Part 4: Lower Extremity
Case 51

Clinical History
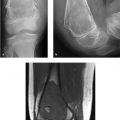
A 39-year-old woman with a history of buttock pain, aggravated by sitting, and “sciatic-like” symptoms (Fig. 51‑1).
Key Finding
Variant sciatic nerve anatomy.
Top 3 Differential Diagnoses
Piriformis syndrome: Piriformis syndrome refers to entrapment of the sciatic nerve at the level of the piriformis muscle. Two general categories are described: primary (i.e., anatomical cause such as sciatic nerve splitting) and secondary (i.e., due to a precipitating case such as a mass or trauma).
Clinical features of piriformis syndrome reported most commonly include tenderness near the greater sciatic notch, buttock pain, pain aggravated on sitting or any maneuver that increases piriformis muscle tension, and limitation in straight leg raising.
On MRI, sciatic nerve variations are relatively common (≤ 20%). Such variant sciatic nerve anatomy is not necessarily associated with piriformis syndrome. Abnormal caliber and T2 hyperintensity in the sciatic nerve may be associated with sciatica. MR neurography and diffusion tensor imaging are increasingly used to evaluate the sciatic nerve.
In patients undergoing cross-sectional imaging for clinically suspected piriformis syndrome, derangements at the level of the piriformis muscle (e.g., tumors) can be identified as important etiologies that drastically alter patient management (e.g., with percutaneous injection, surgery, antibiotics, and radiation therapy).
Incidental anatomic variation: Anatomically, an undivided sciatic nerve most commonly emerges inferior to the piriformis muscle (~80%).
The most common anatomic variation involves the sciatic nerve splitting into two divisions, with one division of the sciatic nerve passing through the piriformis and the other passing inferior to the piriformis. Other variations are also possible (e.g., the sciatic nerve splitting, with one division above and one division below the piriformis).
Double crush syndrome: Multifocal neuropathy, commonly referred to as double crush syndrome, refers to multiple simultaneous nerve insults (e.g., nerve entrapment and diabetes) that results in nerve dysfunction. Double crush syndrome specifically refers to multiple sites of physical compression along a single nerve (e.g., at the level of the piriformis and lumbar spine). Currently, the concept has been expanded to recognize that nonmechanical etiologies (e.g., systemic, pharmacological) may also contribute to neuropathic symptoms. Imaging is most helpful in identifying anatomic sites of nerve entrapment that may be amenable to surgical decompression. In order to avoid suboptimal outcomes after nerve decompression at a single site, successful management focuses on treating all contributing factors (e.g., decompression of both proximal and distal sites of nerve entrapment).
Diagnosis
Piriformis syndrome.
Pearls
Piriformis syndrome may be related to anatomic variations in some cases but, importantly, extraspinal sciatica may be secondary to focal derangements, such as a space-occupying mass.
Anatomic variations in the relationship of the piriformis muscle and sciatic nerve are relatively common, and they may not have a significant association with clinically diagnosed piriformis syndrome.
Double crush syndrome (multifocal neuropathy) occurs when multiple etiologies are responsible for a neuropathy. Recognition of this concept helps physicians avoid “satisfaction of search” errors that can result in suboptimal outcomes.
Suggested Readings
- 135 Bartret AL, Beaulieu CF, Lutz AM. Is it painful to be different? Sciatic nerve anatomical variants on MRI and their relationship to piriformis syndrome.. Eur Radiol 2018; 28 (11) 4681-4686 PubMed 29713768
- 136 Hopayian K, Danielyan A. Four symptoms define the piriformis syndrome: an updated systematic review of its clinical features.. Eur J Orthop Surg Traumatol 2018; 28 (2) 155-164 PubMed 28836092
- 137 Kane PM, Daniels AH, Akelman E. Double crush syndrome.. J Am Acad Orthop Surg 2015; 23 (9) 558-562 PubMed 26306807
- 138 Probst D, Stout A, Hunt D. Piriformis Syndrome: a narrative review of the anatomy, diagnosis, and treatment.. PM R 2019; 11 (Suppl. Suppl 1) S54-S63 PubMed 31102324
- 139 Shah SS, Consuegra JM, Subhawong TK, Urakov TM, Manzano GR. Epidemiology and etiology of secondary piriformis syndrome: A single-institution retrospective study.. J Clin Neurosci 2019; 59: 209-212 PubMed 30528358
- 140 Vassalou EE, Katonis P, Karantanas AH. Piriformis muscle syndrome: a cross-sectional imaging study in 116 patients and evaluation of therapeutic outcome.. Eur Radiol 2018; 28 (2) 447-458 PubMed 28786005
- 141 Wada K, Goto T, Takasago T, Hamada D, Sairyo K. Piriformis muscle syndrome with assessment of sciatic nerve using diffusion tensor imaging and tractography: a case report.. Skeletal Radiol 2017; 46 (10) 1399-1404 PubMed 28616638
Case 52

Clinical History
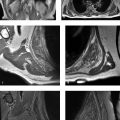
A 21-year-old man with a history of buttock pain associated with elite collegiate athletics (Fig. 52‑1).
Key Finding
Ischiofemoral space narrowing and quadratus femoris muscle edema in a patient with chronic ischial tuberosity avulsion (nonunion).
Top 3 Differential Diagnoses
Ischiofemoral impingement: Ischiofemoral impingement is generally defined by a triad of findings: (1) pain in the hip or buttock region, (2) narrowing of the space between the ischial tuberosity and lesser trochanter, and (3) quadratus muscle abnormality on MRI (edema or atrophy). Ischiofemoral impingement is more common in women, and it may be related to increased ischial and femoral neck angles compared with controls.
Radiographs may show chronic osseous changes associated with narrowing between the ischium and lesser trochanter. Although the ischiofemoral space may change with patient positioning, narrowing to ≤15 mm has an optimal diagnostic sensitivity and specificity (~80%). Imaging also may show associated derangements in the hamstring and iliopsoas tendons.
Management may include image-guided percutaneous treatment (e.g., corticosteroid injection of the quadratus femoris muscle) and, occasionally, surgery (endoscopic or open).
Hamstring disorders: Acute and chronic osseous avulsion injuries at the ischial tuberosity (origin of the hamstring tendons) are commonly observed on radiography, MRI, and CT.
When nonunion or malunion occurs at an ischial avulsion fracture, there is often narrowing of the ischiofemoral space that predisposes patients to ischiofemoral impingement. Other disorders involving the proximal hamstring tendons are also associated with ischiofemoral impingement, including hamstring enthesopathy, tendon tearing, and peritendinitis.
Deep gluteal syndrome: Deep gluteal syndrome is a recently popularized entity that describes the presence of pain in the buttock region and extraspinal entrapment of the sciatic nerve. Disorders involving the deep gluteal space may include a broad array of disorders, including ischiofemoral impingement, piriformis syndrome, obturator internus/gemellus syndrome, and fibrous bands causing sciatic nerve entrapment. Although the term “deep gluteal syndrome” is controversial, proponents suggest it is a unifying concept that should promote improved awareness for diagnosis (e.g., with MR neurography) and treatment (e.g., with percutaneous and endoscopic procedures).
Diagnosis
Ischiofemoral impingement, which is associated with chronic hamstring avulsion injury.
Pearls
Ischiofemoral impingement is a syndrome defined by hip/buttock pain, ischiofemoral space narrowing, and quadratus femoris muscle edema/atrophy.
Hamstring disorders may be primary pain generators, in addition to predisposing patients to ischiofemoral impingement.
Deep gluteal syndrome is an overarching term, encompassing a wide variety of extraspinal disorders that cause sciatica.
Suggested Readings
- 142 Backer MW, Lee KS, Blankenbaker DG, Kijowski R, Keene JS. Correlation of ultrasound-guided corticosteroid injection of the quadratus femoris with MRI findings of ischiofemoral impingement.. AJR Am J Roentgenol 2014; 203 (3) 589-593 PubMed 25148161
- 143 Balius R, Susín A, Morros C, Pujol M, Pérez-Cuenca D, Sala-Blanch X. Gemelli-obturator complex in the deep gluteal space: an anatomic and dynamic study.. Skeletal Radiol 2018; 47 (6) 763-770 PubMed 29218390
- 144 Hernando MF, Cerezal L, Pérez-Carro L, Abascal F, Canga A. Deep gluteal syndrome: anatomy, imaging, and management of sciatic nerve entrapments in the subgluteal space.. Skeletal Radiol 2015; 44 (7) 919-934 PubMed 25739706
- 145 Hernando MF, Cerezal L, Pérez-Carro L, Canga A, González RP. Evaluation and management of ischiofemoral impingement: a pathophysiologic, radiologic, and therapeutic approach to a complex diagnosis.. Skeletal Radiol 2016; 45 (6) 771-787 PubMed 26940209
- 146 Singer AD, Subhawong TK, Jose J, Tresley J, Clifford PD. Ischiofemoral impingement syndrome: a meta-analysis.. Skeletal Radiol 2015; 44 (6) 831-837 PubMed 25672947
- 147 Taneja AK, Bredella MA, Torriani M. Ischiofemoral impingement.. Magn Reson Imaging Clin N Am 2013; 21 (1) 65-73 PubMed 23168183
Case 53
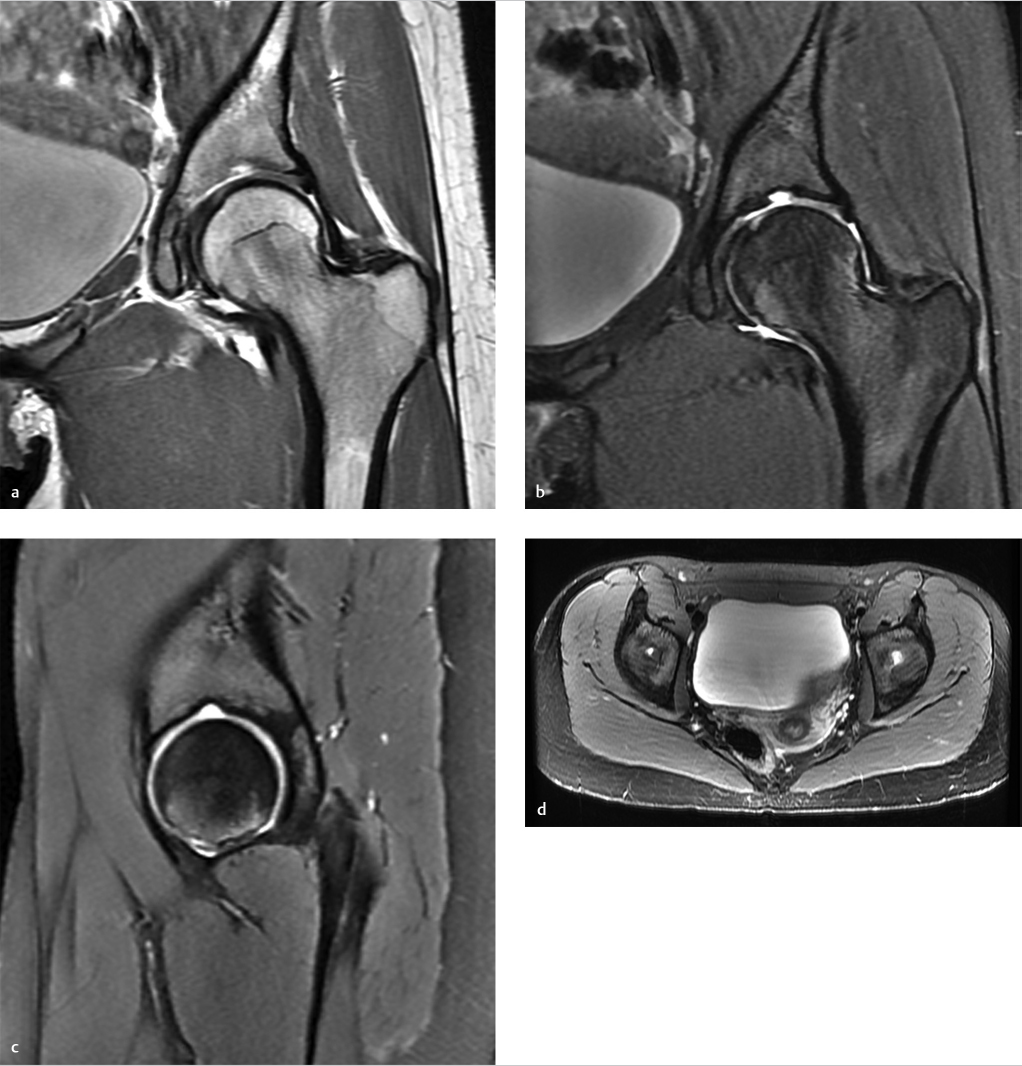
Key Finding
Small smoothly marginated depression (“fossa”) at the mid-acetabular roof.
Top 3 Differential Diagnoses
Supraacetabular fossa: The supraacetabular fossa (pseudodefect of acetabular cartilage) is a focal concavity located in the acetabular roof at the 12 o’clock position (both on coronal and sagittal images). This apparent depression in the articular surface is smoothly marginated and small (generally measures approximately 3 mm in depth and 5 mm in transaxial diameter).
The supraacetabular fossa is considered an incidental anatomic variation. It is observed in approximately 10% of hip MRI examinations (often in adolescents and young adults).
On MRI, the supra acetabular fossa may be filled with articular cartilage or fluid. Unlike a typical chondral/osteochondral defect, there is no adjacent bone edema or loose body.
Superior acetabular notch: The superior acetabular notch is also considered an anatomic variant. This notch is located in the acetabular roof superomedially (superior to the acetabular fossa and medial the chondral surface).
The “superior acetabular notch,” like the “supraacetabular fossa,” is an incidental anatomic variant and is seen in only a minority of patients. These two terms for anatomic variants are easy to confuse with “acetabular fossa” and “acetabular notch,” which are both constant anatomic features. (The “acetabular fossa” is at the medial aspect of the acetabulum; it is occupied by the ligamentum teres and fibrofatty tissue. At the inferior margin of the acetabular fossa, there is an “acetabular notch,” which is the attachment site for the transverse acetabular ligament).
Chondral/osteochondral defect: A chondral or osteochondral lesion must be differentiated from normal anatomic variants in the acetabular roof. Chondral lesions most commonly occur at the anterosuperior or superolateral rim of the acetabulum. In addition to having different locations than anatomic variants, chondral or osteochondral lesions are typically associated with subchondral bone edema/reaction, adjacent chondrosis, or loose bodies.
Diagnosis
Supraacetabular fossa.
Pearls
The supra acetabular fossa and the superior acetabular notch are incidental anatomic variants that occur at specific locations in the roof of the acetabulum.
Anatomic variations in the roof of the acetabulum, especially the supraacetabular fossa, can be mistaken for a chondral/osteochondral defect.
A chondral/osteochondral defect is typically associated with subchondral bone edema, adjacent chondrosis, or loose bodies.
Suggested Readings
- 148 Boutris N, Gardner SL, Yetter TR, Delgado DA, Pulido L, Harris JD. MRI prevalence and characteristics of supraacetabular fossae in patients with hip pain.. Hip Int 2018; 28 (5) 542-547 PubMed 29756502
- 149 Dallich AA, Rath E, Atzmon R et al. Chondral lesions in the hip: a review of relevant anatomy, imaging and treatment modalities.. J Hip Preserv Surg 2019; 6 (1) 3-15 PubMed 31069090
- 150 Dietrich TJ, Suter A, Pfirrmann CW, Dora C, Fucentese SF, Zanetti M. Supraacetabular fossa (pseudodefect of acetabular cartilage): frequency at MR arthrography and comparison of findings at MR arthrography and arthroscopy.. Radiology 2012; 263 (2) 484-491 PubMed 22416250
- 151 Omoumi P, Vande Berg B. Hip Imaging: normal variants and asymptomatic findings.. Semin Musculoskelet Radiol 2017; 21 (5) 507-517 PubMed 29025181
- 152 Sampatchalit S, Chen L, Haghighi P, Trudell D, Resnick DL. Changes in the acetabular fossa of the hip: MR arthrographic findings correlated with anatomic and histologic analysis using cadaveric specimens.. AJR Am J Roentgenol 2009; 193 (2) W127–W133 PubMed 19620414
Case 54

Key Finding
Focal subchondral bone edema in the femoral head.
Top 3 Differential Diagnoses
Subchondral stress fracture: Stress fractures may occur at numerous characteristic locations in the skeleton, including the subchondral bone of the femoral head.
Two types of stress fractures may occur: (1) fatigue fractures and (2) insufficiency fractures. Fatigue-type stress fractures occur in normal bone with chronic repetitive microtrauma (e.g., endurance athletes and military recruits during intensive training). Insufficiency-type stress fractures generally occur when normal stress is exerted on weakened bones. In the femoral head, subchondral insufficiency fractures are diagnosed most commonly in elderly female patients with osteopenia and acute onset severe hip pain. There is an association with insufficiency fractures elsewhere (e.g., vertebral body compression fracture).
Both types of femoral head subchondral fractures cause hip pain, are relatively small, and are subtle (or occult) with radiography. Both types of fractures can benefit from early diagnosis with a high-spatial resolution MRI, which enables appropriate early management (e.g., non-weight bearing) and improves the likelihood of preventing progression to subchondral collapse, secondary osteonecrosis, and early onset osteoarthritis.
At MRI, both subchondral stress fractures and avascular necrosis (AVN) have some overlapping features that can make evaluation difficult, including potentially a “crescent sign,” a low-signal intensity band of demarcation (corresponding to osteosclerosis on CT), and adjacent bone edema.
Two MRI findings can help differentiate a femoral head subchondral stress fracture versus AVN. With a subchondral stress fracture, there is typically (1) a serpentine, hypointense fracture line that parallels the articular surface and (2) hyperintense signal between the fracture line and the subchondral bone plate on fat-saturated T2 and postcontrast images (in other words, bone edema and enhancement occur on both sides of the fracture line).
By comparison, with AVN, there is often (1) a continuous, rounded hypointense line that is concave superiorly, and (2) hypointense, necrotic bone completely fills the zone between the hypointense line and the subchondral bone on fat-saturated T2 and postcontrast images (in other words, bone edema and enhancement occur only outside the necrotic bone).
With follow-up MRI of subchondral stress fractures, improvement is characterized by fracture healing and decreased bone edema, while worsening is characterized by progression to secondary AVN with femoral head collapse.
Osteonecrosis: With femoral head AVN, bone marrow edema is associated with hip pain and radiographically-occult subchondral fracture/femoral head collapse (the last case discusses femoral head AVN in detail).
Reactive subchondral bone edema: A bone marrow edema pattern on MRI is a nonspecific finding seen in association with a wide spectrum of pathological conditions, including posttraumatic contusions, subchondral fractures, AVN, osteochondral lesions, and articular cartilage degeneration (histologically, true edema may not be the predominant finding in the bone marrow, and therefore some purists prefer terms such as “bone marrow edema-like pattern” or “bone marrow lesion”).
The term “reactive bone edema” is sometimes used to indicate that the edema-like pattern of signal in the subchondral bone is secondary to a process such as overlying chondrosis. With MRI, articular cartilage degeneration in the hip may be difficult to visualize, but reactive bone edema and degenerative subchondral cysts are often recognizable.
Notably, the term “idiopathic transient bone marrow edema syndrome” has largely fallen out of favor, because clinical correlation or high-resolution MRI often reveal an underlying etiology for the bone edema pattern, such as stress reaction, stress fracture, or overlying chondrosis.
Diagnosis
Reactive subchondral bone edema.
Pearls
Focal signal alteration in the subchondral bone of the femoral head is not always due to AVN.
Subchondral fracture and AVN can occur at similar anatomical locations, but they often have different imaging appearances and different treatment algorithms.
Reactive subchondral bone edema and subchondral cysts are commonly associated with overlying chondrosis in the hip.
Suggested Readings
- 153 Ando W, Yamamoto K, Koyama T, Hashimoto Y, Tsujimoto T, Ohzono K. Radiologic and clinical features of misdiagnosed idiopathic osteonecrosis of the femoral head.. Orthopedics 2017; 40 (1) e117-e123 PubMed 27755641
- 154 Gonzalez-Espino P, Van Cauter M, Gossing L, Galant CC, Acid S, Lecouvet FE. Uncommon observation of bifocal giant subchondral cysts in the hip: diagnostic role of CT arthrography and MRI, with pathological correlation.. Skeletal Radiol 2018; 47 (4) 587-592 PubMed 29164284
- 155 Hatanaka H, Motomura G, Ikemura S et al. Differences in magnetic resonance findings between symptomatic and asymptomatic pre-collapse osteonecrosis of the femoral head.. Eur J Radiol 2019; 112: 1-6 PubMed 30777197
- 156 Kim SM, Oh SM, Cho CH et al. Fate of subchondral fatigue fractures of femoral head in young adults differs from general outcome of fracture healing.. Injury 2016; 47 (12) 2789-2794 PubMed 27771040
- 157 Lee S, Saifuddin A. Magnetic resonance imaging of subchondral insufficiency fractures of the lower limb.. Skeletal Radiol 2019; 48 (7) 1011-1021 PubMed 30706108
- 158 Leydet-Quilici H, Le Corroller T, Bouvier C et al. Advanced hip osteoarthritis: magnetic resonance imaging aspects and histopathology correlations.. Osteoarthritis Cartilage 2010; 18 (11) 1429-1435 PubMed 20727415
Case 55

Clinical History
A 64-year-old athletic man with persistent pain at the lateral aspect of the hip, presenting for an MRI to “rule out stress fracture, trochanteric bursitis, and abductor tendon tear” (Fig. 55‑1).
Key Finding
Low-grade tearing of the iliotibial band (ITB), with soft-tissue edema along the fascia.
Top 3 Differential Diagnoses
Proximal iliotibial band syndrome: Anatomically, the ITB is a longitudinally-oriented band of fascial tissue that is best seen on coronal images originating from an origin at the iliac tubercle (at the superior–lateral lip of the iliac crest, approximately 5 cm posterior to the anterior superior iliac spine). The ITB extends distally along the superficial aspect of the gluteus medius/minimus and greater trochanter. The ITB continues distally beyond the hip region along the entire lateral thigh and knee, inserting distally at the anterolateral tibia (Gerdy’s tubercle).
Distally, ITB syndrome at the lateral aspect of the knee is a well-known overuse injury in athletes such as runners and cyclists. However, a proximal ITB syndrome is increasingly recognized as an important cause of pain at the lateral aspect of the hip, which can be associated with repetitive use/microtrauma and low-grade inflammation. Proximal ITB syndrome may occur as an overuse injury in athletes as well as in older (predominantly female) nonathletes.
On MRI, proximal ITB syndrome is characterized by the presence of soft-tissue edema along the ITB, typically at the proximal attachment or more distally at the lateral aspect of the hip (without other causes of edema such as contusion). At the iliac tubercle enthesis, edema may be seen in the bone marrow and overlying soft tissue. Regardless of the location in the hip region, soft-tissue edema may be accompanied by subtle thickening or partial tearing.
Greater trochanteric pain syndrome: With the given clinical history, the differential diagnosis could include numerous potential pain generators located at the lateral aspect of the hip.
Regardless of the pain generator, the accuracy of MRI and sonography is enhanced by detailed anatomic knowledge and high-resolution imaging. Causes of greater trochanteric pain syndrome may or may not be well-demonstrated with imaging, and therefore some humility on the part of imagers is appropriate.
The term “greater trochanteric pain syndrome” is important to recognize, however, because it has largely replaced the clinical diagnosis of “trochanteric bursitis.” There is now a more nuanced understanding of pain at the lateral aspect of the hip. In addition to bursitis, pain generators are now known to commonly involve one or more other sites. Examples of conditions include abductor tendinopathy (with or without tears), abductor peritendinitis, calcific tendinitis, and snapping hip (external coxa saltans). Proximal ITB syndrome could also be included under this umbrella term.
Greater trochanteric pain syndrome is often diagnosed in middle-aged women, but it is also reported in other demographic groups such as athletes and patients after hip arthroplasty.
Treatment of greater trochanteric pain syndrome varies with the source, severity, and duration of symptoms. In most cases, nonoperative management suffices (e.g., physical therapy, percutaneous injection). In a minority of cases failing conservative management, surgical interventions may be performed (e.g., tendon repair, tendon release, and bursectomy).
Referred pain: With the given clinical history, the clinical differential diagnosis includes pain referred to the lateral aspect of the hip. For many of these diagnoses, radiologists are extremely helpful with imaging and/or image-guided injections, including the diagnosis of hip osteoarthritis, stress fracture, myopathy/myositis, sacroiliitis, and lumbar spine degeneration.
Diagnosis
Proximal ITB syndrome.
Pearls
The term “greater trochanteric pain syndrome” is a clinical diagnosis. In addition to trochanteric bursitis, there are numerous other potential pain generators located at the lateral aspect of the hip.
Proximal ITB syndrome refers to low-grade edema/inflammation involving the ITB at the ischial enthesis or at the lateral aspect of the hip, often related to overuse (degenerative or athletic). Partial tearing of the ITB may or may not be present.
Proximal ITB syndrome is easily overlooked if one is not aware of the characteristic appearance on MRI.
Suggested Readings
- 159 Flato R, Passanante GJ, Skalski MR, Patel DB, White EA, Matcuk Jr GR. The iliotibial tract: imaging, anatomy, injuries, and other pathology.. Skeletal Radiol 2017; 46 (5) 605-622 PubMed 28238018
- 160 Hirschmann A, Falkowski AL, Kovacs B. Greater trochanteric pain syndrome: abductors, external rotators.. Semin Musculoskelet Radiol 2017; 21 (5) 539-546 PubMed 29025184
- 161 Huang BK, Campos JC, Michael Peschka PG et al. Injury of the gluteal aponeurotic fascia and proximal iliotibial band: anatomy, pathologic conditions, and MR imaging.. Radiographics 2013; 33 (5) 1437-1452 PubMed 24025934
- 162 Khoury AN, Brooke K, Helal A et al. Proximal iliotibial band thickness as a cause for recalcitrant greater trochanteric pain syndrome.. J Hip Preserv Surg 2018; 5 (3) 296-300 PubMed 30393557
- 163 Redmond JM, Chen AW, Domb BG. Greater trochanteric pain syndrome.. J Am Acad Orthop Surg 2016; 24 (4) 231-240 PubMed 26990713
- 164 Sher I, Umans H, Downie SA, Tobin K, Arora R, Olson TR. Proximal iliotibial band syndrome: what is it and where is it?. Skeletal Radiol 2011; 40 (12) 1553-1556 PubMed 21499978
Case 56

Clinical History
A 12-year-old athletic girl with a history of knee pain and negative radiographs (Fig. 56‑1).
Key Finding
Focal periphyseal edema.
Top 3 Differential Diagnoses
Focal periphyseal edema (FOPE) zone: Focal periphyseal edema (FOPE) refers to a focal zone of bone edema centered around a physis. It can be seen in adolescents with pain in the early stages of physiologic physeal closure.
FOPE zones are most commonly observed at the knee, but can occur elsewhere (e.g., in the hip at the greater trochanter apophysis). In the knee, FOPE zones generally occur in patients with bone ages ranging from 11 to 14 years. FOPE zones are characteristically seen in an eccentric central location at sites of early physeal closure and resolve within one year. FOPE zones are managed conservatively and do not need follow-up imaging.
Physeal stress reaction (repetitive microtrauma): Children and adolescents participating in organized sports are vulnerable to stress injury. In addition to bone edema, physeal widening and reactive sclerosis may occur.
Although stress injury is usually reversible with conservative management, there is occasionally, premature bone bridging across the physis and subsequent deformity.
Contusion/fracture: Injuries can involve the physis, such as a contusion or fracture. Imaging findings of physeal injury can include physeal widening, bone edema, fracture lines, and periosteal injury (elevation or disruption).
At the knee, the most common physeal fracture pattern targets the lateral distal femoral metaphysis, with fracture extension through the medial physis (Salter–Harris type II fracture). Subsequent premature physeal fusion with leg length inequality may then occur.
Diagnosis
Focal periphyseal edema (FOPE) zone.
Pearls
FOPE zones are observed on MRI examinations of adolescents during early physiologic physeal closure.
On knee MRI, a FOPE zone is generally seen as a patchy focus of bone edema, abutting the proximal and distal margins of the physis; this bone edema resolves in less than 1 year with conservative management.
Bone edema on MRI is a nonspecific finding and may be caused by stress reaction or fracture, depending on the clinical context.
Suggested Readings
- 165 Giles E, Nicholson A, Sharkey MS, Carter CW. Focal periphyseal edema: are we overtreating physiologic adolescent knee pain?. J Am Acad Orthop Surg Glob Res Rev 2018; 2 (4) e047 PubMed 30211386
- 166 Jaimes C, Jimenez M, Shabshin N, Laor T, Jaramillo D. Taking the stress out of evaluating stress injuries in children.. Radiographics 2012; 32 (2) 537-555 PubMed 22411948
- 167 Leschied JR, Udager KG. Imaging of the pediatric knee.. Semin Musculoskelet Radiol 2017; 21 (2) 137-146 PubMed 28355677
- 168 Sakamoto A, Matsuda S. Focal periphyseal edema zone on magnetic resonance imaging in the greater trochanter apophysis: a case report.. J Orthop Case Rep 2017; 7 (4) 29-31 PubMed 29181348
- 169 Ueyama H, Kitano T, Nakagawa K, Aono M. Clinical experiences of focal periphyseal edema zones in adolescent knees: case reports.. J Pediatr Orthop B 2018; 27 (1) 26-30 PubMed 27606713
- 170 Zbojniewicz AM, Laor T. Focal periphyseal edema (FOPE) zone on MRI of the adolescent knee: a potentially painful manifestation of physiologic physeal fusion?. AJR Am J Roentgenol 2011; 197 (4) 998-1004 PubMed 21940591
Case 57

Key Finding
Curvilinear signal separating the patella into two fragments.
Top 3 Differential Diagnoses
Bipartite patella:The patella begins to ossify at 3 to 5 years of age, with secondary ossification centers that develop and usually fuse during adolescence. When one (or more) of these ossification centers fail to fuse, a bipartite (or multipartite) patella occurs. The prevalence is approximately 1 to 2% (9 times more common in males than females; bilateral in >25 %).
A bipartite patella may be discovered as an asymptomatic incidental finding, or it may be associated with symptoms. With MRI of an asymptomatic bipartite patella, bone edema is not present. Conversely, in most symptomatic patients, bone edema abuts the interface (fibrocartilaginous synchondrosis or pseudarthrosis) between the fragment and the adjacent patella.
With imaging, the bipartite patella is seen as a well-corticated fragment that is located at the superolateral patella in the vast majority of cases (i.e., insertion site for the vastus lateralis and lateral retinaculum in ≥75% of cases). Less commonly, a bipartite patella has a longitudinal orientation along the lateral patellar facet. A small fragment at the distal margin was originally described as a “type I” bipartite patella, but many investigators now categorize this finding as Sinding–Larsen–Johansson (SLJ) syndrome
Imaging features that favor a bipartite patella (rather than fracture) include bilateral distribution, superolateral location, curvilinear orientation, and smooth, sclerotic, corticated margins.
Patellar fracture: Fractures of the patella are commonly due to a traumatic event involving a direct blow or excessive tension through the extensor mechanism. Osteochondral fractures are commonly seen after a transient lateral patellar dislocation.
Other patellar fractures are often described by their orientation as transverse (~80% occur in the mid to lower third of the patella, typically from indirect trauma), vertical (~20% of patellar fractures), or stellate (comminuted, typically from direct trauma).
Patellar stress fractures are rare, but they may occur in athletes (runners, jumpers, and kickers). These fractures are typically located either in the distal third of the patella (transversely) or in the lateral patella facet (longitudinally).
Patellar calcar: A calcar is a normal condensation of bone trabeculae. In the subchondral bone of the lateral patellar facet, a calcar can be observed in most knees, particularly in adolescents and young adults (~80%).
The patellar calcar is displayed as curvilinear low-signal with anterior convexity, which is generally best seen on two to three contiguous, nonfat-suppressed images. The patellar calcar is often inconspicuous and is not normally associated with any bone edema, but it rarely may mimic a patellar stress fracture or an osteochondral lesion.
Diagnosis
Bipartite patella.
Pearls
A bipartite patella is a developmental anomaly caused by failed fusion of an accessory ossification center with the remainder of the patella, usually superolaterally.
A bipartite patella is commonly an incidental finding; these patients do not have bone edema along the synchondrosis between fragments. In most symptomatic patients with a bipartite patella, MRI shows bone edema.
In the lateral patellar facet, a patellar calcar is a normal incidental finding seen as curvilinear low signal, without bone edema. The patellar calcar should not be confused with a fracture or osteochondral lesion.
Suggested Readings
- 171 Brown GA, Stringer MR, Arendt EA. Stress fractures of the patella. In: Miller TL, Kaeding CC (eds). Stress Fractures in Athletes. Cham, Switzerland: Springer; 2015:125–135
- 172 Collins MS, Tiegs-Heiden CA, Stuart MJ. Patellar calcar: MRI appearance of a previously undescribed anatomical entity.. Skeletal Radiol 2014; 43 (2) 219-225 PubMed 24287959
- 173 Jarraya M, Diaz LE, Arndt WF, Roemer FW, Guermazi A. Imaging of patellar fractures.. Insights Imaging 2017; 8 (1) 49-57 PubMed 27905071
- 174 Kavanagh EC, Zoga A, Omar I, Ford S, Schweitzer M, Eustace S. MRI findings in bipartite patella.. Skeletal Radiol 2007; 36 (3) 209-214 PubMed 17151849
- 175 O’Brien J, Murphy C, Halpenny D, McNeill G, Torreggiani WC. Magnetic resonance imaging features of asymptomatic bipartite patella.. Eur J Radiol 2011; 78 (3) 425-429 PubMed 19954912
- 176 Oohashi Y. Developmental anomaly of ossification type patella partita.. Knee Surg Sports Traumatol Arthrosc 2015; 23 (4) 1071-1076 PubMed 24531356
Case 58

Clinical History
A 62-year-old woman with a history of chronic knee osteoarthritis and “swelling” (Fig. 58‑1).
Key Finding
Frond-like lobules of fatty tissue in the suprapatellar recess, with effusion.
Top 3 Differential Diagnoses
Lipoma arborescens: Lipoma arborescens is a non-neoplastic lipomatous proliferation of the subsynovial connective tissue. Although an idiopathic (primary) type is described in younger patients, the vast majority of cases are in older patients, secondary to chronic synovial irritation in the setting of osteoarthritis or (much less commonly) rheumatoid arthritis. Affected joints have a chronic or recurring effusion.
Lipoma arborescens is generally a monoarticular process. The most common location for lipoma arborescens is the knee, particularly in the suprapatellar recess. With knee MRI, the frequency of this finding is approximately 0.3%. Lipoma arborescens may also be seen in other synovial-lined locations (e.g., glenohumeral joint in the setting of chronic osteoarthritis). More rarely, bursae or tendon sheaths may be affected.
Diagnosis can be made on MRI examinations showing hypertrophic lipomatous tissue with a lobular or villous morphology projecting into an effusion. Unlike pigmented villonodular synovitis (PVNS), there is no low-T2 signal or susceptibility artifact to suggest hemosiderin.
Arthroscopic synovectomy is an accepted treatment to address the proliferative process itself, although management of any underlying cause (e.g., osteoarthritis) is also an important consideration.
Intraarticular (loose) bodies: Multiple intraarticular (loose) bodies are associated with internal derangements of joints, most commonly osteoarthritis. These fragments vary in signal intensity, depending on whether they are cartilaginous (chondral), osteochondral, or osseous.
Fragments containing fatty marrow, such as osseous loose bodies, are commonly seen in joints with substantial osteoarthritis. This condition is sometimes referred to as secondary osteochondromatosis to differentiate it from primary synovial chondromatosis.
Osseous loose bodies contain fatty signal (like lipoma arborescens), but typically have a thin sclerotic rim and are not confined to the suprapatellar recess. Unlike lipoma arborescens, osseous loose bodies are mineralized (radio dense) on radiography and CT.
Rice bodies: Rice bodies are fibrinoid fragments that grossly resemble grains of rice. They are thought to occur when hypertrophic synovium undergoes fibrinoid necrosis and is shed into a joint, bursa, or tendon sheath.
Rice body formation is typically seen in patients with chronic synovitis which is caused by inflammatory arthritis (e.g., rheumatoid arthritis, juvenile chronic arthritis) or mycobacterial infection (e.g., tuberculosis arthritis).
On MRI, rice bodies are recognized as numerous, very small fragments of uniform caliber outlined by synovial fluid (not fixed to the synovium).
Diagnosis
Lipoma arborescens.
Pearls
Lipoma arborescens is displayed as synovial-based hypertrophic lipomatous tissue projecting into an effusion.
Lipoma arborescens is typically seen in the suprapatellar recess of older patients with knee osteoarthritis.
Both lipoma arborescens and osseous loose bodies can be seen as foci of high-T1 signal intensity in arthritic joints.
Suggested Readings
- 177 Coll JP, Ragsdale BD, Chow B, Daughters TC. Best cases from the AFIP: lipoma arborescens of the knees in a patient with rheumatoid arthritis.. Radiographics 2011; 31 (2) 333-337 PubMed 21415182
- 178 Evenski AJ, Stensby JD, Rosas S, Emory CL. Diagnostic imaging and management of common intra-articular and peri-articular soft tissue tumors and tumor like conditions of the knee.. J Knee Surg 2019; 32 (4) 322-330 PubMed 30449023
- 179 Susa M, Horiuchi K. Rice bodies in a patient with oligoarticular juvenile idiopathic arthritis.. J Rheumatol 2019; 46 (9) 1157-1158 PubMed 31474614
- 180 Wang CK, Alfayez S, Marwan Y, Martineau PA, Burman M. Knee arthroscopy for the treatment of lipoma arborescens: a systematic review of the literature.. JBJS Rev 2019; 7 (4) e8 PubMed 31021893
Case 59

Clinical History
A 70-year-old man with a palpable mass and pain at the medial aspect of the knee (Fig. 59‑1).
Key Finding
Cystic-appearing lesion centered inferior to the medial joint line of the knee.
Top 3 Differential Diagnoses
Bursitis: Bursae are sac-like structures lined by synovial tissue which function to mitigate friction in the musculoskeletal system. When inflamed, these bursae characteristically fill with fluid, which is considered the sine qua non of bursitis. Classic symptoms can include pain, swelling, and tenderness.
Bursitis at the medial aspect of the knee is associated most commonly with osteoarthritis and overuse (e.g., in runners). Fluid may collect in three bursae at the medial aspect of the knee (in order of frequency).
The semimembranosus–tibial collateral ligament bursa is located near the posteromedial joint line, typically along the superficial aspect of the semimembranosus tendon.
The pes anserinus bursa is located between the medial tibial plateau region and the pes anserine tendons (i.e., sartorius, gracilis, and semitendinosus). With imaging, there is a rounded fluid collection with well-defined margins located inferior to the joint line along the medial aspect of the tibia, superficial to the medial collateral ligament. These fluid collections do not communicate with the knee joint or with popliteal bursal fluid collections. However, in rare cases of pes anserine bursitis (< 2%), there are underlying bone changes (e.g., cortical scalloping, subcortical sclerosis, reactive bone marrow edema, and even intramedullary extension at the medial tibial surface), which may mimic a neoplasm.
The medial collateral ligament (MCL) bursa is located between the superficial and deep MCL fibers (at the central third of the medial knee). Fluid can collect proximal to the joint line, distal to the joint line, or both.
Parameniscal cyst: Parameniscal cysts are well-defined, rounded collections that are typically in continuity with a horizontal or complex tear of the subjacent meniscus.
Almost all patients going to arthroscopy with parameniscal cysts have meniscus tears at surgery. An important exception to this “rule” is observed at the anterior and anterolateral regions of the lateral meniscus; underlying meniscus tears are not present in approximately one-third of patients with cysts at the anterolateral joint line.
Synovial sarcoma: Solid lesions, such as synovial sarcoma, may have a high-water content and mimic fluid collections on noncontrast MRI. Synovial sarcoma can appear relatively homogeneous when small, but larger lesions usually are seen as heterogeneously hyperintense on T2-weighted images that undergo diffuse heterogeneous contrast enhancement. Dystrophic calcification is present in approximately one-third of synovial sarcomas. Underlying bone erosion is rarely seen. Unlike a periarticular cyst, there is no pedicle connecting to the joint.
Diagnosis
Pes anserinus bursitis.
Pearls
Bursitis at the medial aspect of the knee occurs at characteristic anatomic locations, typically involving one of three bursae: the semimembranosus–tibial collateral ligament bursa, pes anserinus bursa, or MCL bursa.
Parameniscal cysts are very highly associated with underlying meniscus tears, except at the anterolateral joint line.
Synovial sarcoma may mimic a fluid collection on noncontrast MRI. However, synovial sarcoma typically does not occur in the anatomic distribution of bursa and is not associated with an underlying meniscus tear.
Suggested Readings
- 181 Colak C, Ilaslan H, Sundaram M. Bony changes of the tibia secondary to pes anserine bursitis mimicking neoplasm.. Skeletal Radiol 2019; 48 (11) 1795-1801 PubMed 31093712
- 182 Curtis BR, Huang BK, Pathria MN, Resnick DL, S , mitaman E. Pes anserinus: anatomy and pathology of native and harvested tendons.. AJR Am J Roentgenol 2019; 213 (5) 1107-1116 PubMed 31361527
- 183 De Maeseneer M, Shahabpour M, Van Roy F et al. MR imaging of the medial collateral ligament bursa: findings in patients and anatomic data derived from cadavers.. AJR Am J Roentgenol 2001; 177 (4) 911-917 PubMed 11566705
- 184 De Smet AA, Graf BK, del Rio AM. Association of parameniscal cysts with underlying meniscal tears as identified on MRI and arthroscopy.. AJR Am J Roentgenol 2011; 196 (2) W180–6 PubMed 21257860
- 185 Rennie WJ, Saifuddin A. Pes anserine bursitis: incidence in symptomatic knees and clinical presentation.. Skeletal Radiol 2005; 34 (7) 395-398 PubMed 15940489
- 186 Steinbach LS, Stevens KJ. Imaging of cysts and bursae about the knee.. Radiol Clin North Am 2013; 51 (3) 433-454 PubMed 23622093
Case 60

Clinical History
A 38-year-old female with recurrent posterior ankle pain after exercising (Fig. 60‑1).
Key Finding
Accessory muscle around ankle.
Top 3 Differential Diagnoses
Accessory soleus: Accessory soleus muscle is typically asymptomatic but can manifest as a soft-tissue mass, and hypertrophy may cause compression of the posterior tibialnerve. The accessory soleus is more common in males. Radiographs reveal obscuration of the normal triangular appearance of Kager fat pad by a well-defined area of increased soft-tissue density. Cross-sectional imaging is more sensitive and specific for identification. MR imaging demonstrates the accessory muscle anterior to the Achilles tendon and superficial to the flexor retinaculum, typically extending medially to the area between the medial edge of the Achilles tendon and medial malleolus. Five types of accessory soleus have been described based on the insertion characteristics. The insertion points include the Achilles tendon, the superior surface of the calcaneus either with a fleshy muscular insertion or a tendinous insertion, and the medial surface of the calcaneus either with a fleshy muscular insertion or a tendinous insertion.
Peroneus quartus: Peroneus quartus is commonly encountered as an asymptomatic variant but may cause lateral ankle pain or ankle instability in athletes. Peroneus quartus extends medial and posterior to the other peroneal tendons and has a variety of insertion sites, the most common insertion is into the calcaneus. MR imaging delineates the muscle optimally. On axial MR images, a peroneus quartus is visualized posteromedial or medial to the peroneus brevis and is separated from it by a fat plane. The muscle belly of peroneus quartus may vary in size. It can be associated with longitudinal tears of the peroneal tendons secondary to crowding of the peroneal tendons deep to the peroneal retinaculum.
Flexor digitorum accessorius longus: The accessory flexor digitorum originates either from the medial margin of the tibia or from the lateral margin of the fibula, and the origin can vary widely, arising from any structure in the posterior compartment. The tendon descends posterior and superficial to the tibial nerve, courses beneath the flexor retinaculum through the tarsal tunnel, and is intimately related to the posterior tibial artery and tibial nerve. It can be associated with tarsal tunnel syndrome because of its close relationship to the neurovascular bundle in the tarsal tunnel. On axial MR images, the muscle is demonstrated within the tarsal tunnel, typically superficial to the neurovascular bundle.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree