- •
Clinical manifestations of CS include heart block, ventricular arrythmias, SCD, and HF.
- •
Among patients with biopsy-proven or a clinical diagnosis of extracardiac sarcoidosis, any signs or symptoms of cardiac involvement should prompt evaluation with advanced imaging with CMR and/or FDG PET.
- •
Imaging for suspected CS is also indicated in patients who do not have a prior history of extracardiac sarcoidosis if there is a history of unexplained atrioventricular block (second-degree Mobitz type II or third degree) in individuals who are under the age of 60 or who sustained monomorphic VT in the absence of any known etiology.
- •
Approximately 25% of CS cases may be isolated to the heart and do not involve other organs.
- •
Diagnosis of CS is challenging and there is no single reliable reference standard. The likelihood of CS (e.g., possible [<50%], probable [>50%], highly probable [>90%]) can be estimated by integrating clinical data and advanced imaging findings.
- •
Echocardiography has a limited sensitivity to detect CS. Thus, a normal echocardiogram should not be used to exclude the diagnosis of CS.
- •
CMR and cardiac PET are the most useful advanced imaging tests used to evaluate patients with suspected CS. Data from both of these tests are often complementary and can aid in diagnosis, prognosis, and patient management.
- •
Among patients with suspected CS, resting perfusion defects on nuclear imaging and focal areas of increased FDG uptake on PET are associated with a higher rate of VT or death.
- •
Among patients with suspected CS, the absence of late gadolinium enhancement on MRI is associated with a low likelihood of CS and an excellent prognosis.
- •
In addition to dedicated cardiac PET images, a limited whole-body FDG PET scan provides an assessment of extracardiac involvement and identifies potential biopsy sites, which may be easier to approach than endomyocardial biopsy.
- •
FDG uptake on cardiac PET is not specific to CS. The differential diagnosis includes some forms of myocarditis, hibernating myocardium, artifacts related to incomplete suppression of FDG from normal myocardium, and other forms of inflammatory cardiomyopathy (see Table 23.9 ).
TABLE 23.9
Differential Diagnosis for Fluorodeoxyglucose Uptake on Positron Emission Tomography Imaging
Category
Example
Artifact/physiologic uptake
- •
Inability to suppress fluorodeoxyglucose from normal myocardium
- •
Severe end-stage cardiomyopathy
Other cardiac pathologies
- •
Hibernating myocardium
- •
Arrhythmogenic cardiomyopathy
Myocarditis
- •
Bacterial, giant cell, tuberculosis
Other inflammatory cardiomyopathies
- •
Related to systemic rheumatologic disease
- •
Antigen-mediated response (environmental or drug related)
Genetic/familial
- •
Lamin cardiomyopathy
- •
PET imaging can be useful for quantifying the intensity and amount of myocardial inflammation and for monitoring the response to therapy.
- •
Additional nuclear imaging tracers are needed to improve the diagnostic accuracy of CS. Ideally, such tracers would not have any uptake by the heart under normal conditions and would be specific to sarcoidosis or to the presence of myocardial inflammation.
Introduction
Sarcoidosis is a multisystem granulomatous and inflammatory disorder that most commonly affects the lungs or its associated lymph nodes but can involve any organ. Although genetic, environmental, occupational, and infectious factors have all been implicated, the exact etiology of sarcoidosis remains unknown. Noncaseating granulomas characterize sarcoidosis and consist of aggregates of epithelioid histiocytes with inflammation and large multinucleated giant cells. The severity of disease is not proportional to the number of granulomas because advanced cases develop a fibrotic reaction that may result in permanent organ dysfunction. The true prevalence of sarcoidosis remains unknown but is likely underestimated; a 2016 prospective cohort study estimated a baseline prevalence of 100 in 100,000 women between the ages of 25 and 44 years old. Other data suggest a higher prevalence in certain parts of the world, particularly in Scandinavia, Ireland, and Japan, and in African Americans, suggesting an important interaction between genetic and environmental factors influencing the risk for developing sarcoidosis. Without cardiac involvement, sarcoidosis is generally associated with a low mortality rate, although the clinical course and prognosis are highly variable and dependent on age, disease severity at presentation, and major organ involvement.
Prevalence and prognosis of cardiac sarcoidosis
Prior data have estimated that 5% of patients with sarcoidosis have clinically manifest cardiac involvement. Autopsy and imaging series, however, report a substantially higher occurrence, ranging from more than 20% in the United States to more than 50% in Japan. Based on imaging studies, 20% to 25% of patients with sarcoidosis have cardiac involvement. The rate of isolated cardiac involvement among patients with cardiac sarcoidosis (CS) is approximately 25%. Cardiac involvement accounts for 10% to 25% of all deaths from sarcoidosis in the United States and more than half of all deaths from sarcoidosis in Japan, where studies have identified the extent of left ventricular (LV) dysfunction as an independent predictor of mortality. Notably, among patients with CS, the most common mode of death is sudden cardiac death (SCD).
Clinical manifestations and presentation of cardiac sarcoidosis
Depending on the type and extent of involvement, the presentation of CS is highly variable ( Table 23.1 ), ranging from asymptomatic heart involvement to sudden death as the initial presentation. CS most commonly involves the myocardium, particularly the basal ventricular septum, followed by the LV free wall, papillary muscles, and right ventricle (RV). Conduction abnormalities are common because of the predilection for granulomas along the conduction system. Although complete heart block in a young patient should raise suspicion for CS, other causes of such a presentation, including Lyme disease and congenital heart block, should be excluded. Acute sarcoid myocarditis, a rare presentation of CS characterized by high-grade atrioventricular block, malignant ventricular arrhythmias, and congestive heart failure (HF), may be indistinguishable clinically from giant cell myocarditis. Frequent ventricular arrhythmias may mimic arrhythmogenic RV cardiomyopathy, classic outflow tract or fascicular ventricular tachycardia (VT), or VT secondary to coronary artery disease (CAD). Establishing the diagnosis is essential given the distinct management of the different conditions. Atrial arrhythmias are increasingly recognized in CS, but small autopsy series previously suggested that CS may involve the atria less extensively than ventricles. Systolic and diastolic HF may also occur because of myocardial changes caused by granulomatous deposition and subsequent fibrosis, which typically occurs in a noncoronary distribution but may be indistinguishable from other infiltrative cardiomyopathies. Valvular disorders have also been reported as a result of papillary muscle involvement. Pericardial effusion, with or without hemodynamic instability, may result from sarcoidosis of the pericardium, but frank clinical pericarditis is relatively uncommon. Myocardial ischemia and infarction from coronary involvement is relatively rare; however, CAD is likely more common among individuals with sarcoidosis.
| Clinical Manifestations |
| Arrhythmias |
| AV block |
| Bundle branch block |
| Supraventricular tachycardia |
| Ventricular tachycardia |
| Sudden cardiac death |
| Cardiomyopathy |
Heart failure
|
| Pericardial Involvement |
|
Diagnosis of cardiac sarcoidosis
The diagnosis of CS is challenging and there is no widely agreed on diagnostic criteria or reference standard. Blood tests are nonspecific and nondiagnostic. Traditional endomyocardial biopsy (EMB) has a low sensitivity (<25%) compared with autopsy, likely because of the patchy involvement of CS, the typical midwall distribution, and the limited biopsy sites of standard EMB techniques. Newer biopsy techniques guided by imaging and electroanatomic mapping may increase the sensitivity of EMB for CS but are not readily available. Acknowledging the low yield and attendant risks of EMB, several diagnostic criteria that do not rely on cardiac histology have been proposed. Clinical criteria typically incorporate clinical, electrocardiographic, imaging, and histologic findings and allows cases of suspected or probable CS to be diagnosed even in the absence of a diagnostic EMB. The most commonly used criteria are the Japanese Ministry of Health and Welfare (JMHW) guideline revised by the Japanese Circulation Society in 2017 , and the Heart Rhythm Society (HRS) criteria ( Table 23.2 ). Both sets of criteria have limited supporting data and lack prospective validation. Importantly, the HRS algorithm requires a histologic diagnosis of extracardiac sarcoidosis and does not provide any pathway for identifying isolated CS. On the other hand, the revised Japanese diagnostic guidelines now offer criteria for isolated CS that do not require evidence of extracardiac disease ( Table 23.3 ). Because of the limitations of the aforementioned criteria, advanced cardiovascular imaging has an important role in estimating the likelihood of cardiac involvement ( Table 23.4 ).
Prerequisite
| ||||||||||||
|
| Role of Imaging | Details |
|---|---|
| Diagnosis | Estimate likelihood of cardiac involvement:
|
| Prognosis | Identify patients who have a higher risk for sudden cardiac death/ventricular arrythmias. |
| Identify patients who benefit from therapy | Individuals who have more severe or a greater amount of cardiac and extracardiac inflammation may benefit more from immunosuppressive therapies. |
| Follow response to therapy | Because treatment response is variable, imaging can identify individuals who have a favorable response as well as those who do not respond and in whom more aggressive treatment should be considered. |
Role of imaging in cardiac sarcoidodis
Imaging plays a major role in the diagnosis of CS. Both the JMHW and HRS diagnostic algorithms include a variety of imaging findings in their clinical diagnostic pathway, ranging from echocardiography to single photon emission computed tomography (SPECT) myocardial perfusion imaging (MPI), 67 gallium ( 67 Ga), cardiac positron emission tomography (PET), and magnetic resonance imaging (MRI) techniques.
Echocardiography
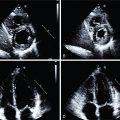
Echocardiography generally demonstrates nonspecific findings in the presence of CS but may be most helpful if abnormal in the presence of extracardiac sarcoidosis. Several studies have estimated that the sensitivity of echocardiography to detect CS ranges from 25% to 30%. Nevertheless, the HRS Expert Consensus Statement suggests echocardiography as one of the initial screening tests in conjunction with a clinical history and a 12-lead electrocardiogram (ECG) in patients with biopsy-proven extracardiac sarcoidosis ( Fig. 23.1 ). Echocardiographic findings in CS include reduced LV function, wall motion abnormalities, and changes in ventricular wall thickness, particularly basal septal thinning or aneurysm. Per the HRS algorithm, cardiac symptoms, an abnormal ECG, or abnormal echocardiographic findings in the setting of biopsy-proven extracardiac sarcoidosis should prompt referral to advanced imaging (see Fig. 23.1 ).
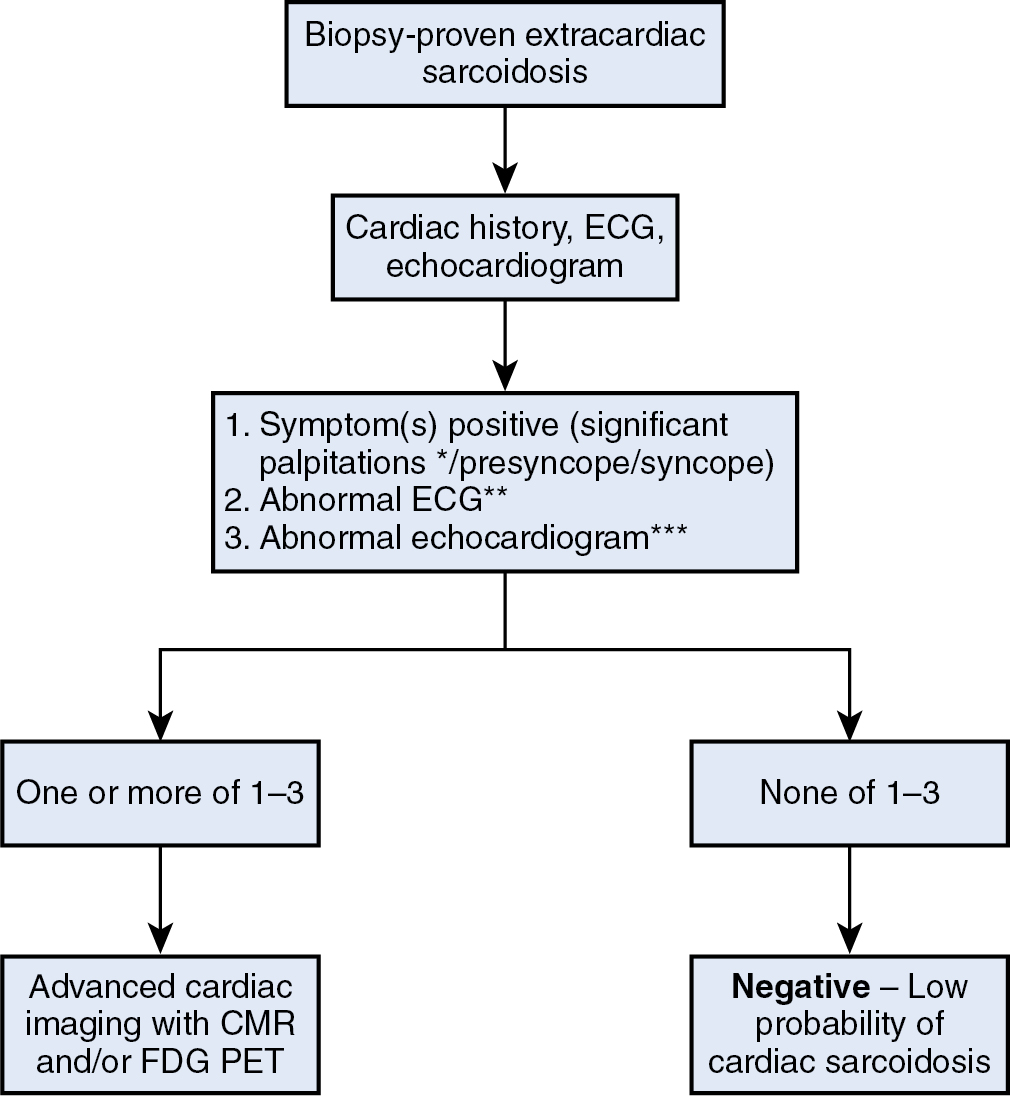
Among patients who are found to have CS, echocardiography may be useful to evaluate the extent of involvement (particularly nonmyocardial involvement, such as the valves or pericardium) and LV and RV function. Given the prognostic importance of LV systolic function in CS, echocardiography may be helpful in identifying high-risk patients.
Single photon emission computed tomography myocardial perfusion imaging and 67 gallium
SPECT 201 thallium (Tl)-based and 99m technetium (Tc)-based findings in CS may also be nonspecific and include focal perfusion defects at rest. When performed in conjunction with radionuclide-based inflammation imaging, SPECT MPI may help differentiate areas of scar or fibrosis from active inflammation. A reverse-redistribution pattern on rest and stress SPECT MPI has also been described whereby focal resting perfusion defects decrease in size or even disappear with vasodilator stress. , This is believed to be due to focal reversible microvascular constriction in the coronary arterioles surrounding the granulomatous inflammation. Although the HRS clinical algorithm does not include SPECT myocardial perfusion defects in its diagnostic pathway, likely because of the low sensitivity and specificity of these findings for CS, the 2017 revised Japanese clinical diagnostic pathway lists SPECT perfusion defects as minor criteria. SPECT MPI may be combined with 18 F-fluorodeoxyglucose (FDG) PET imaging for evaluating CS when perfusion PET is not available.
67 Gallium ( 67 Ga) accumulates in areas of active inflammation and is included as a major criterion in the 2017 revised Japanese criteria. 67 Ga has a high specificity but low sensitivity (<50%), in part because of the challenges of distinguishing cardiac from pulmonary and mediastinal tracer uptake. Combined SPECT MPI and 67 Ga scintigraphy has not been extensively evaluated but may have higher diagnostic accuracy; however, higher radiation exposure from 67 Ga and its relatively low-contrast resolution, along with the availability of superior imaging techniques, have limited its use.
Cardiac positron emission tomography
Cardiac PET is widely recognized as the advanced imaging technique of choice for evaluation and management of CS. Throughout this chapter, PET refers to both combined PET/computed tomography (CT) systems and to PET systems with line sources, unless otherwise specified.
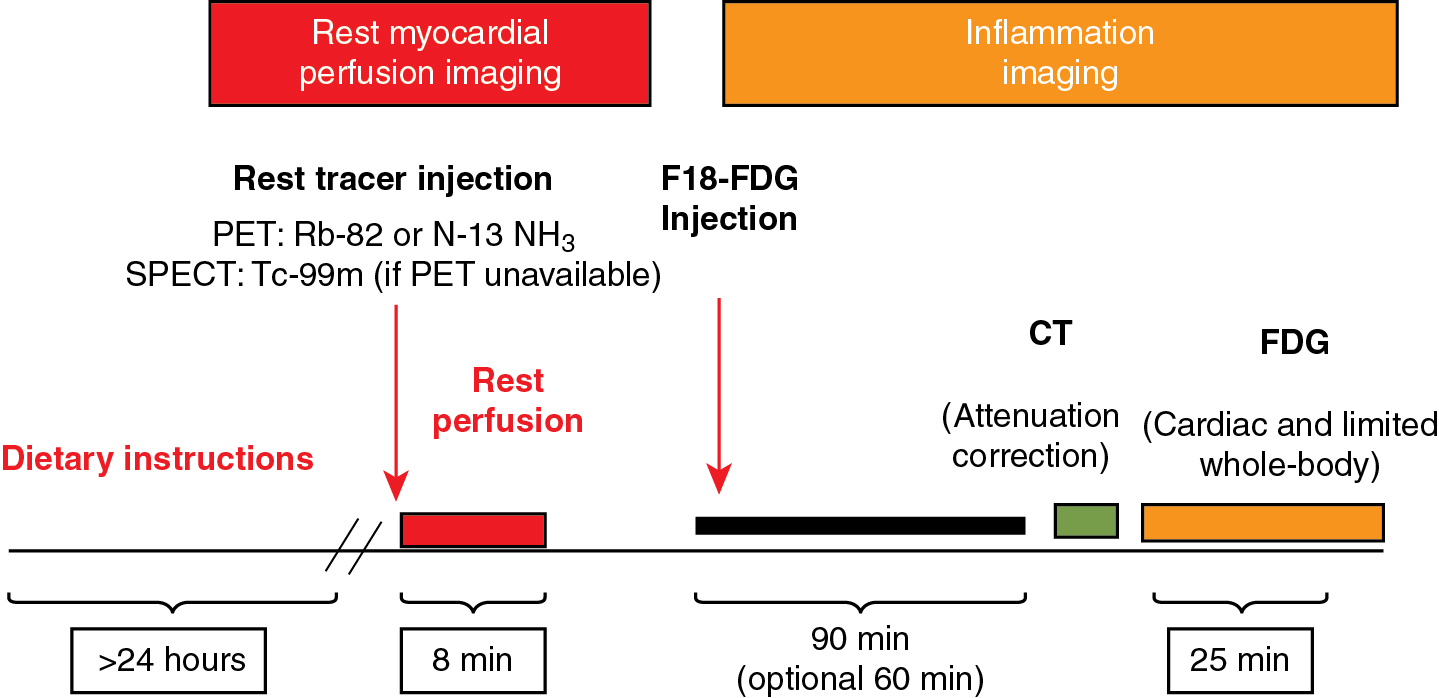
Because of the growing literature demonstrating the diagnostic accuracy and clinical utility of cardiac PET in CS, the 2017 revised Japanese diagnostic guidelines and 2014 HRS Expert Consensus documents both list cardiac FDG uptake as clinical criterion for the diagnosis of CS (see Table 23.2 ). The basis for using FDG to image inflammation in sarcoidosis relates to the increased uptake of FDG, a glucose analog, in macrophage-dense lesions. Although inflammatory cells have increased metabolic activity and are more reliant on external glucose as an energy substrate than normal myocardial cells, the normal heart uses a mixture of free fatty acids and glucose for energy production under normal conditions. Therefore, to improve specificity in identifying pathologic myocardial glucose uptake, patient preparation to suppress physiologic myocardial FDG uptake is a critical aspect of FDG PET imaging ( Fig. 23.2 ).