Pediatric Chest
Susan D. John
Leonard E. Swischuk
Abnormal Lung Opacity
Pulmonary opacities in children are classified in the same way as in adults: as primarily alveolar or interstitial, focal or diffuse, and unilateral or bilateral. Some abnormalities occur in a central or parahilar distribution, whereas others are predominantly peripheral or basal in location. Mixed patterns also occur. An understanding of the causes of these various patterns is necessary to provide a useful interpretation of abnormal lung opacities in children.
Alveolar Patterns
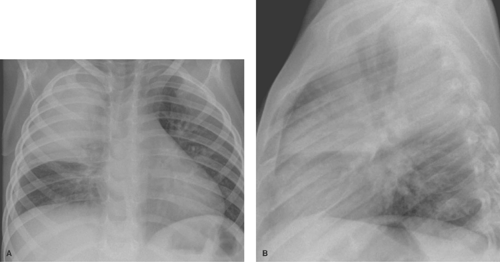
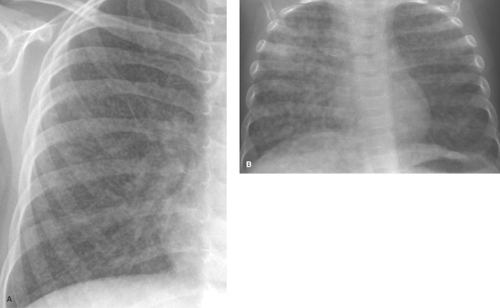
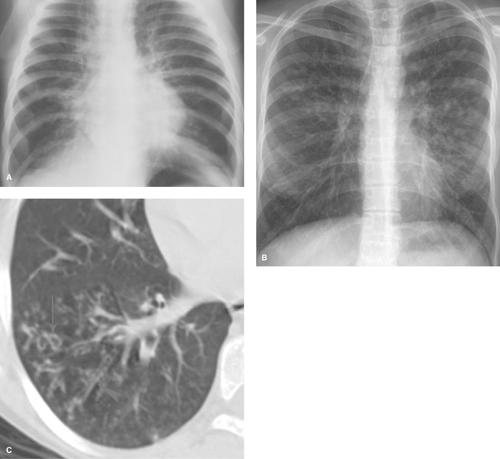
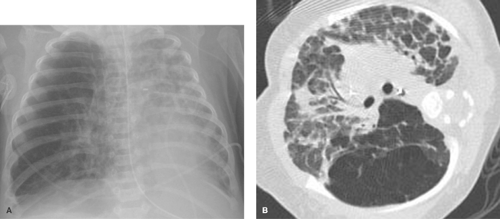
Alveolar consolidation occurs when the alveolar airspace is replaced by a substance, usually fluid. Focal consolidations most often represent exudates associated with bacterial pneumonia (Table 50.1). Bacterial consolidation begins as an oval, round, ill-defined, or fluffy area of solid opacification, often more peripheral than central in location. The pneumonia may progress to involve an entire lobe, but involvement of an entire lung is uncommon. Bacterial pneumonia is a space-occupying process within the lung and, therefore, little or no volume will be lost in the affected lung during the acute stage of infection (Fig. 50.1).
Streptococcus pneumoniae is the most common cause of lobar pneumonia throughout childhood. The incidence of Haemophilus influenzae pneumonia has dramatically decreased in the United States and other developed countries because of the use of the H. influenzae type b vaccine. Mycoplasma infections may also occasionally produce focal consolidating pneumonia.
Consolidations with viral infections are not particularly common but can occur with more serious viral infection, such as adenovirus, influenza, parainfluenza, and respiratory syncytial virus. There is some question as to whether these opacities represent true airspace consolidations. Most likely they represent intense interstitial disease compressing the alveoli. A similar process may occur with Mycoplasma infections.
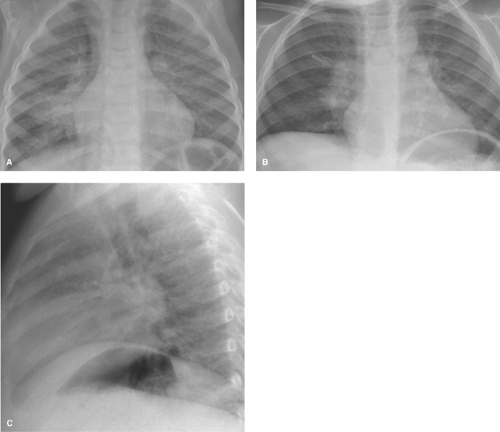
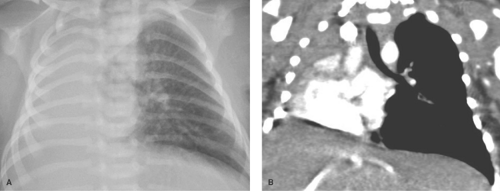
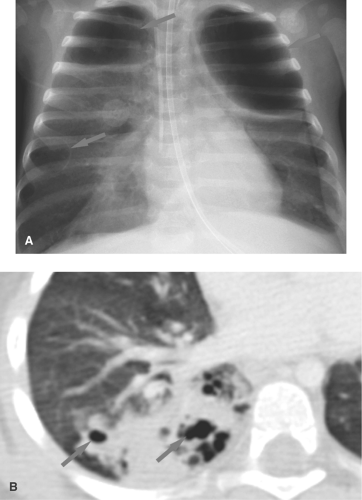
Pneumonia caused by gram-negative bacilli is uncommon in children; it occurs primarily in infants and immunocompromised children. Primary tuberculosis should be considered when the infiltrate is accompanied by hilar lymphadenopathy (Fig. 50.2). Other causes of isolated lung consolidation in children include fungal infection, pulmonary infarction, lung contusion, and focal pulmonary hemorrhage.
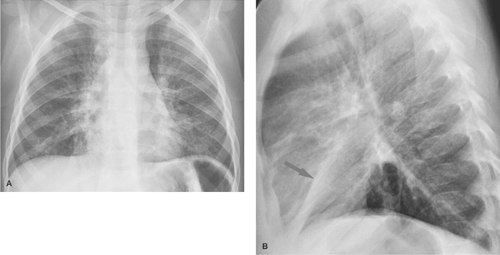
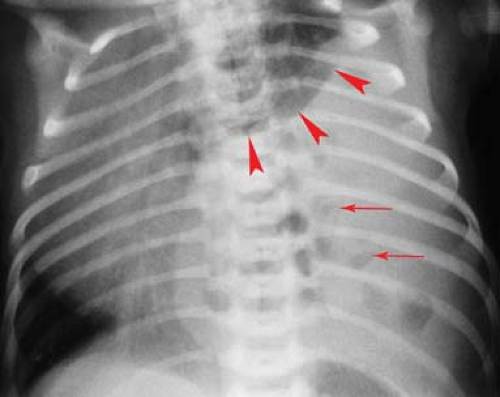
Atelectasis is a common occurrence in children, especially those with bronchial disease such as acute viral respiratory tract infections, reactive airway disease, and asthma. Atelectasis can sometimes resemble a bacterial consolidation. The findings of volume loss, such as shift of the fissures or the mediastinum, help to distinguish atelectasis from bacterial consolidation. Generally, volume loss will not be seen with a bacterial pneumonia until it begins to resolve. A flattened or linear shape in a pulmonary opacity should also suggest that it represents atelectasis rather than consolidation (Fig. 50.3). Atelectasis is particularly problematic in children with asthma, who are also at increased risk for bacterial pneumonia. Clinical information may be necessary to help distinguish atelectasis from pneumonia in such children. Opacities seen in a child with acute asthmatic exacerbation but without high fever, chest pain, or leukocytosis are much more likely to be caused by atelectasis than pneumonia.
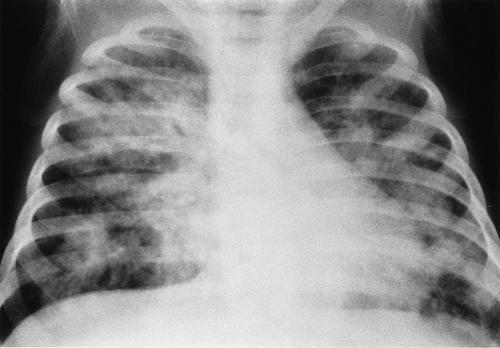
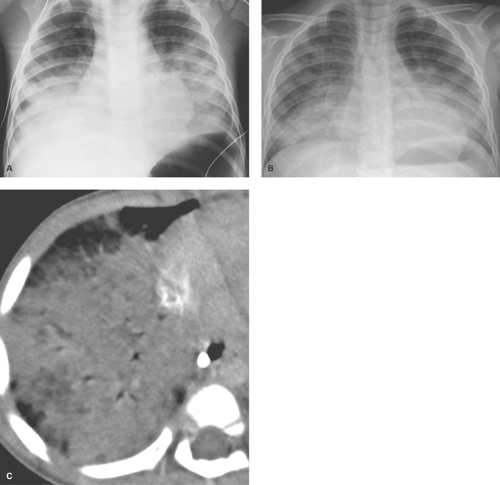
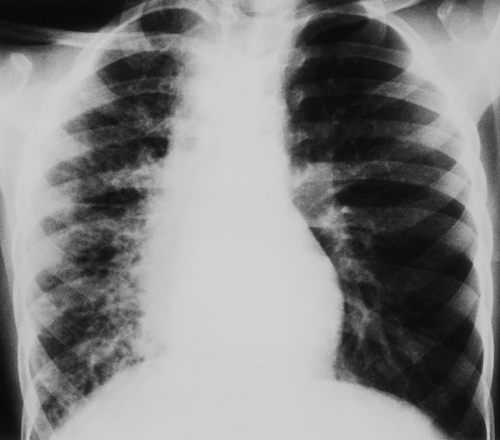
Multiple patchy lung opacities is a pattern seen in a wide variety of conditions (Table 50.2). Such opacities reflect filling of the alveolar space with exudates, edema, or blood. Multiple bilateral alveolar opacities suggest bacterial infection (most commonly staphylococcal) (Fig. 50.4) or fungal disease. Opportunistic infections in immunocompromised patients are much more likely to be multiple and bilateral. Aspiration pneumonia also tends to present with multiple patchy pulmonary opacities. The pneumonitis associated with hydrocarbon ingestion typically occurs in the medial portions of the lung bases (Fig. 50.5). Other less common causes of patchy alveolar opacities include milk allergy, hypersensitivity pneumonitis, uremic lung disease, near drowning, and pulmonary
hemorrhage (i.e., idiopathic pulmonary hemosiderosis). Lipoid pneumonia due to aspiration of mineral oil can develop in children who are given oral mineral oil for treatment of constipation. The high viscosity of mineral oil suppresses the cough reflex, increasing the chance of aspiration during swallowing. Radiographs tend to show bibasilar alveolar opacities, similar to those seen with hydrocarbon aspiration (Fig. 50.5C); however, on CT the air space opacities may show lower attenuation, indicating the presence of fat.
hemorrhage (i.e., idiopathic pulmonary hemosiderosis). Lipoid pneumonia due to aspiration of mineral oil can develop in children who are given oral mineral oil for treatment of constipation. The high viscosity of mineral oil suppresses the cough reflex, increasing the chance of aspiration during swallowing. Radiographs tend to show bibasilar alveolar opacities, similar to those seen with hydrocarbon aspiration (Fig. 50.5C); however, on CT the air space opacities may show lower attenuation, indicating the presence of fat.
Table 50.1 Causes of Focal Alveolar Consolidation | ||||
|---|---|---|---|---|
|
Table 50.2 Sources of Multiple Patchy Lung Opacities | |||||
|---|---|---|---|---|---|
|
Peribronchial and Interstitial Patterns
The vast majority of upper respiratory tract infections in childhood are viral in nature and primarily bronchial in location. Such infections may result in pulmonary opacities that differ significantly from those seen with bacterial pneumonia.
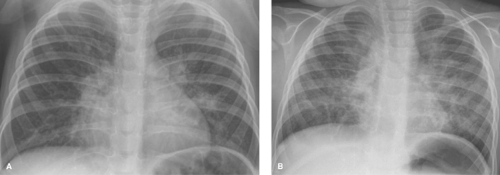
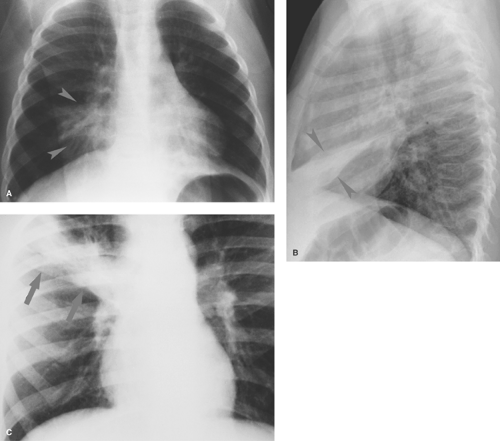
Parahilar peribronchial opacities are sometimes seen and are the result of peribronchial inflammation and edema associated with bronchitis (Table 50.3) (Fig. 50.6A). The pattern consists of bilateral, ill-defined, hazy soft tissue opacity in the hilar region of the lungs. When extensive, these opacities may cause a “shaggy” appearance to the cardiac borders (Fig. 50.6B). Acute peribronchial opacities are most often caused by viral respiratory infections (1,2). Bilateral hilar adenopathy and scattered areas of subsegmental atelectasis are common associated findings (Fig. 50.7). This pattern is very different from the more peripheral alveolar opacification that is usually seen with bacterial pneumonias. However, it should be noted that a superimposed consolidating bacterial pneumonia can develop later in the course of a viral lower respiratory tract infection. Mycoplasma pneumoniae and pertussis infections also commonly produce this pattern (3). Follicular bronchitis, associated with proliferation of lymphoid follicles along the airways, is indistinguishable radiographically. Chlamydia trachomatis infection has a similar appearance and usually occurs just after the newborn period (Fig. 50.8). Chronic bronchial inflammation associated with conditions such as asthma, cystic fibrosis (4) (see Fig. 50.18), immunologic deficiency diseases, and recurrent aspiration may result in persisting patterns of parahilar peribronchial opacity and may eventually lead to bronchiectasis.
Table 50.3 Causes of Parahilar Peribronchial Opacity | ||
|---|---|---|
|
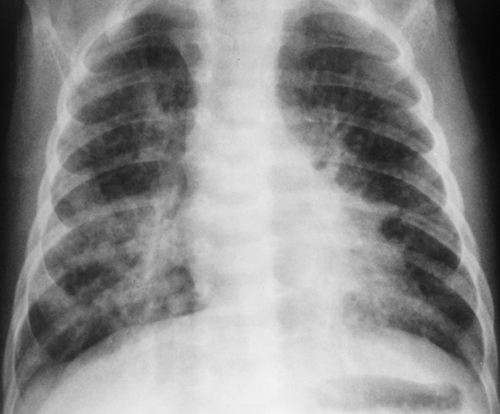 Figure 50.8. Chlamydia Pneumonitis. Prominent bilateral peribronchial opacities with slight nodularity are seen in the lung bases. The appearance is similar to that seen with viral infections. |
Hazy, reticular, or reticulonodular opacities that occur diffusely in the lungs indicate interstitial lung pathology, and the causes include many of the same conditions that cause parahilar peribronchial opacities (Table 50.4). The most common cause of an interstitial pattern in the lungs of a child is viral or Mycoplasma infection (Fig. 50.9). In general, bacterial infections of the lung do not have this appearance, except in the neonate, when bacterial pneumonia can present as diffuse haziness or reticulonodularity. Infections with fungi, such as Histoplasma capsulatum and Coccidioides immitis, can also occasionally result in an interstitial pattern.
Table 50.4 Conditions Causing Hazy, Reticular, or Reticulonodular Patterns | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
|
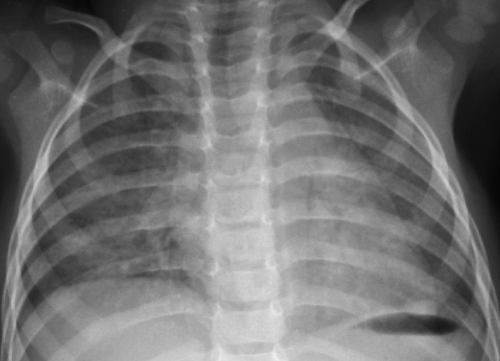 Figure 50.9. Viral Lower Respiratory Tract Infection. A fine interstitial pattern in the lungs of this child with a viral infection causes the lungs to appear diffusely hazy. |
Pulmonary edema, when it is confined to the interstitial space, often produces a hazy or reticular pattern in the lungs. Cardiogenic pulmonary edema occurs when the pulmonary venous pressures are elevated because of left-sided myocardial failure or congenital lesions that impede blood flow through the left side of the heart (e.g., pulmonary vein atresia, cor triatriatum, hypoplastic left heart syndrome). Noncardiogenic causes of pulmonary edema predominate in children. One of the most common causes of pulmonary edema in children is acute glomerulonephritis (Fig. 50.10). Sodium and fluid retention leads to hypervolemia, which can then result in cardiomegaly and pulmonary vascular congestion with edema. The radiographic appearance can be indistinguishable from that of edema caused by cardiac failure. Other noncardiogenic causes of pulmonary edema in children include near drowning, increased intracranial pressure, inhalation injuries, drug overdose, and acute respiratory distress syndrome.
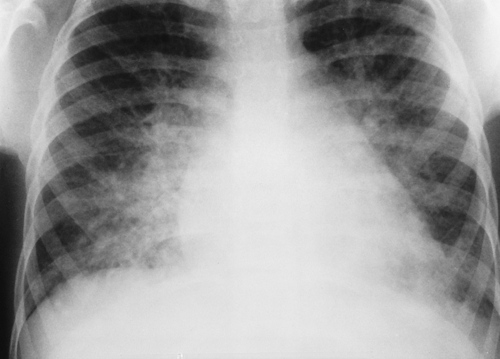 Figure 50.10. Acute Glomerulonephritis. The heart is mildly enlarged with marked bilateral vascular congestion edema reflecting fluid overload. |
Pulmonary lymphangiectasia is a rare condition that consists of dilated lymphatic channels secondary to either abnormal embryonic development of the lymphatic system or obstruction
of lymphatic drainage. The dilated lymphatics cause a coarsely nodular or reticular pattern in the lungs, usually developing early in infancy (Fig. 50.11A) (5,6). Pulmonary hemangiomatosis is a similar rare condition. Recurrent hemorrhage into the lungs in patients with idiopathic pulmonary hemosiderosis eventually leads to a chronic diffuse, hazy or reticular pattern in the lungs, representing pulmonary fibrosis (Fig. 50.11B,C). Langerhans cell histiocytosis (LCH) causes an interstitial pattern that often is more prominent in the upper lung zones. The lung volumes in Langerhans cell histiocytosis are normal or increased, which differs from fibrotic conditions, in which lung volumes tend to be decreased. High-resolution CT may show cysts and nodules (7). High-resolution CT can be used successfully to better evaluate interstitial and airway abnormalities in pediatric patients under certain conditions (Fig. 50.19C). Low-dose techniques with limited coverage are advised (8).
of lymphatic drainage. The dilated lymphatics cause a coarsely nodular or reticular pattern in the lungs, usually developing early in infancy (Fig. 50.11A) (5,6). Pulmonary hemangiomatosis is a similar rare condition. Recurrent hemorrhage into the lungs in patients with idiopathic pulmonary hemosiderosis eventually leads to a chronic diffuse, hazy or reticular pattern in the lungs, representing pulmonary fibrosis (Fig. 50.11B,C). Langerhans cell histiocytosis (LCH) causes an interstitial pattern that often is more prominent in the upper lung zones. The lung volumes in Langerhans cell histiocytosis are normal or increased, which differs from fibrotic conditions, in which lung volumes tend to be decreased. High-resolution CT may show cysts and nodules (7). High-resolution CT can be used successfully to better evaluate interstitial and airway abnormalities in pediatric patients under certain conditions (Fig. 50.19C). Low-dose techniques with limited coverage are advised (8).
Interstitial lung disease that predominates in the lower lobes can be seen with tuberous sclerosis, connective tissue diseases, and primary interstitial pneumonitis. Leukemia, lymphoma, and lymphatic metastases to the lungs can also cause a reticular or reticulonodular infiltrative pattern. Mycoplasma pneumonitis sometimes presents as an interstitial pattern confined to one lobe of the lung (3).
Miliary nodules usually consist of tiny nodules (smaller than 5 mm) that are randomly distributed throughout the lungs. This pattern in children is most often caused by hematogenous dissemination of tuberculosis or histoplasmosis (Fig. 50.12), although viral pneumonitis, idiopathic pulmonary hemosiderosis, and metastatic disease can also have this appearance (Table 50.5). The tiny nodules can be difficult to see on radiographs in some cases, and CT can better define the nodules and other associated abnormalities, such as lymphadenopathy (9). Acute disseminated tuberculosis in infants and young children can sometimes produce larger nodules (Fig. 50.12B), and CT may show larger areas of opacity caused by coalescent nodules and interstitial thickening.
Opportunistic infections may occur in children with HIV infection and other forms of congenital or acquired immunodeficiency. Infection with common viral, bacterial, and fungal organisms creates a pattern similar to that seen in immunocompetent children, but the findings tend to be more rapidly progressive and more pronounced. Lymphocytic infiltrative disease produces a reticulonodular pattern that is indistinguishable from infection (Fig. 50.13), except for its chronicity.
Abnormal Lung Volume
Pulmonary aeration abnormalities are best evaluated on the chest radiograph by observing the following criteria: (1) the relative size of a lung or hemithorax, (2) the degree of radiolucency of the lung, and (3) the pulmonary vascularity or blood flow to the lung. Bilateral smallness of the lungs is commonly caused by less than complete inspiration. The technical difficulties of obtaining good inspiratory chest films in children are significant. The lungs may appear small if the diaphragm is elevated, because of either neuromuscular abnormality or the presence of large masses, fluid collections, or bowel distension in the abdomen. Infrequently, inspiratory obstruction of the trachea can lead to bilateral underaeration of the lungs. Causes of such obstruction include intratracheal masses or foreign bodies, or extrinsic compression of the trachea by anomalous vascular structures. A hyperlucent but small hemithorax usually signifies some degree of pulmonary hypoplasia, either congenital or acquired.
Table 50.5 Causes of Miliary Nodules | |||
|---|---|---|---|
|
Pulmonary Hypoplasia or Agenesis
Congenital pulmonary hypoplasia is associated with hypoplasia or absence of the ipsilateral PA (10); thus, pulmonary vascular markings will be diminished in size on radiographs. Congenital lung hypoplasia is sometimes associated with congenital heart disease, most often tetralogy of Fallot or persistent truncus arteriosus. In cases of tetralogy of Fallot, the left lung is hypoplastic. A hypogenetic lung is one of the features of congenital pulmonary venolobar (scimitar) syndrome. Usually, the right lung is the hypoplastic lung in this condition. The scimitar is a curvilinear vertical vein that usually extends along the right heart border and empties into the inferior vena cava, resulting in partial anomalous pulmonary venous return. Other variable components of this syndrome include hypoplasia or absence of the PA, pulmonary sequestration, systemic arterialization of the hypoplastic lungs, accessory diaphragm, and absent inferior vena cava (11,12). Pulmonary agenesis is a rare anomaly that results from an insult during the fourth week of fetal life. The right and left lungs are affected with equal frequency. Right pulmonary agenesis has an increased association with other congenital malformations involving the heart, skeleton, GI tract, and genitourinary tract. Chest radiographs or CT demonstrates severe volume loss and opacity on the side of agenesis, often with close spacing of the ribs. The bronchus and PA to the affected lung are absent, and blood flow to the contralateral lung is increased (Fig. 50.14).
Pulmonary hypoplasia in the neonate can be unilateral or bilateral. Bilateral pulmonary hypoplasia is most often the result of compression of the lungs during fetal development. Congenital bone dysplasias and syndromes associated with short ribs and a small thoracic cage (asphyxiating thoracic
dystrophy, thanatophoric dwarfism, Ellis-van Creveld syndrome) compress the lungs and cause hypoplastic lungs (Fig. 50.15). The degree of hypoplasia is often severe and leads to the demise of these infants. Chromosomal abnormalities such as the trisomies are associated with hypoplastic lungs, and in some infants, hypoplasia is “primary” and unexplained.
dystrophy, thanatophoric dwarfism, Ellis-van Creveld syndrome) compress the lungs and cause hypoplastic lungs (Fig. 50.15). The degree of hypoplasia is often severe and leads to the demise of these infants. Chromosomal abnormalities such as the trisomies are associated with hypoplastic lungs, and in some infants, hypoplasia is “primary” and unexplained.
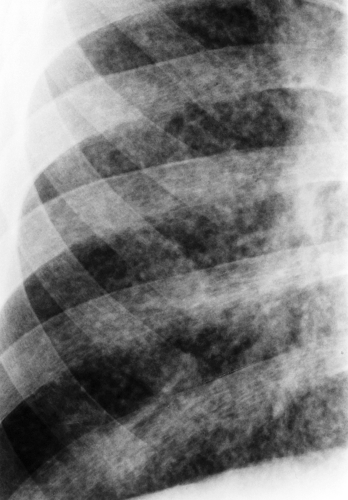 Figure 50.13. Lymphocytic Interstitial Pneumonitis. The diffuse interstitial pattern in the lungs of an HIV-positive child represents lymphocytic pneumonitis. |
The most common cause of intrathoracic compression of the fetal lungs is congenital diaphragmatic hernia. Although the hernia itself is most often unilateral, the increased volume of the thorax on the side of the hernia causes compression of the contralateral lung, resulting in bilateral and asymmetric lung hypoplasia (Fig. 50.16). The degree of hypoplasia varies in severity; the earlier in gestation that the hernia occurs, the more severe the lung hypoplasia. Pulmonary insufficiency is the most significant cause of morbidity and mortality in these infants. Infants with severely hypoplastic lungs can be supported with artificial ventilation or extracorporeal membranous oxygenation (ECMO) until their lungs develop enough to permit survival. Other causes of intrathoracic compression leading to bilateral pulmonary hypoplasia include bilateral chylothorax, large intrathoracic cysts or tumors (neuroblastoma, teratoma, cystic adenomatoid malformation), or marked cardiomegaly.
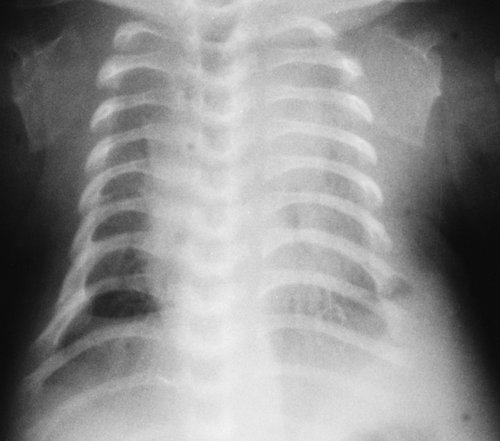 Figure 50.15. Lung Hypoplasia. The very small thoracic cage caused by rib shortening in this thanatophoric dwarf is associated with marked lung hypoplasia. |
Extrathoracic compression of the fetal lungs is most often caused by oligohydramnios secondary to fetal urinary tract abnormalities or by abnormal amniotic fluid production or leakage. Potter syndrome, associated with bilateral renal agenesis, congenital renal cystic disease, or obstructive uropathy
(posterior urethral valves, prune belly syndrome), commonly results in hypoplastic lungs. Additional causes include neuromuscular abnormalities with persistent elevation of the diaphragm or prolonged distension of the abdomen by large abdominal masses, enlarged kidneys, or ascites.
(posterior urethral valves, prune belly syndrome), commonly results in hypoplastic lungs. Additional causes include neuromuscular abnormalities with persistent elevation of the diaphragm or prolonged distension of the abdomen by large abdominal masses, enlarged kidneys, or ascites.
Swyer-James syndrome is an acquired hypoplastic lung that develops following severe obliterative bronchiolitis, leading to bronchiolar obstruction, bronchiectasis, and distal airspace destruction. Bronchiectasis is not present in all cases (13). Air enters the lung by air drift phenomenon but becomes trapped because of the bronchiolar obstruction. Air trapping results in a lung that changes very little in size between inspiration and expiration. This important feature helps to distinguish the hypoplastic Swyer-James lung from the congenitally hypoplastic lung. Radionuclide ventilation/perfusion studies can be used to verify the expiratory airway obstruction as well as the diminished perfusion of the hypoplastic lung. CT is more sensitive than radiographs in detecting areas of air trapping and helps to exclude other causes of central bronchial obstruction (14). Although the Swyer-James lung is classically clear and hyperlucent (Fig. 50.17), some patients show a fibrotic reticular pattern in the hypoplastic lung. Other causes of a unilateral small reticular lung include scarring after radiation therapy or congenital unilateral pulmonary vein atresia or stenosis (Fig. 50.18). The reticular pattern of the lung in pulmonary vein atresia is caused by a combination of interstitial pulmonary edema, fibrosis, and dilated interstitial lymphatics.
 Figure 50.17. Swyer-James Lung. The left lung is small and relatively hyperlucent when compared with the right lung. The left pulmonary vascularity is decreased. |
Bilateral Lung Hyperinflation
Bilateral overaeration of the lungs is most often caused by airway obstruction that can be central or diffuse and peripheral (Table 50.6).
Table 50.6 Possible Causes of Bilateral Lung Hyperinflation | ||
|---|---|---|
|
Small Airway Obstruction. Widespread obstruction of the peripheral airway is a common cause of obstructive emphysema and is most often the result of viral bronchitis and bronchiolitis or asthma. Acute bronchiolitis in infants is often
accompanied by severe air trapping and overinflation of the lungs, with little or no other visible pulmonary abnormality. Hyperinflation tends to be less severe in older children with viral lower respiratory tract infections, but the mechanism (i.e., mucosal edema and bronchospasm secondary to inflammation) is the same. Infants with cystic fibrosis can present with an appearance identical to that of bronchiolitis. Cystic fibrosis should be considered in any infant who presents with multiple episodes of bronchiolitis (Fig. 50.19). Peripheral small airway obstruction with parahilar peribronchial opacities is seen with certain immunologic deficiency diseases, chronic aspiration, and graft versus host disease. Bronchiolitis obliterans can be caused by various inflammatory or toxic insults to the airway and is commonly a postinfectious complication in children. The imaging findings in bronchiolitis obliterans in children are variable but usually involve some degree of hyperlucency of the lungs, sometimes accompanied by atelectasis, hyperinflation, or peribronchial thickening (15,16).
accompanied by severe air trapping and overinflation of the lungs, with little or no other visible pulmonary abnormality. Hyperinflation tends to be less severe in older children with viral lower respiratory tract infections, but the mechanism (i.e., mucosal edema and bronchospasm secondary to inflammation) is the same. Infants with cystic fibrosis can present with an appearance identical to that of bronchiolitis. Cystic fibrosis should be considered in any infant who presents with multiple episodes of bronchiolitis (Fig. 50.19). Peripheral small airway obstruction with parahilar peribronchial opacities is seen with certain immunologic deficiency diseases, chronic aspiration, and graft versus host disease. Bronchiolitis obliterans can be caused by various inflammatory or toxic insults to the airway and is commonly a postinfectious complication in children. The imaging findings in bronchiolitis obliterans in children are variable but usually involve some degree of hyperlucency of the lungs, sometimes accompanied by atelectasis, hyperinflation, or peribronchial thickening (15,16).
Central airway obstruction leading to bilateral overaeration of the lungs is less common than peripheral obstruction. Intratracheal foreign bodies, neoplasms, granulomas, and intrinsic stenoses of the trachea are all rather rare. More commonly, tracheal obstruction is the result of extrinsic compression caused by cysts, neoplasms, adenopathy, and congenital vascular abnormalities.
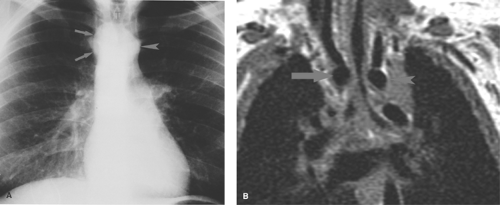
A right-sided aortic arch is the key radiographic clue to the presence of an obstructing vascular ring (Fig. 50.20). Most cases of double aortic arch (70%) consist of a large posterior, right-sided arch and a small anterior, left-sided arch that encircle the esophagus and the trachea (16). The diagnosis may be verified by barium esophagram, which shows a reverse S configuration caused by the bilateral vascular impressions on the esophagus (Fig. 50.21). Lateral radiographs demonstrate increased retrotracheal opacity, tracheal narrowing, and anterior tracheal bowing (17). Similar radiographic abnormalities are seen when the vascular ring consists of a right ascending aortic arch, an aberrant left subclavian artery that passes posterior to the esophagus, and a ligamentum arteriosum or persistent ductus arteriosus stretching from the left subclavian artery to the PA anterior to the trachea. Definitive evaluation of the vascular anatomy is best accomplished with MRI or helical CT (18,19) (Fig. 50.20).
The pulmonary sling anomaly is a rare condition that may also result in tracheal compression and bilateral hyperaeration of the lungs. The left PA arises from the right PA, and as it courses to the left lung, the left PA passes between the trachea and the esophagus and compresses the trachea posteriorly (Fig. 50.22). The right lung may be either underaerated or overaerated.
Asymmetric/Unilateral Aeration Abnormalities
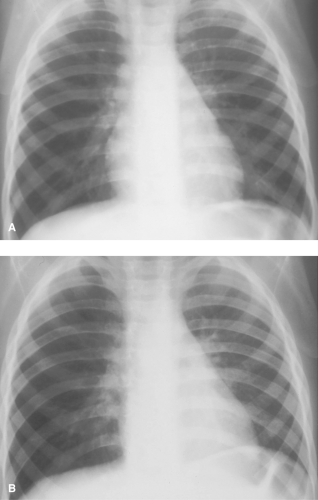
Pulmonary aeration abnormalities are frequently asymmetric or unilateral. A large, hyperlucent hemithorax most often indicates overinflation of an entire lobe or lung. Such hyperaeration may represent obstructive emphysema (Table 50.7) or compensatory overinflation resulting from decreased volume of the contralateral lung. Pulmonary vascularity is the key to differentiation. Obstructive emphysema generally results in diminished size of the pulmonary vessels because of compression and hypoxia-induced reflex arterial spasm. With compensatory hyperinflation, the pulmonary vessels are normal or even increased in size. If doubt remains, inspiratory and expiratory frontal views of the chest or fluoroscopy can be helpful. The lung that changes the least in volume between inspiration and expiration is the abnormal lung (Fig. 50.23). This holds true whether the lung is obstructed and overinflated or is small because of atelectasis or hypoplasia. The only exception to this rule is mild congenital pulmonary hypoplasia, which is associated with relatively normal lung dynamics.
Table 50.7 Causes of Unilateral Obstructive Emphysema | |||||||
|---|---|---|---|---|---|---|---|
|
Congenital lobar hyperinflation or emphysema consists of obstructive emphysema of a single lobe of the lung, most commonly the left upper, right middle, or right upper lobe.
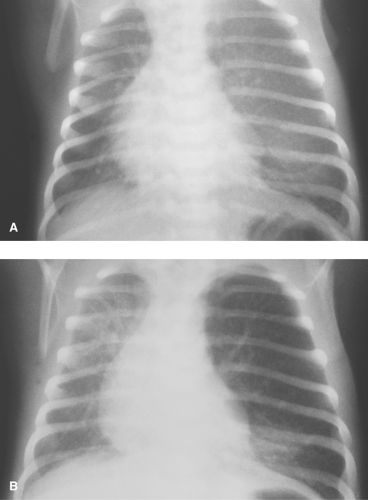
Usually, emphysema is the result of underdevelopment of the segmental bronchial cartilage, which leads to expiratory airway collapse and a ball valve type of obstruction. Early in the newborn period, the obstructed lobe may be opaque because of delayed clearance of fluid distal to the obstruction. Gradually, the fluid clears, and the involved lobe is filled with air and overinflated (Fig. 50.24). A severely enlarged emphysematous lobe can occupy the entire hemithorax and erroneously suggest a pneumothorax or emphysema of the entire lung. Careful inspection reveals the collapsed, compressed adjacent lobes of the lung, confirming the diagnosis. A similar appearance can occur in the rare case of bronchial stenosis or atresia. Classically, an oval opacity is seen adjacent to the overinflated lung, near the hilum, representing a collection of mucus (mucocele) within the obstructed bronchus. Acquired lobar emphysema can occur as a result of bronchial damage associated with inflammatory conditions or bronchopulmonary dysplasia (BPD) (Fig. 50.25). BPD is a common complication of preterm birth, the manifestations of which are changing slightly in the postsurfactant therapy era. Current therapies involving prenatal corticosteroids, postnatal surfactant treatment, and ventilation for shorter times with lower pressures have caused changes in the pathogenesis of BPD (25). Peripheral abnormalities have become more common, such as decreased alveolarization of the lungs and abnormal pulmonary vascular development (20). However, bronchial pathology remains an important cause of morbidity in BPD. CT is not commonly used in the routine evaluation of BPD in infants and children; however, CT can be helpful for evaluating severe obstructive emphysema in some patients that may require surgical resection (Fig. 50.25B).
Usually, emphysema is the result of underdevelopment of the segmental bronchial cartilage, which leads to expiratory airway collapse and a ball valve type of obstruction. Early in the newborn period, the obstructed lobe may be opaque because of delayed clearance of fluid distal to the obstruction. Gradually, the fluid clears, and the involved lobe is filled with air and overinflated (Fig. 50.24). A severely enlarged emphysematous lobe can occupy the entire hemithorax and erroneously suggest a pneumothorax or emphysema of the entire lung. Careful inspection reveals the collapsed, compressed adjacent lobes of the lung, confirming the diagnosis. A similar appearance can occur in the rare case of bronchial stenosis or atresia. Classically, an oval opacity is seen adjacent to the overinflated lung, near the hilum, representing a collection of mucus (mucocele) within the obstructed bronchus. Acquired lobar emphysema can occur as a result of bronchial damage associated with inflammatory conditions or bronchopulmonary dysplasia (BPD) (Fig. 50.25). BPD is a common complication of preterm birth, the manifestations of which are changing slightly in the postsurfactant therapy era. Current therapies involving prenatal corticosteroids, postnatal surfactant treatment, and ventilation for shorter times with lower pressures have caused changes in the pathogenesis of BPD (25). Peripheral abnormalities have become more common, such as decreased alveolarization of the lungs and abnormal pulmonary vascular development (20). However, bronchial pathology remains an important cause of morbidity in BPD. CT is not commonly used in the routine evaluation of BPD in infants and children; however, CT can be helpful for evaluating severe obstructive emphysema in some patients that may require surgical resection (Fig. 50.25B).
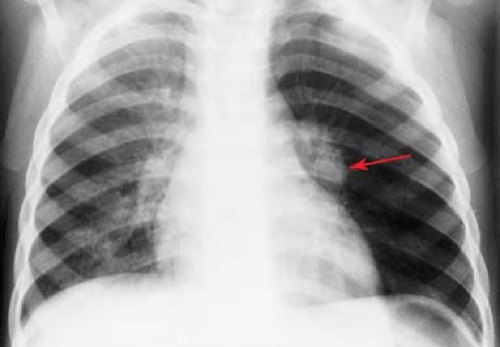
Endobronchial Lesions. In older infants and children, obstructive emphysema is most often caused by an endobronchial foreign body or a mucous plug (Fig. 50.23). Mucous plugs occur most commonly in asthmatics and in children with viral lower respiratory tract infections. Other less common causes of unilateral obstructive emphysema include endobronchial masses such as tuberculous granulomas (Fig. 50.26) and extrinsic compressing lesions such as anomalous blood vessels and mediastinal tumors and cysts. Pneumothorax may cause a large hyperlucent hemithorax that mimics obstructive emphysema. In supine patients, the pleural air may lie entirely along the anterior surface of the lung and
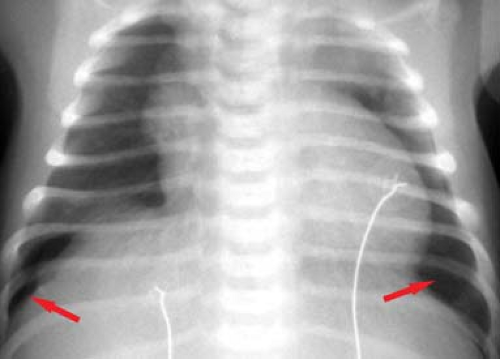
no free lung edge will be visible. Clues to the presence of an anterior pneumothorax include increased radiolucency of the hemithorax and increased sharpness of the mediastinal border (Fig. 50.27). In newborns, a pneumothorax can compress the normal thymus gland, creating a mediastinal “pseudomass.” Rarely, an air-filled lung cyst or pneumatocele, or a markedly dilated stomach in a diaphragmatic hernia, can occupy an entire hemithorax, rendering it hyperlucent.
no free lung edge will be visible. Clues to the presence of an anterior pneumothorax include increased radiolucency of the hemithorax and increased sharpness of the mediastinal border (Fig. 50.27). In newborns, a pneumothorax can compress the normal thymus gland, creating a mediastinal “pseudomass.” Rarely, an air-filled lung cyst or pneumatocele, or a markedly dilated stomach in a diaphragmatic hernia, can occupy an entire hemithorax, rendering it hyperlucent.
Pulmonary Cavities
Cavities in the lungs of children are most often inflammatory or postinflammatory.
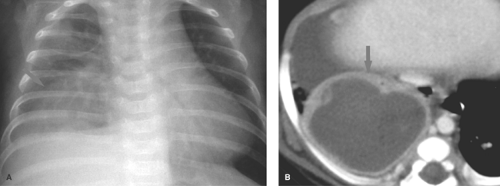
Lung abscesses usually develop as a complication of a bacterial pneumonia and can be solitary or multiple. The wall of an abscess is thick and irregular and some contain air–fluid levels (Fig. 50.28). CT is valuable for distinguishing intrapulmonary
abscess from loculated empyema in the pleural space. An abscess may also form distal to a bronchial obstruction, which in children is often caused by a retained foreign body. Cavitary tuberculosis is rare in childhood, and echinococcal cysts are rare outside of endemic areas.
abscess from loculated empyema in the pleural space. An abscess may also form distal to a bronchial obstruction, which in children is often caused by a retained foreign body. Cavitary tuberculosis is rare in childhood, and echinococcal cysts are rare outside of endemic areas.
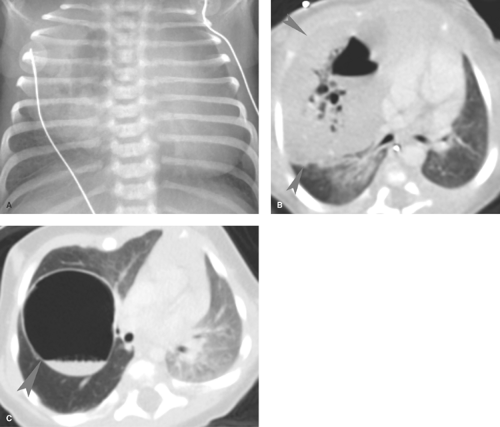
Pneumatoceles are thin-walled lung cavities that commonly occur with pulmonary infections in children. Staphylococcal pneumonia is classically associated with lung necrosis and pneumatocele formation, but cavitation now is more commonly seen with S. pneumoniae and can occur with other infections, including tuberculosis (20) (Fig. 50.29). Pneumatoceles develop from a bronchiolar obstruction that leads to air trapping and alveolar rupture. Pneumatoceles can become large and cause significant mass effect. More often, the pneumatoceles remain relatively small and resolve spontaneously. Occasionally, pneumatoceles rupture, leading to pneumothorax or pneumomediastinum. Other causes of pneumatoceles in children include blunt chest trauma, hydrocarbon pneumonitis, and Langerhans cell histiocytosis.
Congenital lung cysts are uncommon and may be indistinguishable from pneumatoceles. Congenital cysts are usually thin walled and more commonly occur in the lower lobes. Most are asymptomatic unless they become infected or undergo rapid expansion with the development of a tension phenomenon.
Congenital pulmonary airway malformation (cystic adenomatoid malformation) (CPAM) is a heterogeneous group of
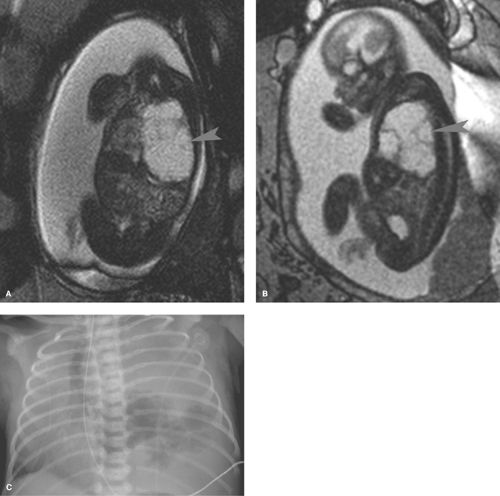
lesions that result from early maldevelopment of the airway. Although the lesions are commonly cystic, noncystic varieties occur (21,22,23). CPAMs are commonly identified at prenatal US and are well evaluated with fetal MRI (Fig. 50.30). MRI shows a hyperintense cystic lesion often multilocular, with well-defined walls (23). Various classifications of CPAMs have been developed, primarily based on cyst size (24). Failed alveolarization of the lung and abnormal pulmonary vascular development now appear to play a more prominent role (26). The radiographic appearance can vary from a predominantly solid lesion with multiple tiny cysts to multiple, large, thin-walled cysts that mimic congenital lobar emphysema (Fig. 50.31) (26). In the first days of life, the cysts are fluid filled and the lesion has the appearance of a solid mass. With time, air replaces the fluid, and small radiolucent cysts become apparent. The cysts gradually enlarge and can cause enough mass effect to lead to respiratory distress. The malformation is usually unilateral and can affect any portion of the lung. Recent literature suggests that CPAM and bronchopulmonary sequestration share a common developmental pathway, triggered by airway obstruction in the fetus (27).
lesions that result from early maldevelopment of the airway. Although the lesions are commonly cystic, noncystic varieties occur (21,22,23). CPAMs are commonly identified at prenatal US and are well evaluated with fetal MRI (Fig. 50.30). MRI shows a hyperintense cystic lesion often multilocular, with well-defined walls (23). Various classifications of CPAMs have been developed, primarily based on cyst size (24). Failed alveolarization of the lung and abnormal pulmonary vascular development now appear to play a more prominent role (26). The radiographic appearance can vary from a predominantly solid lesion with multiple tiny cysts to multiple, large, thin-walled cysts that mimic congenital lobar emphysema (Fig. 50.31) (26). In the first days of life, the cysts are fluid filled and the lesion has the appearance of a solid mass. With time, air replaces the fluid, and small radiolucent cysts become apparent. The cysts gradually enlarge and can cause enough mass effect to lead to respiratory distress. The malformation is usually unilateral and can affect any portion of the lung. Recent literature suggests that CPAM and bronchopulmonary sequestration share a common developmental pathway, triggered by airway obstruction in the fetus (27).
Congenital Diaphragmatic Hernia. Air-filled loops of bowel in a congenital diaphragmatic hernia can resemble the multiple cysts of cystic adenomatoid malformation. An important clue to the correct diagnosis of diaphragmatic hernia is the absence or paucity of gas-filled bowel loops within the abdomen (Fig. 50.32). Congenital diaphragmatic hernias most often occur through the foramen of Bochdalek, which lies posteriorly and medially in each hemidiaphragm (28). Left-sided hernias are more common (80%) and more frequently involve bowel herniation. Solid abdominal viscera are more likely to herniate into the chest through right-sided
hernias (Fig. 50.32B). Hernias through the foramen of Morgagni, which lies anteriorly, are less common and usually are less severe (Fig. 50.32C).
hernias (Fig. 50.32B). Hernias through the foramen of Morgagni, which lies anteriorly, are less common and usually are less severe (Fig. 50.32C).
Infants with large diaphragmatic hernias usually present with severe respiratory distress immediately after birth. Compression of the ipsilateral lung in utero causes it to be hypoplastic, and often the contralateral lung is also small. The patients are profoundly hypoxic, and persistent fetal circulation caused by hypoxia-induced pulmonary hypertension usually further compromises the infant’s condition. Despite recent advances in early diagnosis and management, the morbidity and mortality with this condition remains high. ECMO has improved the survival of some patients by circumventing the problem of pulmonary hypertension and the right-to-left shunting of blood away from the lungs. Congenital diaphragmatic hernia may occasionally be minimally symptomatic at birth, presenting later in life.
Lung Disease in the Neonate
The conditions leading to respiratory distress in the newborn infant are numerous and can be divided into those that can be treated medically and those that require surgical intervention. Surgical conditions consist primarily of congenital and developmental abnormalities that result in a space-occupying lesion within the chest (diaphragmatic hernia, congenital lobar emphysema, chylothorax, pneumothorax, cystic adenomatoid malformation). This section will deal with diffuse pulmonary disease of the newborn.
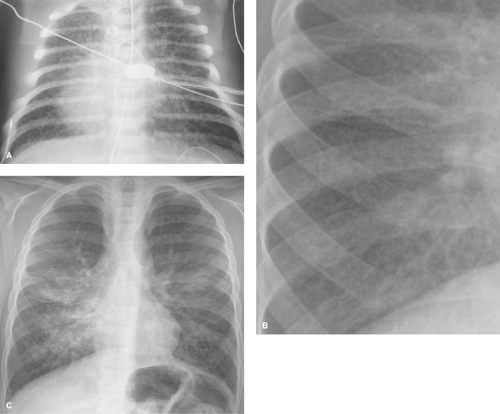
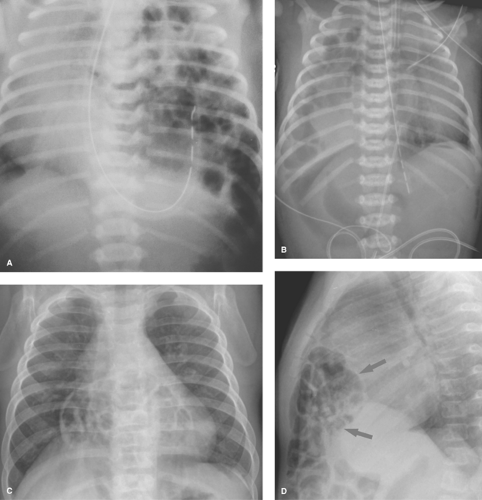
Surfactant deficiency disease (hyaline membrane disease) is one of the most common causes of respiratory distress in the newborn (29,30). It is most common in premature infants; however, it occasionally occurs in full-term infants of diabetic mothers. In both cases, lung immaturity is the main predisposing factor. The primary abnormality is a lack of surfactant normally produced by the type II alveolar cells. This substance is responsible for decreasing the surface tension of the alveoli. When absent, the alveoli are poorly distensible and remain collapsed. A cycle of hypoxia, acidosis, and diminished perfusion results. Clinically, these infants present with respiratory distress within the first few hours after birth. The classic radiographic findings of surfactant deficiency disease consist of lungs that are small in volume and have a finely granular pattern, with air bronchograms that extend into the lung periphery (Fig. 50.33). The granular pattern reflects the histologic findings of distended alveolar ducts and terminal
bronchioles superimposed over generalized alveolar collapse. When the alveoli and terminal bronchioles overdistend, small, round, 1- to 2-mm bubbles result. During expiration, the air bronchograms and granular pattern disappear and the lungs become totally opaque. With surfactant therapy, these changes are very transient.
bronchioles superimposed over generalized alveolar collapse. When the alveoli and terminal bronchioles overdistend, small, round, 1- to 2-mm bubbles result. During expiration, the air bronchograms and granular pattern disappear and the lungs become totally opaque. With surfactant therapy, these changes are very transient.
Similar lung opacities can be seen with neonatal pneumonia, pulmonary lymphangiectasia, neonatal retained fluid syndrome, and congenital heart abnormalities associated with severe pulmonary venous obstruction. However, unlike patients with surfactant deficiency, the lung volumes in these conditions are usually normal to increased (Fig. 50.34). In a few cases of neonatal pneumonia, the lung pattern is indistinguishable from that seen in surfactant deficiency. Until recently, the primary form of therapy for surfactant deficiency consisted of positive pressure-assisted ventilation, which attempts to force air deeper into the respiratory tree and alveoli. Although in some patients the use of assisted ventilation significantly improves oxygenation, in others, the elevated airway pressures result in complications caused by air leakage from the distended terminal airways. Air dissects through the interstitium and lymphatics (pulmonary interstitial emphysema), creating a radiographic pattern of serpiginous bubbles that can extend well into the lung periphery
(Fig. 50.35). Pneumomediastinum and pneumothorax are other common complications of positive pressure ventilation. Air can also dissect into the pericardium and peritoneum, and occasionally air embolism can develop, with devastating consequences. Early surfactant therapy has significantly reduced the incidence of these complications. The hypoxemia associated with the premature lung disease sometimes leads to persistent patency of the ductus arteriosus. Suggestive radiographic findings include lungs that are large and increasingly opaque, with loss of the granular pattern, cardiomegaly, and pulmonary vascular congestion (Fig. 50.36). The increased lung opacity represents pulmonary edema. Poor renal function and neurogenic pulmonary edema resulting from cerebral hypoxic injury and hemorrhage are common noncardiac causes of pulmonary edema in the premature infant.
(Fig. 50.35). Pneumomediastinum and pneumothorax are other common complications of positive pressure ventilation. Air can also dissect into the pericardium and peritoneum, and occasionally air embolism can develop, with devastating consequences. Early surfactant therapy has significantly reduced the incidence of these complications. The hypoxemia associated with the premature lung disease sometimes leads to persistent patency of the ductus arteriosus. Suggestive radiographic findings include lungs that are large and increasingly opaque, with loss of the granular pattern, cardiomegaly, and pulmonary vascular congestion (Fig. 50.36). The increased lung opacity represents pulmonary edema. Poor renal function and neurogenic pulmonary edema resulting from cerebral hypoxic injury and hemorrhage are common noncardiac causes of pulmonary edema in the premature infant.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree