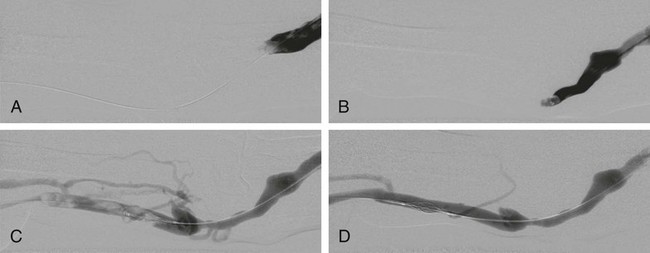
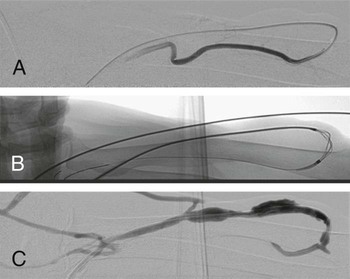
Percutaneous procedures for hemodialysis shunts are becoming increasingly important for interventional radiologists. A growing number of patients with renal insufficiency are enrolled in dialysis programs, the majority of them undergoing hemodialysis. In the West, this affects some 150 to 200 persons per million inhabitants. Given the increase in patients’ life expectancy, maintaining access to the vascular system continues to be a problem.1 For example, the number of long-term functioning shunts is estimated to be 15%.1 Primary patency of hemodialysis shunts is low: approximately 65% of Brescia-Cimino shunts and 50% of polytetrafluoroethylene (PTFE)-covered shunts exhibit primary patency after 1 year, and the numbers sink after 2 and 4 years to 60% and 45% for Brescia-Cimino shunts and to 43% and 10% for PTFE-covered shunts.1 Shunt thrombosis of native fistulas is a complication not as frequent as in grafts.1 Besides an underlying stenosis that is present in almost all cases, cofactors are manifold: clotting abnormalities, thrombocytosis, disturbed fluid balance, hypotension, and aneurysmatic degeneration of the native vein causing low flow may lead to shunt thrombosis. There are, however, different types of shunt thrombosis that require different strategies of percutaneous intervention. Indications for interventional treatment are acute or subacute occlusions in native fistulas that prevent use of the arteriovenous connection for dialysis. Relative indications are occluded fistulae in patients with a functioning renal allograft. Percutaneous intervention is contraindicated in acute infections of the vein or perivenous space. It is, however, worth trying to recanalize even thrombosed immature fistulas because there is a good chance of restoring flow and allowing development of a mature fistula after percutaneous recanalization.2,3 Natario et al. achieved clinical success in immature fistulas in 97%, with a 1-year primary patency of 51%.3 Miller et al. achieved 85% technical success in thrombosed immature fistulas, 79% clinical success, and a secondary patency of 90% at 12 months.2 In lesions near the anastomosis, retrograde puncture of the upper cephalic vein is performed at a position near the trunk. If the lesion is located more centrally, a puncture site is chosen close to the anastomosis (Figs. 117-1 and 117-2). A double access is often advisable in thromboses of upper arm shunts, whereby the puncture can be made in the thrombosed venous portion and thrombectomy first performed on one segment via retrograde access and then the second segment via antegrade access. Ipsilateral puncture of the internal jugular vein with retrograde probing has been described as an alternative access to the brachiocephalic vein.4 Published literature describes a number of approaches to treating clotted native fistulas: Frequently a combination of methods becomes necessary to finalize a case. All methods have been used with success, and interventional radiologists should be familiar with several of them to achieve best possible results. Thrombolysis is (relatively) infrequently described to treat native fistulas. Rajan et al.5 described their experience in 25 fistulas with 30 episodes of occlusion, including 19 forearm radiocephalic fistulas in 18 patients and 6 upper arm brachiocephalic fistulas in 6 patients. Lytic therapy with urokinase or recombinant tissue plasminogen activator (rtPA) was administered as a bolus into the fistula in 24 cases, with the exception of one case in which a 16-hour infusion of rtPA was initiated. Clinical success was achieved in 73% of cases (22 of 30). All patients were followed for a maximum of 66 months (mean 12 months). Primary patency rates were 36% at 3 months and 24% at 1 year. The assisted primary patency rate was 40% at 3 months and 32% at 6 months. The secondary patency rate stabilized at 3 months was 44%. Patency rates after clot removal were not significantly different between upper and lower arm fistulas.
Percutaneous Management of Thrombosis in Native Hemodialysis Shunts
Clinical Relevance
Indications
Technique
Anatomy and Approaches
Native Upper Arm Shunts
Technical Aspects
Percutaneous Treatment
Thrombolysis
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Percutaneous Management of Thrombosis in Native Hemodialysis Shunts