The Need for Reducing Unnecessary Biopsies in Breast MRI
Dynamic contrast-enhanced (DCE) magnetic resonance imaging (MRI) of the breast—referred to as standard breast MRI throughout this chapter—is considered the most sensitive test for diagnosis of malignant breast lesions. Standard breast MRI relies on highlighting tumor vasculature by means of intravenously applied gadolinium (Gd)-based contrast media. Because (biologically active) cancers require nutrients, a complex cascade of cytokines induces the growth of new vessels, a process known as neoangiogenesis. This ensures that practically all breast cancers show contrast uptake on DCE MRI images. A lack of enhancement in a technically adequate breast MRI examination excludes biologically significant breast cancer with high certainty. Nonenhancing breast cancers are uncommon, and the negative predictive value of a negative MRI scan for invasive breast cancer has been reported as 99% in two meta-analyses. The high sensitivity is the reason why—besides classical indications, such as high-risk screening and staging and therapy monitoring of breast cancer—breast MRI is gaining increasing acceptance for applications of problem-solving and intermediate risk screening. On the other hand, enhancement itself is not specific: benign ductal proliferation and ductal carcinoma in situ (DCIS) can show similar patterns of angiogenesis as invasive breast cancer. Breast MRI reporting according to the Breast Imaging Reporting and Data System (BI-RADS) lexicon is standardized, but BI-RADS does not include a clinical decision rule such as PI-RADS for the prostate. The lack of formal guidance makes breast MRI interpretation a subjective, experience-dependent task. In addition, BI-RADS semantic features contain redundant information and show significant overlap between benign and malignant lesions. Insecurity of the reporting physician will lead to biopsy and follow-up recommendations that are ultimately unnecessary. False-positive findings pose a challenge to the broad implementation of breast MRI, as the availability of MRI-guided biopsy facilities is limited, and cost-effectiveness of supplemental breast MRI screening in women with dense breasts critically depends on MRI specificity.
Diffusion-weighted imaging (DWI) can help with this clinical problem: it provides additional quantitative information that is fast to acquire. Malignant neoplastic tissue changes cause a characteristic decrease in water diffusivity as measured by the apparent diffusion coefficient (ADC) in standard DWI. This can be put to clinical use, as the probability of cancer decreases with increasing ADC values, so that breast cancer can be excluded practically beyond a certain ADC threshold.
DWI and the ADC in Clinical Practice
CHOICE OF b VALUES
The general physical principle of DWI is covered in Chapter 1 in this book. From a clinical perspective, DWI is a technique sensitive to the molecular (Brownian) motion of water and, in some circumstances, the “pseudoincoherent motion” caused by flowing blood in small vessels (perfusion). To assess water diffusion in a quantitative manner, at least two sets of images must be acquired. The first set of images is a non- (or very low) diffusion-weighted image with recommended b values of 0 (or at least lower than 50) s/mm 2 . As the clinical relevance of using nonzero low b values for suppressing signal from larger vessels in the breast is less evident than in the liver, for example, there is no clear benefit of using b 50 s/mm 2 instead of b 0 s/mm 2 images. The second set of images is diffusion weighted, and a b value of 800 s/mm 2 is currently recommended. This b value is determinant for the DWI contrast level and the calculated ADC value. The simple diffusion model used for DWI assumes a monoexponential signal loss, independent of the choice of b value, assuming free (not hindered) water diffusion. However, in tissues, water diffusion is highly hindered, which results in a decrease of the amount of signal loss when the b value increases. This hindrance is what makes DWI so sensitive to changes in tissue microstructure, as seen in tumors, but it also implies that choice of b values is a major issue for DWI acquisition standardization. Other less important issues for standardization are the pulse gradient setup and timing, which might also affect the ADC to some degree. It is recommended to perform the acquisition of DWI before contrast medium injection, as the local background magnetic gradient created by Gd-filled vessels might result in small ADC underestimation. However, this effect has not been shown to be clinically significant in measuring breast lesion ADC, so that the acquisition of DWI after DCE MRI might be acceptable when clinically necessary. Calculation of the ADC is usually automatically performed by scanner software; the main adjustment that can be performed is a low b value filter that ignores voxels with a base signal below the chosen arbitrary value. This reduces noisy voxels, including those covering suppressed fat signal, but it may make anatomical orientation and correlation with anatomical images difficult. The ADC map itself is a parametric map, the signal intensity of each voxel corresponds to an ADC value that can be considered a spatially resolved quantitative imaging biomarker.
Integrating DWI in the Overall Multiparametric Breast MRI Protocol
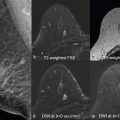
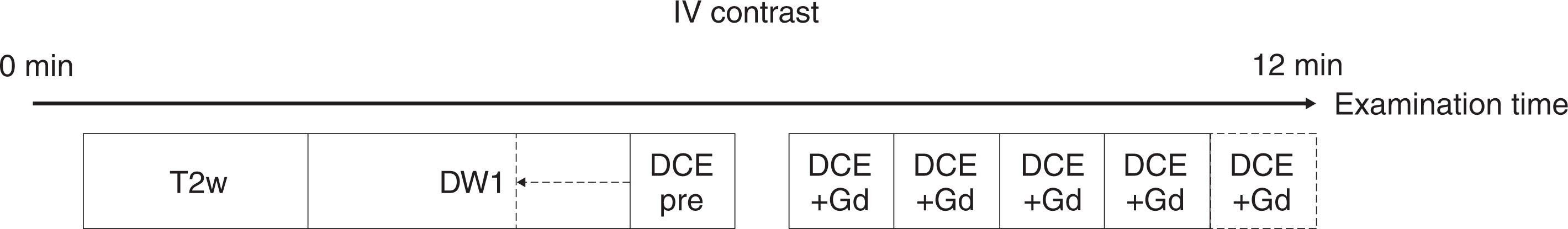
A standard breast MRI protocol comprises T2- and T1-weighted MRI sequences, the latter being repeated as a part of the DCE protocol. DWI usually comes as an added MRI sequence that should ideally be performed before DCE. DWI should be performed in axial orientation using an echo-planar imaging (EPI) sequence. Recommended acquisition parameters are a repetition time (TR) of ≥3000 ms, a minimal echo time (TE), an in-plane resolution ≤2 × 2 mm 2 , a slice thickness ≤4 mm, and application of parallel imaging to reduce image distortion. Fat suppression is commonly incorporated in breast MRI sequences and is mandatory for DWI. There are different fat suppression techniques available, which vary in their robustness and signal-to-noise ratio (SNR). The spectrally adiabatic inversion recovery (SPAIR) technique has been recommended for breast DWI as the best compromise between signal and robustness. T2-weighted acquisitions can be acquired with an acquisition time of 2 to 4 minutes. Although some authors suggested that b = 0 images with submillimeter resolution (using 7 T MRI) might be used as a replacement of T2w images—providing the T2-weighted contrast while saving on acquisition times—currently, low b value images acquired at 1.5 T and 3 T cannot replace T2-weighted TSE images. The total DWI sequence can be acquired within 2 to 4 minutes only if the relevant two b values are acquired. The central part of the breast MRI protocol is a DCE T1-weighted sequence acquired before and several times after contrast (see Chapter 2 ). This sequence is used to identify lesions, and in addition to the enhancement curve type, most other BI-RADS features are obtained from these images. The higher the temporal resolution, the higher the degree of information available from signal intensity curve type analysis, and the higher the spatial resolution, the higher the amount of diagnostic data available from contrast enhancement morphology. The necessary trade-off in clinical practice usually does favor spatial over temporal resolution, though there are multiple acceleration techniques able to alleviate this loss of temporal resolution. There are only loose recommendations regarding how the technique should be acquired, but usually 5 minutes postcontrast scanning suffices to identify wash-out curves and enhancement heterogeneity (see Chapter 2 ). Further techniques, such as MR spectroscopy and T1 or T2 mapping, are currently not recommended in standard protocols due to the lack of consensus regarding their integration in multiparametric protocols, while their incremental diagnostic value is not entirely clear. A full multiparametric breast MRI therefore can take less than 15 minutes in total acquisition time, as shown in Fig. 3.1 .
Analysis of DWI Images
Qualitative Assessment
What diagnostic information does the DWI sequence provide? As malignant lesions show a typical diffusion hindrance, they appear of intermediate to high signal on high b value ( b ≥800 s/mm 2 ) images. Though the appearance of malignant lesions on DWI is highly variable regarding shape, margins, and mass-like features and thus not very specific, these images could be used to potentially detect cancer lesions (see Chapter 6 ). This use of DWI is hampered by the fact that not only malignant lesions show high signal on high b value DWI images: complicated cysts with bloody, putrid, or proteinaceous content all show high to very high signal and may thus impair the ability of the reading physician to identify breast cancer, and some cancers are not visible on DWI images. Therefore cancer detection currently relies on the contrast-enhanced T1w images. DWI can further provide fluid-sensitive T2w contrast on low b value (e.g., b = 0 s/mm 2 ) images, similar to that of the T2w sequence though usually at lower image quality and spatial resolution, which may be helpful to identify cysts, edema, and masses with very high signal intensity that often represent benign fibroadenoma. Finally, DWI provides the ADC map in which diffusivity may be assessed quantitatively. For this purpose, the DWI images are carefully compared with the contrast-enhanced and T2w images in order to localize the lesion.
Quantitative Assessment: Downgrading Suspicious Contrast-Enhancing Lesions By Means of the ADC
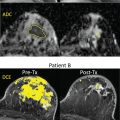
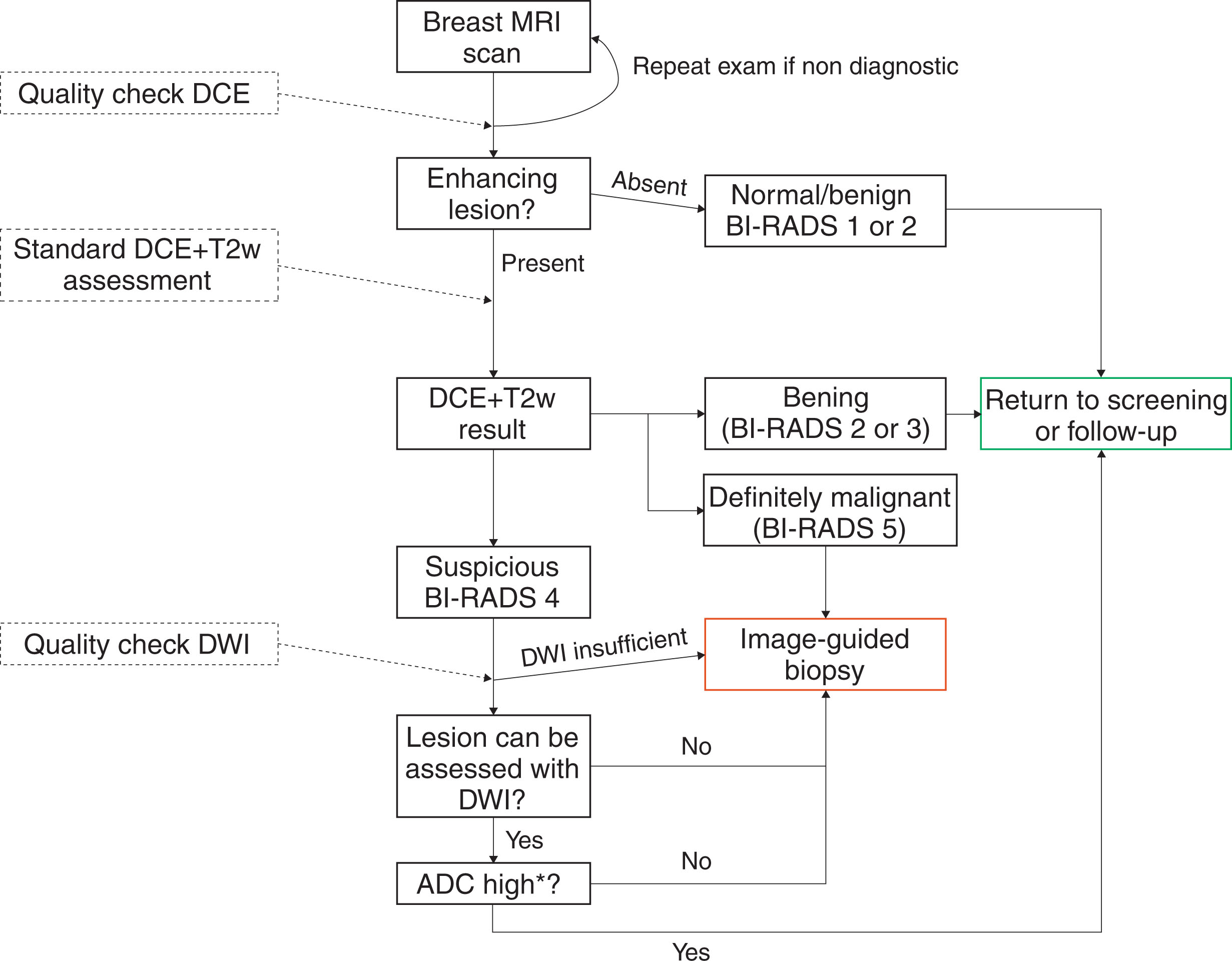
Quantitative assessment of ADC maps requires drawing a region of interest (ROI) around the lesion either directly on the ADC map or copied from DWI images. The positioning of the ROI may greatly influence ADC measurements, so ROIs must be placed with great care to avoid cystic or necrotic areas and rather target the biologically most active parts of the lesions. Malignant lesions show a typical diffusion hindrance reflected by low ADC values, whereas a substantial number of benign lesions present with high ADC values. But how can the ADC be put to use in clinical practice? The dilemma is the same as with other diagnostic features: does the absence of diffusion hindrance overrule a suspicious wash-out curve type? Should a lesion presenting with a low ADC value be biopsied despite benign morphological features and curve type? The ultimate aim in breast imaging to detect as many cancers as possible while keeping the costs as low as possible in terms of false-positive biopsy and follow-up recommendations. As the sensitivity of contrast-enhanced MRI is without question, reducing false-positive findings without compromising sensitivity is what best describes the clinical need. A number of authors have addressed the topic. The most straightforward approach is applying a high ADC cutoff value above which a lesion is automatically downgraded, thereby omitting biopsy. More complicated algorithms adapting the ADC threshold to the specific lesion type and contrast-enhanced MRI BI-RADS category assignment were also successfully developed and tested ( Fig. 3.2 ).

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree