PET and PET-CT of Esophageal and Gastric Cancers
Jamshed B. Bomanji
Farrokh Pakzad
Darren L. Francis
Peter J. Ell
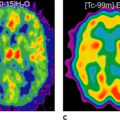
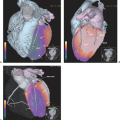
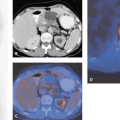
The incidence of gastroesophageal cancers, particularly the incidence of cancer of the esophagus, has been increasing in the Western world. Despite many advances in management, the prognosis from these tumors has remained poor. Currently, anatomical imaging modalities such as computed tomography (CT) and endoscopic ultrasound (EUS) are routinely used in diagnosing and staging esophageal and gastric cancers. However, they have several shortcomings with regard to demonstrating the full extent of disease and in predicting response to therapy. Biological imaging with fluorine 18 fluorodeoxyglucose (18F-FDG) positron emission tomography (PET) and more recently dual modality PET-CT offers significant advantages. The aim of this chapter is to review the available evidence for the continually expanding role of imaging with PET and PET-CT in these conditions (see Fig. 41.1).
Background
Esophageal Cancer
The incidence of carcinoma of the esophagus has been increasing in the Western world at a rate exceeding that of all other types of cancer (1). Geographically, the high risk areas are known as the “Asian esophageal cancer belt,” which runs through Turkey, the northern provinces of Iran, central Asia, and China and where the incidence of esophageal cancer reaches as high as 150 per 100,000 population (2). It is a cancer that is more common between the ages of 50 and 70 years, with a male to female ratio of 3:1.
Esophageal cancer can be divided into two major histological types, squamous cell carcinoma and adenocarcinoma. The incidence of adenocarcinoma has risen dramatically and is now the most prevalent histological type in western Europe and the United States (3). Accompanying this increase, there has been a shift in the presentation of tumors at the distal esophagus and gastroesophageal junction (4). Squamous cell carcinomas are largely attributed to alcohol, tobacco, and nitrogen compounds, whereas adenocarcinomas are strongly associated with gastroesophageal reflux disease and Barrett esophagus. This has led to recommendations that patients with chronic symptoms that might lead to Barrett esophagus undergo endoscopic screening (5).
Over the last 3 decades, there has been little improvement in the long-term survival from esophageal carcinoma (6,7). Early surgical resection provides the only potential for long-term survival or even cure, an approach that despite its significant morbidity is increasingly advocated in older patients (8). However, up to 50% of patients have metastatic disease either at initial presentation or following apparent curative local therapy. In those who are treated surgically, the primary cause of death is disease recurrence. Clinical stage of the disease is a strong determinant of long-term survival, with 5-year survival decreasing dramatically when the disease is upstaged from stage I–III. In patients with stage IV disease, the 5-year survival is virtually zero (4).
Regional lymph node involvement is a major prognostic factor in esophageal cancer. The presence of lymph node metastases has been shown to be associated with poorer survival (5-year survival of 10% to 12%, compared with 42% to 72% in node-negative disease) (9). Lymph node staging of esophageal cancer is, however, complicated by the rich network of lymphatics that drain the esophagus. For instance, cervical and supraclavicular lymph node chains tend to drain the cervical esophagus, and in tumors of the distal esophagus and gastroesophageal junction, perigastric, celiac, or even para-aortic lymph nodes may be involved.
The distribution of metastases can also be widespread. This has been demonstrated by a 1995 study by Quint et al., who looked at the distribution of metastases in 838 newly diagnosed cases of esophageal cancer (10). Overall, metastases were found in 18% of the cases, of which 75% were preoperatively detected by imaging and/or clinical findings. Table 41.1 summarizes their findings. It was interesting to note that in those with a negative CT of the abdomen and chest, no additional metastases were detected on CT brain or bone scintigraphy. Therefore, the distribution of metastases, being mainly thoracoabdominal, led them to conclude that imaging of the abdomen and chest would be an effective screening method for M1 disease prior to an operation.
Gastric Cancer
Gastric cancer remains the second most common cause of cancer-related death in the world. Most patients are elderly, and the median age at diagnosis is 65 years (range, 40 to 70). As with esophageal cancer, the incidence of gastric cancer is high in many Asian countries.
Table 41.1 METASTASES DETECTED AT INITIAL STAGING OF ESOPHAGEAL CANCER | ||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||
Histologically, 90% to 95% of gastric cancers are adenocarcinomas. Early-stage gastric cancers tend to be asymptomatic, which is the predominant reason why only 10% to 20% are diagnosed early. The remainder present with stage III or IV disease, and gastric cancers at these stages have an overall 5-year survival rate of no greater than 15% to 20%. Tumors arising in the distal stomach have a slightly better prognosis (5-year survival of approximately 55%). However, the involvement of regional nodes reduces this to 20%. Following a curative gastrectomy, staging and lymph node involvement have also been implicated as strong independent predictors of early recurrence (11).
Tables 41.2 and 41.3 summarize the universally accepted staging system for esophageal and gastric cancers (12). The prognostic significance of this system has been supported by the German Gastric Cancer Group (13). It is at this level of patient assessment where historically most difficulty has arisen. Thus the direction of treatment toward either surgery or chemoradiotherapy depends on accurate staging of the disease.
Table 41.2 STAGING OF ESOPHAGEAL CANCER | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 41.3 STAGING OF GASTRIC CANCER | |||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||||||||||||||||||
Staging
Initial Staging
The key aims of staging both esophageal and gastric cancer are
To determine recurrence risk by assessing the locoregional (T and N) stage, and
To rule out distant metastases.
The gold standard method for the diagnosis of carcinoma of the esophagus and stomach is endoscopy. It offers both direct visualization of the tumor and the ability to acquire a histological diagnosis. There is limited data on the safety and efficacy of intensive endoscopic surveillance for patients with high-grade dysplasia, and no study has established that endoscopic screening and surveillance programs for Barrett esophagus decrease the rate of death from cancer (5). Fluorodeoxyglucose (FDG) positron emission tomography (PET) currently plays no established role in diagnosis but may have a use in the detection of the severe dysplasia that may be found in areas of Barrett esophagus.
Although endoscopy alone provides superb visualization of the mucosa, it does not provide information regarding the depth of tumor invasion or the extent of metastatic disease. This is where endoscopic ultrasound (EUS) is emerging as a powerful tool for accurate assessment of the luminal wall and local lymph node involvement (14,15,16).
Computed tomography (CT) is currently the most frequently used initial staging tool (17,18,19,20,21). Anatomical imaging with CT, however, has its limitations, as size criteria, such as that used for characterizing lymph nodes, cannot reliably differentiate benign from malignant disease. This is also a particular problem where small nodules in the liver and lung are encountered on CT (22,23). Staging laparoscopy is another tool that has gained favor as an adjunct to CT, and several studies have confirmed its accuracy compared to conventional imaging (24,25). Two prospective randomized trials have in fact shown a higher sensitivity in detecting small distant metastases than achievable with CT (23,26). In addition to staging, laparoscopy can aid palliation of patients with inoperable tumors by avoiding an unnecessary laparotomy (27).
Staging of Recurrent Disease
Development of recurrent gastric or esophageal cancer is nearly always fatal. It has been argued that early detection combined with aggressive treatment may offer improved survival in esophageal cancer (28). However, there seems to be no such evidence for gastric cancer, as current treatment options for recurrent disease do not offer any significant survival benefit (29,30,31). That said, in the majority of cases, detection and restaging of recurrent disease is performed using CT and, more recently, EUS. However, both these approaches are limited by their reliance on structural change. This, together with technical difficulties in performing endoscopy, invariably leads to loss of accuracy.
PET
The fact that detection is also dependant on the signal intensity of metabolically active tissue (as opposed to lesion size only) gives PET the unique ability to detect functional or metabolic changes prior to the structural changes observed. This in turn has important clinical advantages with regard to diagnosing, staging, and prognosticating the disease. In this section, the role of PET with FDG will be reviewed. Currently, the breadth of evidence in this field is narrow, and data from series using PET-CT scanners are not yet available. Where possible, however, references will be made to the potential advantages of dual-modality imaging.
Esophageal Cancer
Detection and Staging of Primary Disease
In esophageal cancer, FDG-PET has been consistently demonstrated to be highly sensitive for detecting both adenocarcinomas and squamous cell carcinomas (32,33,34,35). In Block’s study of 58 patients with histologically confirmed tumors, PET
failed to detect the disease in only 2 patients, who subsequently were found to have small and early T-stage tumors (32). In 2000, Flamen et al. reported a sensitivity of 95% for primary tumor visualization in a cohort of 74 patients (36). Once gain, false negatives were associated with small T1 lesions. In 2001, in a tabulated summary of published data on 277 patients and 168 patient studies, Gambhir et al. showed FDG-PET to be 96% sensitive, compared with 81% for CT. On a lesion-by-lesion basis (545 lesions), the sensitivity, specificity, and accuracy of 18F-FDG-PET was 80%, 95%, and 86%, compared with 68%, 81%, and 73% for CT (37).
failed to detect the disease in only 2 patients, who subsequently were found to have small and early T-stage tumors (32). In 2000, Flamen et al. reported a sensitivity of 95% for primary tumor visualization in a cohort of 74 patients (36). Once gain, false negatives were associated with small T1 lesions. In 2001, in a tabulated summary of published data on 277 patients and 168 patient studies, Gambhir et al. showed FDG-PET to be 96% sensitive, compared with 81% for CT. On a lesion-by-lesion basis (545 lesions), the sensitivity, specificity, and accuracy of 18F-FDG-PET was 80%, 95%, and 86%, compared with 68%, 81%, and 73% for CT (37).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree