PET and PET-CT of Colorectal and Anal Carcinoma
Farrokh Pakzad
Jamshed B. Bomanji
Svetislav Gacinovic
Peter J. Ell
Colorectal cancer represents the commonest malignancy of the gastrointestinal tract. The application of positron emission tomography (PET) using fluorodeoxyglucose (FDG) has been frequently reported in the management of advanced and metastatic disease, and emerging data are continually redefining its clinical indications. The emergence of dual-modality imaging with integrated PET–computed tomography (CT) has further enhanced the power of imaging with PET, and the development of newer radiopharmaceuticals is bridging the gap between our understanding of cancer biology, from the bench to the patient.
The aims of this chapter are to review the most relevant current applications of FDG-PET, particularly PET-CT, in colorectal cancer. The use of some of the newer PET radiopharmaceuticals will also be outlined. Currently, there is little evidence supporting the use of FDG-PET in anal cancer. Yet, because of the recent overhaul in the management of this relatively uncommon malignancy, the potential use of FDG-PET will also be briefly discussed.
Colorectal Cancer
Background
Worldwide, an estimated 1 million cases of colorectal cancer were diagnosed in 2002. This accounts for more than 9% of all newly diagnosed cancers. Around 372,000 cases occur each year in Europe and 166,000 in the United States. In eastern Europe, the incidence of colorectal cancer (CRC) has been on the increase, while remaining stable in western Europe. The disease is commoner in patients over the age of 65 years, and although overall there is a slight male preponderance, over the age of 65 years this difference becomes negligible.
In the United Kingdom, approximately 55% of patients with CRC present at initial diagnosis with advanced disease (Dukes stages C and D). Of those with earlier disease who receive a curative resection, some 30% to 40% still go on to develop a local recurrence or metastatic disease. Therefore, early diagnosis and accurate staging represents the principal determinant of successful management of CRC.
Currently, the gold standard in diagnosing primary disease is endoscopy. This is followed by computed tomography (CT), which is routinely used for primary staging, surveillance for metastases, and detection of recurrent disease. Other conventional imaging tools such as ultrasound, magnetic resonance imaging (MRI), and, more recently, CT pneumocolonography are routinely applied according to institutional preferences and availability. Overall, however, the indications are that conventional imaging modalities seem to play a suboptimal role in the management of colorectal cancer, and hence much attention has been focused on molecular imaging techniques such as positron emission tomography (PET).
PET offers the clinician both an alternative and complementary means of assessing the gastrointestinal tract. Although fluorodeoxyglucose (FDG) has been the most successful and widely used PET radiopharmaceutical in
oncology, molecular profiling with alternative tracers and the advent of dual-modality PET-CT are reshaping the way we manage colorectal cancer.
oncology, molecular profiling with alternative tracers and the advent of dual-modality PET-CT are reshaping the way we manage colorectal cancer.
Detection of Premalignant Colonic Lesions
Adenomatous polyps are benign neoplasms of colonic mucosa. In asymptomatic patients of average risk, its prevalence is approximately 10% from sigmoidoscopy and 25% from colonoscopy series (1). Worldwide, however, this figure varies among different countries. Colonic adenomas are well established as precursors of CRC (2), where the prevalence in these patients is approximately 1%. Barium enema and colonoscopy represent the gold standard for detecting these lesions, the later offering therapeutic as well as diagnostic benefits. More recently, CT pneumocolonography has also emerged as an alternative and accurate way of detecting polyps in patients unsuitable for colonoscopy.
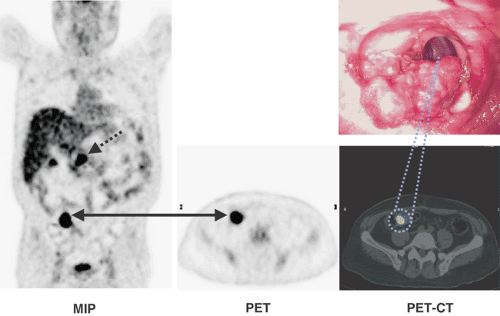
Adenomatous polyps exhibit an enhanced glycolytic activity, and therefore the incidental detection of polyposis has been reported with FDG-PET (3). In a retrospective study completed by Yassuda et al. in 2001, FDG-PET was shown to have a true positive rate of only 24% (3). This figure did rise to 90%, particularly in polyps greater that 13 mm in size, suggesting that smaller polyps (less than 1 cm) may not be accumulating enough FDG to be detected at the resolution limit of PET. Although PET-CT may help in the localization of intraluminal lesions, the size and FDG avidity of the polyps would remain a limiting factor (Fig. 44.1).
Overall, while FDG-PET and PET-CT are not indicated for the routine detection of colonic polyps, focal FDG accumulation in the colon should not be ignored.
Screening
To date, the use of FDG-PET in screening for CRC has been reported by one study of 3,600 patients (4). Here, the prevalence of CRC was 2.1%, and FDG-PET had a true-positive rate and false-negative rate of 54% and 46%, respectively. These figures, coupled with the cost, availability, and radiation exposure risk of 18F-FDG, do not justify the routine use of FDG-PET and FDG PET-CT for screening purposes.
Role of FDG-PET in the Diagnosis of Primary Colorectal Cancer
The role of FDG-PET in routine preoperative diagnosis of primary CRC has only been reported in small patient
numbers. Abdel Nabi et al., in 1998, reported FDG-PET to have a sensitivity and specificity of 100% and 48% (N = 48) (5). Similarly, in a 2000 study, Mukai et al. reported a true-positive rate of 98.5% (N = 24) (6). False positives often arise due to focal areas of FDG accumulation in, for example, diverticulitis or adenomatous polyps. Where physiological tracer uptake in the bowel is encountered, lack of anatomical localization with PET can also lead to diagnostic uncertainty. This has been addressed with PET-CT, and although its clinical impact in primary CRC has not yet been conclusively evaluated, early results have shown that PET-CT can improve the certainty of lesion characterization by 30% and the accuracy of staging by 11% (7).
numbers. Abdel Nabi et al., in 1998, reported FDG-PET to have a sensitivity and specificity of 100% and 48% (N = 48) (5). Similarly, in a 2000 study, Mukai et al. reported a true-positive rate of 98.5% (N = 24) (6). False positives often arise due to focal areas of FDG accumulation in, for example, diverticulitis or adenomatous polyps. Where physiological tracer uptake in the bowel is encountered, lack of anatomical localization with PET can also lead to diagnostic uncertainty. This has been addressed with PET-CT, and although its clinical impact in primary CRC has not yet been conclusively evaluated, early results have shown that PET-CT can improve the certainty of lesion characterization by 30% and the accuracy of staging by 11% (7).
Conventional anatomical imaging modalities remain the cornerstone for staging CRC. Local (T staging) of the tumor cannot be reliably carried out with PET due to its limited resolution and the effect of partial-volume averaging of the FDG signal. Although at present PET-CT imaging protocols limit the diagnostic capability of its CT component, future developments may allow PET-CT to confer advantages over CT alone or MRI alone.
In terms of lymph node staging (N staging), the sensitivity of FDG-PET has been shown to be similar to that of CT and generally poor (22% to 29%) (5,6). As the presence of lymph node metastases on CT are detected according to size criteria, image fusion with PET-CT can help further confirm the presence of disease, even in nonenlarged lymph nodes. A pitfall of this, however, is that false positives could result from the possible accumulation of 18F-FDG in reactive lymph nodes.
In preoperative staging, FDG-PET has performed best in detecting hepatic and extra-abdominal metastases. Sensitivities of 88% to 91% and specificities of 91% to 100% have been reported, as compared with 38% and 97% for CT, respectively (5,8). This can lead to a change in treatment modality and the extent of surgery (8).
Overall, the indications are that FDG-PET or PET-CT play a limited role in the routine diagnosis and staging of primary CRC. However, in high-risk patients, its accuracy in detecting extracolonic disease may result in the avoidance of futile surgery.
FDG-PET and PET-CT in the Detection of Recurrent and Metastatic Disease
Locoregional Recurrence
After apparent curative resection of the primary tumor, the recurrence rate for colorectal cancer is between 30% and 40%, with the majority of recurrences occurring within the first 3 years after surgery (9,10). Of these, approximately 25% are isolated locoregional recurrences, which may be amenable to resection. In order to minimize unnecessary morbidity and mortality from surgical treatment, accurately identifying the extent of recurrent disease is vital for appropriate patient selection.
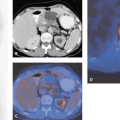
The role of FDG-PET in detecting recurrent and metastatic colorectal cancer has been demonstrated by several authors (11,12,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31) (Table 44.1 and Fig. 44.2). A particular challenge of anatomical imaging modalities has been to differentiate scar from local recurrence. The reported accuracy of FDG-PET in detecting pelvic recurrences is 95%, as compared with 65% for CT (25). In particular, Valk et al. demonstrated the sensitivity and specificity of FDG-PET to be 93% and 98%, whereas for CT they were 69% and 96%, respectively. This led to significant cost savings from the avoidance of futile surgical intervention (28). Image fusion with PET-CT further adds to the diagnostic accuracy of 18F-FDG-PET. It is particularly effective in characterizing a presacral mass, with a quoted sensitivity of 100% and a quoted specificity of 96% (32).
Intensive follow-up after treatment with regular imaging (ultrasound or CT) and tumor marker levels has consistently been shown to correlate with improved survival figures. Measurement of serum carcinoembryonic antigen (CEA) levels is a simple method that can give a first indication of tumor recurrence in approximately 60% of cases, with a mean sensitivity of 80% (range, 17% to 89%). However, up to 30% of patients have been reported not to express the antigen.
One of the areas where FDG-PET plays a significant role is in the assessment of patients with rising CEA levels and negative conventional imaging (Fig. 44.3). Flanegan el al. showed FDG-PET to have positive and negative predictive values of 89% and 100%, respectively (16). Flamen et al. also reported similar findings, but their false-positive rate of 21% was significant (15). The question remains whether all patients with a rising CEA should receive an FDG-PET scan. A pitfall of this approach is the false-positive rate of CEA (5% to 16%). On the other hand, routine 18F-FDG PET-CT as a first-line imaging modality, followed by further investigation guided by PET-CT, is also a valid strategy (33,34). This paradigm requires in-depth examination.
Metastatic Disease
The principal advantages of FDG-PET are that it is a whole-body modality and that biological signals of disease can be detected before morphological changes become apparent. Detection of these signals often results in the detection of unsuspected metastases, which can occur in 13% to 36% of cases and can have a clinical impact in 14% to 65% (12,15,16,19,22,23,24,25,26,35,36). In whole-body imaging, Valk et al. showed the sensitivity of FDG-PET to be higher than that of CT at all sites except for the lungs, where the two were equivalent (28). The differences in detection rate were greatest in the abdomen, pelvis, and retroperitoneum, where almost 30% of lesions missed on CT were detected with 18F-FDG-PET. Lai et al. also reported similar findings, but their study suggested FDG-PET to be superior in the lungs (22). The significant advantage of dual-modality PET-CT imaging is that it aids in differentiating areas of pathology and normal uptake, which is especially relevant to the issues mentioned in this paragraph.
Table 44.1 Tabulated summary of published data comparing detection rates of fdg-pet and ct imaging of colorectal liver metastases. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree