25
Pelvic and Obstetric Hemorrhage
Life-threatening pelvic hemorrhage may result from blunt or penetrating trauma, pregnancy, malignancy, or arterial venous malformation. The pelvis is supplied by an extensive network of collateral vessels. Consequently, surgical ligation of the hypogastric artery origins has minimal effect on bleeding. Operative exposure and ligation of the bleeding distal hypogastric artery branches are a technical nightmare.1 Dissection of the retroperitoneum to find the hypogastric artery origins at laparotomy often exacerbates bleeding by removing compression of the bleeding site provided by adjacent anatomic structures. Because of technical difficulties posed by the surgical approach, angiographic embolization is the treatment of choice for pelvic arterial hemorrhage. Embolic materials released into the internal iliac artery are carried by the bloodstream to occlude the artery beyond the larger collaterals. The arterial pressure at the bleeding site then decreases sufficiently to permit hemostasis.
 Trauma
Trauma
Approach to the patient with pelvic fractures and shock: Stop the bleeding!
The patient with life-threatening pelvic hemorrhage may be in florid shock or may not appear to be seriously injured at all when he or she is brought to the emergency department. Sometimes, flank and buttock hematomas are present. Displaced pelvic fractures may cause obvious leg-length discrepancies,2 or no injuries may be visible. Blood pressure may be normal; baseline blood pressure varies from individual to individual. An adult can lose 30% of the blood volume acutely before systolic pressure begins to drop (although it then drops rapidly). Initially, the hematocrit is likely to be normal because it takes hours of equilibration with extravascular fluids before the serum hematocrit reflects an acute blood loss. Initially, the only abnormalities that may be observed are tachycardia (pulse > 100/min), tachypnea, coolness of the skin caused by peripheral vasoconstriction, a narrowed pulse pressure (actually an elevated diastolic pressure caused by catecholamine release), and low urine output. If the trauma patient has signs of shock after intravenous infusion of 2 L of fluid, it is most likely the consequence of active internal bleeding.3 The overall mortality from pelvic fractures is 6 to 19%, and half of these deaths are from hemorrhage and the others from associated injuries. If tachycardia is attributed to pain from pelvic fractures, the diagnosis of shock may not be made until systolic blood pressure falls. The mortality rate rises to 50% when hypotension is present.4 Hypotensive patients are treated with large volumes of intravenous crystalloid solution and packed red blood cells. If platelets and plasma are not infused as well, a dilutional coagulopathy develops. If the intravenous solutions are not warmed, the patient may become hypothermic. Hypothermia interferes with normal coagulation.5 Consequently, fluid resuscitation for hypotension may worsen the bleeding. Patients who are hypotensive for prolonged periods often develop multiple organ-system failure and sepsis.6 Therefore, it is essential to find the site of bleeding and to stop it before the patient’s condition deteriorates.
The trauma patient with active hemorrhage usually arrives in the emergency department with little clinical history but with many injuries. Complete evaluation of the patient requires numerous diagnostic tests. If the patient is to be saved, the sequence of tests must be prioritized so that potentially lethal injuries are addressed first and less dangerous (although frequently more obvious) injuries are evaluated later. The workup must proceed rapidly despite the fact that the patient may be hemodynamically unstable. Resuscitation and evaluation must take place simultaneously. Suspending the workup so that the patient can be “tuned-up” first will delay the time when a surgical or interventional procedure can be performed to stop the bleeding. Often, it is necessary to institute therapeutic procedures based on a probable diagnosis before a complete clinical picture is available.7,8
Diagnostic peritoneal lavage
The pelvic fracture patient with a patent airway in whom hemothorax, pneumothorax, cardiac contusion, and pericardial tamponade have been ruled out who shows signs of shock after vigorous intravenous fluid infusion is presumed to be actively bleeding. Supraumbilical diagnostic peritoneal lavage then is performed. If gross blood is found at lavage, it is likely that the bleeding emanates from a ruptured intraperitoneal organ (usually spleen or liver). The patient is taken for laparotomy. If the lavage does not reveal gross blood or if the patient remains unstable after laparotomy, ongoing retroperitoneal hemorrhage is probable. The patient then undergoes diagnostic pelvic angiography and possible embolization.9
Angiography and embolization
Hemorrhage from branches of the internal iliac artery requiring embolization occurs in 7 to 11% of patients with pelvic fractures.10 The most commonly injured arteries are the superior gluteal, iliolumbar, and lateral sacral (associated with sacral fracture and sacroiliac joint disruption), and the internal pudendal and the obturator arteries (associated with pubic ramus and acetabular fractures) (Fig. 25-1).11 Transcatheter embolization is successful in treating pelvic arterial hemorrhage in 85 to 90% of cases.6,12,13 There is great potential for cross-pelvic collateralization via anterior and posterior division branches of the internal iliac arteries. Therefore, selective injections of both internal iliac arteries should be performed, and bilateral embolization is often necessary.
External fixation
Venous bleeding is common after pelvic fractures. An extensive venous plexus exists adjacent to the pelvic bones and is more fragile than the pelvic arteries (Fig. 25-2). The venous bed is a low-pressure, but high-capacity, structure. The pelvic veins are valveless and are therefore in open communication with the inferior vena cava, and they are also in open communication with the portal vein via the superior rectal vein. In dogs simultaneous ligation of the lumbar arteries, inferior mesenteric artery, medial sacral artery, right and left hypogastric, external iliac, and profunda femoral arteries had no effect on pelvic venous pressure.14 Pelvic venous bleeding is stopped by the tamponade (compression) effect of surrounding structures. An unstable pelvic fracture (particularly of the open-book type) can greatly increase the potential volume of venous hemorrhage. A 3-cm diastasis of the pubic symphysis will increase the potential volume of the pelvis from the normal 4 L to 8 L.15 Realigning the fractured and displaced bones by placement of external fixators is an orthopedic procedure performed to control venous bleeding. External fixation is helpful when there is a diastasis of the pubic symphysis. It decreases the potential space in which bleeding can take place and aids coagulation by preventing the fracture fragments from moving in relation to each other.2 Angiographic embolization and external fixation treat different types of bleeding. External fixation controls osseous and venous bleeding but not arterial bleeding. Angiographic embolization controls arterial but not venous bleeding. At present, it is not possible to know initially whether a pelvic fracture patient is bleeding primarily from an arterial or a venous source; thus, which procedure should be performed first is a subject of controversy.
 Indications for Other Radiographic Studies in the Pelvic Fracture Patient
Indications for Other Radiographic Studies in the Pelvic Fracture Patient
Plain films
The only radiographs that an unstable blunt trauma patient should have are cervical spine, anteroposterior (AP) chest, and AP abdomen.3 These studies will identify most of the potentially lethal injuries that can be visualized by plain films. The patient may well have obvious extremity fractures and facial lacerations. Evaluation and treatment of these must wait until life-threatening injuries have been treated or ruled out. Foley et al.16 reported on four trauma patients who bled to death of ruptured spleens while plain films were being obtained in the emergency department.
Computed tomography scanning
Trauma patients with neurosurgical emergencies requiring immediate evacuation of intracranial hematomas usually present with one or more of the following: hypertension (not shock), bradycardia (not tachycardia), altered mental status (Glasgow coma scale of 8 or less), a dilated pupil on the side of the intracranial bleeding, and a peripheral neurological deficit on the side opposite the injury. For these patients, immediate computed tomography (CT) scan of the head is indicated.3
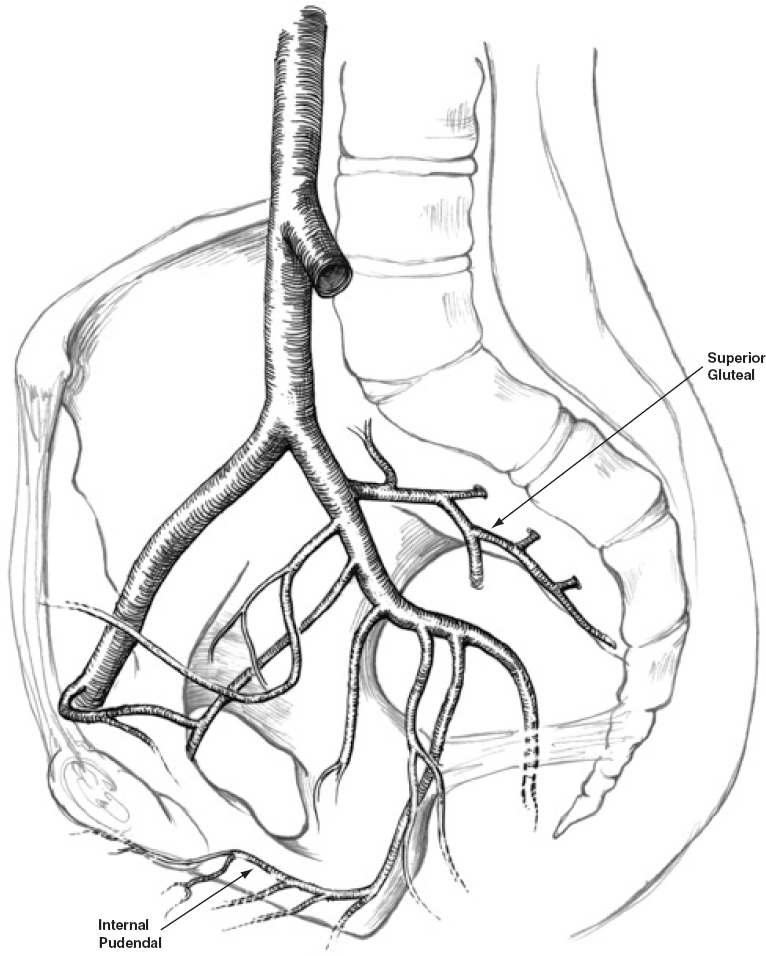
FIGURE 25-1. Internal iliac artery branches vulnerable to pelvic fractures. The superior gluteal and internal pudendal arteries are most commonly affected.
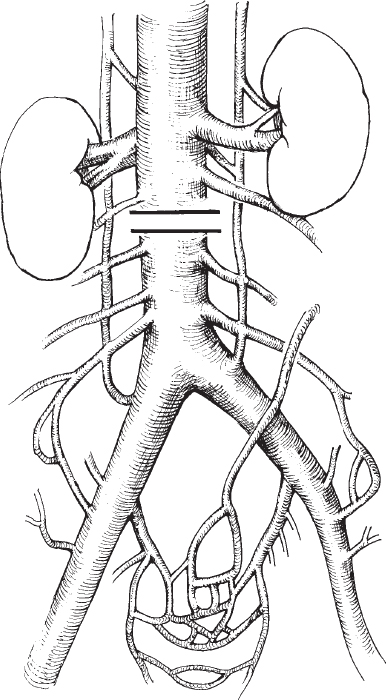
FIGURE 25-2. Pelvic venous plexus and collateral pathways.
Chest and body CT scanning is appropriate for evaluation of the hemodynamically stable trauma patient but in general should not be performed on the patient in shock. Oral contrast should not be used when there is a possibility that angiography may follow CT scanning. In the unstable patient with pelvic fractures and when findings on the chest film suggest great vessel injury, both thoracic aortography and pelvic angiography should be performed.10
Uroradiographic studies
A patient in shock must have a catheter to drain the bladder and to measure urine output. Urethral injury in females is rare. If a male with pelvic trauma is unable to void, has blood at the meatus, has a prostate that is high riding or is not palpable, or has a scrotal hematoma, he may have sustained a urethral injury. To avoid converting a partial urethral tear to a complete transection, urethrography should be performed before a Foley catheter is placed.17 A patient in shock should not undergo urethrography, cystography, or intravenous urography for two reasons: First, time is better spent stopping the bleeding than performing studies to evaluate injuries that are not life threatening; second, contrast that extravasates from an injured bladder may render pelvic angiography uninterpretable. Patients in shock who may have urethral injury and have an easily palpable bladder should have a suprapubic cystostomy placed at the bedside, which can be performed quickly and without contrast and is therapeutic. Many of these patients will require elective repair of urethral strictures after the urethral injury has healed.18
 Obstetric Hemorrhage
Obstetric Hemorrhage
Bleeding in excess of 1 L occurs in 1 to 2% of deliveries;19 11 to 13% of pregnancy-related maternal deaths in the United States are caused directly by hemorrhage, averaging 53 maternal deaths from bleeding each year.20 The pregnant uterus is extremely vascular. It is perfused by the uterine branches of the internal iliac arteries as well as by the ovarian arteries, which in most cases originate from the abdominal aorta (Fig. 25-3). The ovarian arteries may arise from the renal, iliac, lumbar, or adrenal arteries.21 The collateral blood supply to the uterus is excellent. Fatal obstetric hemorrhage can take place within minutes. Bilateral hypogastric artery ligation is successful in curtailing hemorrhage only 50% of the time. Bilateral uterine artery ligation is successful 95% of the time.19 Packing the uterus with gauze when coagulopathy is present and even hysterectomy occasionally fails to stop uterine hemorrhage. When medical management fails, angiographic embolization in many cases can obviate laparotomy and may preserve fertility by making hysterectomy unnecessary.22–26 When a patient is considered at high risk for postpartum bleeding, an angiographic catheter can be placed into the aorta through a left axillary approach while a lead apron shields the pelvis before the patient is taken to the operating room for cesarian section. Then intraoperative embolization can be performed using C-arm fluoroscopy if hemorrhage is severe.26,27 Because the placenta and the full-term uterus are extremely vascular structures, embolization to stop bleeding from a postpartum uterus or from an ectopic placenta frequently requires far larger quantities of Gelfoam than needed to control posttraumatic hemorrhage. Obstetric hemorrhage may be caused by bleeding from the placental implantation site, by trauma during delivery, or by the HELLP (hemolysis, elevated liver enzymes, and low platelet count) syndrome.
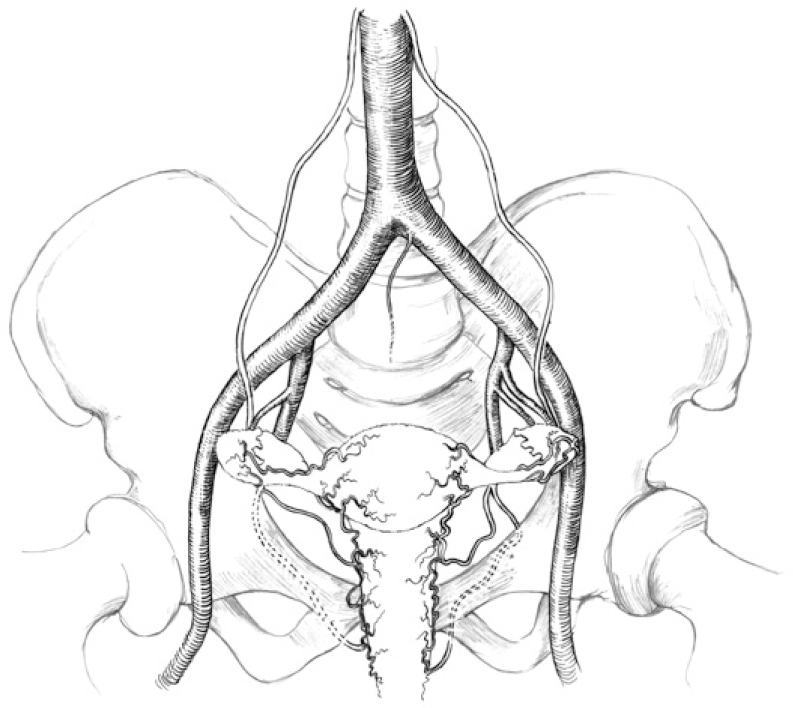
FIGURE 25-3. Arterial anatomy of the female reproductive organs.
Bleeding from the site of placental implantation
The arterial flow to the placenta at term is approximately 600 mL/min.20 When the placenta is expelled at delivery, the vessels connecting the uterus to the placenta are torn. Approximately 500 mL of blood is usually lost at this time. Contractions of the uterus occlude the disrupted vessels. If part of the placenta remains adherent to the uterus, bleeding will continue until the residual placenta is removed. If the uterus does not contract after delivery (atonic uterus), bleeding will persist. Atonic uterus usually responds to administration of oxytocin or, if this fails, to prostaglandin (15-methyl prostaglandin F2a).19 If the placenta attaches to the myometrium instead of to the endometrial decidua, it is called placenta accreta; if it invades deeply into the myometrium, placenta increta; and if it extends through the uterus and sometimes into adjacent organs, termed placenta percreta. Massive postpartum hemorrhage is probable in these situations because the abnormal placenta will not separate from the uterus at delivery. If the placenta separates from the uterus before labor begins (placental abruption), severe bleeding may occur and frequently is exacerbated by a coagulopathy caused by substances released by the placenta. Placental abruption associated with fetal distress or with continuing hemorrhage is treated by delivery of the fetus. If the placenta is near or covers the cervix (placenta previa
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree