCONIOSIS | |
| Progressive massive fibrosis in silicosis | Typically perihilar, bilateral, and upper lobes. Other findings of silicosis |
| NEOPLASTIC | |
| Pulmonary carcinoma | Smooth, lobulated, or most commonly, spiculated margins |
| Carcinoid tumor | Relatively uncommon. Usually smooth margins |
| Hamartoma | Uncommon. Tends to have lobulated margins. Sixty percent have areas of fat density on CT. Popcorn calcification characteristic but uncommon |
| Metastasis | Solitary metastasis uncommon except for sarcoma. Tends to have smooth margins |
| TRAUMATIC | |
| Hematoma | Uncommon. Smooth margins. Usually in a peripheral location. Slow but progressive decrease in size |
Most of the nodules detected radiographically measure more than 1 cm in diameter. Noncalcified nodules less than 7 mm in diameter are seldom visible on radiographs. In contrast, nodules as small as 1 to 2 mm in diameter are readily seen on CT. More than 50% of smokers who undergo thin-section CT have small lung nodules, almost all of which measure less than 7 mm in diameter. The vast majority of these small nodules are benign.
The first step in assessing a lesion detected on a chest radiograph is to determine that it is indeed a lung nodule rather than a pleural or chest wall abnormality, such as a healing rib fracture, pleural neoplasm, or skin lesion projecting over the lung ( Fig. 4.1 ). The next step is to evaluate the imaging characteristics of the nodule to determine whether there are findings that favor a benign or a malignant lesion. After this, one must integrate the imaging characteristics with clinical findings such as patient age, risk factors such as cigarette smoking or a known primary extrathoracic neoplasm, recent travel history, and symptoms such as fever or hemoptysis. For example, a nodule in an adolescent patient is likely to be benign, regardless of its margin characteristics. A growing noncalcified nodule in an older smoking patient is likely to be malignant even though it has smooth borders. In the vast majority of cases, pulmonary metastases are multiple. However, single lung metastases may also occur, particularly with sarcomas and testicular tumors.

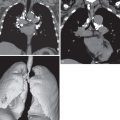
Advances in multidetector CT have improved nodule detection and characterization with thin-section imaging, acquisition within a single breath-hold, high-quality volumetric image data, as well as multiplanar and maximum intensity projection (MIP) reconstructions ( Fig. 4.2 ). Of note, multiplanar CT images should be fully used in assessing a nodule’s shape and appearance because linear opacities, as seen with atelectasis or scar, may appear nodular in a single plane. In the vast majority of cases, no intravenous contrast material is necessary for nodule characterization.

In evaluating lung nodules, it is useful to divide them into two categories: (1) those that are clearly benign, as determined by rigidly defined radiologic signs, and (2) those of indeterminate nature, which represent all other lesions. This allows one to distinguish benign from malignant nodules with a reasonable degree of confidence.
Location of the Lesion
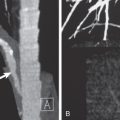
An intrapulmonary nodule tends to form an acute angle with the contiguous pleura and to have well-defined margins when seen en face (see Fig. 4.1A and B ). A nodule originating within the pleural space, rib, or chest wall displaces the pleura and underlying lung inward (see Fig. 4.1C and D ). It therefore tends to have tapered margins and form an obtuse angle with the chest wall and to have one side demonstrating a well-defined margin and the opposite side a poorly defined margin when seen en face (incomplete border sign). Similar to all other radiologic signs, this sign is fallible. Occasionally, an extrapulmonary nodule relates to the lung at an acute angle and an intrapulmonary mass at an obtuse angle.
Imaging Characteristics of Lung Nodules
Distinction between benign and malignant lesions can often be established with a reasonable degree of confidence from the clinical and radiographic findings, although findings are seldom pathognomonic. The main diagnostic radiologic criteria are nodule size, change in size over time, attenuation (including presence of calcification or fat), margin characteristics, and 18 F-fluorodeoxyglucose (FDG) avidity.
Size
The prevalence of malignancy typically varies by size with less than 1% for nodules less than 5 mm, 0.5% to 28% for nodules 5 to 10 mm, and 64% to 82% for nodules greater than 20 mm. When interpreting these figures it is important to remember that the likelihood of carcinoma is influenced strongly by other factors such as age and smoking history.
Change in Size
Because uncontrolled growth is a characteristic feature of cancer, in the absence of an infectious or inflammatory process, an enlarging pulmonary nodule should be considered malignant until proven otherwise by tissue sampling. However, because some benign lesions such as hamartoma and histoplasmoma can enlarge and some adenocarcinomas can grow slowly, an increase in the size of a nodule, by itself, should not be the sole consideration governing the therapeutic approach. Traditionally, 2-year stability in size has been considered a reasonably reliable criterion of benignity; however, the validity of this statement has been questioned, particularly with respect to ground-glass or part-solid nodules, in which a slight change in size may be difficult to appreciate. Patients with ground-glass and part-solid nodules require longer monitoring as outlined by the Fleischner criteria ( Tables 4.2 and 4.3 ).
| SOLID NODULE | |||||
| <6 mm | <6 mm | 6–8 mm | 6–8 mm | >8 mm | |
| Low Risk | High Risk | Low Risk | High Risk | Any Risk | |
| Solitary | No follow-up | 12-mo CT if suspicious a | 6- to 12-mo CT, consider 18- to 24-mo CT if suspicious a | Both 6- to 12-mo CT and 18- to 24-mo CT | Consider CT, PET/CT, or tissue sampling at 3 mo |
| Multiple | No follow-up | 12-mo CT if suspicious b | 3- to 6-mo CT, consider 18- to 24-mo CT if suspicious b | Both 3- to 6-mo CT and 18- to 24-mo CT | 3- to 6-mo CT; additional 18- to 24-mo CT if high risk or suspicious b |
a Suspicious by morphology or with upper lobe location.
| SOLITARY SUBSOLID NODULE | |||
| <6 mm | ≥6 mm | ||
| Initial follow-up: | Additional follow-up if stable: | ||
| Ground-glass | No follow-up | 6- to 12-mo CT | Biannual CT until 5 years |
| Part-solid | No follow-up | 3- to 6-mo CT | Annual CT at least 5 years (Note: Solid component must remain <6 mm; persistent part-solid nodules with solid component measuring ≥6 mm should be considered highly suspicious for adenocarcinoma.) |
| MULTIPLE SUBSOLID NODULES | |||
| <6 mm | ≥6 mm | ||
| Initial follow-up | 3- to 6-mo CT | 3- to 6-mo CT | |
| Additional follow-up (if stable) | Consider 24- and 48-mo CT | Per most suspicious nodule | |
Use of the doubling time provides a more accurate assessment of the nature of a pulmonary nodule than a simple increase in size does. Doubling refers to volume, not diameter. Assuming a nodule to be spherical, its diameter must be multiplied by 1.25 to obtain the diameter of a sphere whose volume is double (e.g., the volume of a nodule 2 cm in diameter is doubled by the time that its diameter reaches 2.5 cm). A doubling of diameter represents an eightfold increase in volume. Measurement of doubling time requires at least two serial chest radiographs or CT scans showing a roughly spherical lesion whose diameter can be averaged from measurements in at least two planes. In a study of 218 pulmonary nodules (177 malignant and 41 benign), virtually all nodules whose doubling time was 7 days or less or 465 days or longer were benign. A pulmonary nodule whose rate of growth falls between these limits is often malignant. A pitfall to be aware of is indolent lung cancer (particularly adenocarcinoma), which may manifest as a ground-glass nodule gradually enlarging over 5 years or longer. For this reason, ground-glass nodules merit longer follow-up, per the Fleischner criteria (see Table 4.3 ). Perhaps the most useful application of the growth rate principle in assessing solitary nodules is in patients older than 40 years, in whom the incidence of malignancy increases markedly. In a study of individuals in this older age group, almost every solitary nodule whose doubling time was less than 37 days was benign.
Attenuation: Calcification
Benign patterns of calcification within a solitary pulmonary nodule are one of the most reliable signs that a lesion is benign. Four patterns of calcification are characteristically associated with benign lesions: diffuse, laminated, central, and popcorn ( Fig. 4.3 ). The diffuse ( Fig. 4.4 ) and laminated (synonym: concentric or laminar) types of calcification are virtually diagnostic of a granuloma, although this cannot be applied to pulmonary masses (>3 cm in size). A small central nidus of calcification is seen most commonly with granulomatous lesions (typically tuberculosis and histoplasmosis), although it also occurs in some hamartomas. Popcorn calcification is characteristic of hamartoma ( Fig. 4.5 ). By contrast, the presence of an eccentric calcification in a nodule or mass may represent incorporation of a calcified granuloma within the substance of a carcinoma.



Calcification within a pulmonary nodule is more readily seen on low-kilovoltage than on high-kilovoltage radiographs and much better characterized on CT than on a radiograph. Consequently, CT is the study of choice in assessing pulmonary nodules. In a study of 634 nodules assessed with CT, 153 were diagnosed correctly as benign based on the presence of central or diffuse calcification; although focal areas of calcification were identified in 13% of the primary lung tumors, only 1 carcinoid tumor had a benign pattern of calcification. The other malignant tumors had neither central nor diffuse calcification.
Calcification in malignant tumors may be seen in several circumstances: (1) most important from a differential diagnosis point of view, the isolated instance of a peripheral primary carcinoma engulfing a calcified granuloma, in which case the calcification is usually eccentric; (2) a solitary metastasis of osteogenic sarcoma or chondrosarcoma with bone formation in the osteoid or calcification of the malignant cartilage; (3) occasional primary pulmonary carcinoid tumors; and (4) calcification in large pulmonary carcinomas ( Fig. 4.6 ), particularly in tumors larger than 3 cm in diameter. The calcification in these large tumors may be punctate, chunky, or amorphous in appearance and central, peripheral, or diffuse in distribution. It is related to psammoma bodies, dystrophic calcification of necrotic carcinoma, or incorporation of a focus of previous granulomatous inflammation or calcified bronchial cartilage within the tumor.

Optimal assessment of lung nodules for the presence of calcification by CT requires the use of thin sections (1- to 2-mm collimation) through the nodule so as to minimize volume averaging and errors in nodule classification based on imaging appearance (e.g., solid vs. subsolid). Foci of calcification that are visible on thin-section CT usually have attenuation values of 400 Hounsfield units (HU) or higher; in the absence of such foci, attenuation values of 200 HU or higher can be considered to represent calcification.
Attenuation: Fat Density
Demonstration of the presence of fat (−30 to −120 HU) attenuation values on thin-section CT within a smoothly marginated lung nodule is virtually diagnostic of hamartoma; however, this should be interpreted with caution as volume-averaging or necrosis can also result in similar attenuation values. In a study of 47 hamartomas, 18 (38%) showed areas of fat, and 10 (21%) showed foci of calcium and fat (see Fig. 4.5 ). Fat within a spiculated lesion is most suggestive of lipoid pneumonia ( Fig. 4.7 ). The radiographic and CT findings of lipoid pneumonia include single or multiple nodules or masses and localized or confluent areas of consolidation. In approximately 80% of patients, focal areas of fat attenuation can be seen on CT using soft tissue windows.

Attenuation: Water Density
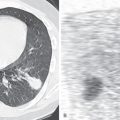
Water density (0 HU) on CT plus a thin or invisible wall is diagnostic of a cystic lesion. The differential diagnosis includes bronchogenic cyst, congenital pulmonary airway malformation, hydatid cyst, and fluid-filled bulla ( Fig. 4.8 ). Only 50% of bronchogenic cysts have water density on CT; however, virtually all have characteristic high signal intensity similar to that of cerebrospinal fluid on T2-weighted MR images. Of note, air-fluid levels may occur in lung abscesses or infected cavitary lesions, which require follow-up to ensure resolution.

Attenuation: Air Bronchogram
On thin-section CT, air bronchograms and bronchiolograms occur more commonly with pulmonary carcinomas than with benign nodules. For example, in a review of 132 patients, these features were identified in 33 (29%) of 115 carcinomas and in only 1 (6%) of 17 benign nodules. The patent airways are frequently tortuous and ectatic.
Attenuation: Bubble Lucencies (Pseudocavitation)
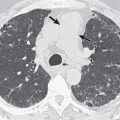
Nodules containing round or oval foci of air attenuation are highly suspicious for malignancy, particularly lung adenocarcinoma ( Fig. 4.9 ). These bubble lucencies (synonym: pseudocavitations) are rarely identified in benign lesions. Correlation with pathologic findings has shown that the lucencies usually represent patent airways, often ectatic, or foci of emphysema surrounded by carcinoma.

Attenuation: Focal Nodular Ground-Glass Opacity
Ground-glass opacity is a term used to describe increased lung attenuation through which pulmonary vessels can still be seen. The differential diagnosis of a focal area of ground-glass opacity or consolidation is broad and includes focal bacterial, fungal, or viral pneumonia; organizing pneumonia; focal fibrosis; lipoid pneumonia; atypical adenomatous hyperplasia; adenocarcinoma (primary and metastatic); and primary lymphoma of the lung (MALToma). Given this broad differential and the likelihood that infectious/inflammatory lesions will regress or resolve quickly, ground-glass nodules 6 mm and larger should be followed with a 6- to 12-month CT; whereas those smaller than 6 mm can generally be ignored. However, given the possibility of slow-growing cancers ( Fig. 4.10 ), if such nodules persist the revised Fleischner criteria recommend CT follow-up for 5 years rather than 3 years as previously recommended (see Tables 4.2 and 4.3 ). Nodules with mixed ground-glass opacity and a solid component on CT (part-solid nodule) are more likely to be malignant than purely soft tissue nodules (see Fig. 4.10 ). These nodules often represent adenocarcinoma and require close follow-up or resection.