PET and PET-CT of Non-Small Cell Lung Cancer
Hans C. Steinert
Lung cancer is the most common cancer in men worldwide and mostly caused by smoking. The risk decreases with cessation of smoking but never disappears. However, the majority of smokers do not develop lung cancer, demonstrating the role of genes and other factors. Among the common cell types of lung cancer are adenocarcinoma, including bronchioloalveolar carcinoma (50%); squamous cell carcinoma (30%); small cell carcinoma (15%); and large cell carcinoma (5%). In non-small cell lung cancer (NSCLC), surgical resection offers the best chance of cure in the early stages, either alone or in combination with chemo- or radiotherapy at the more advanced stages. However, most patients present with metastatic disease at the time of diagnosis.
Both computed tomography (CT) and positron emission tomography (PET) using fluorodeoxyglucose (FDG) play an important role in the diagnosis and staging of patients with lung cancer. CT provides excellent morphological information but has significant limitations in differentiating between benign and malignant lesions either in an organ or in lymph nodes. It has been shown that FDG-PET is highly accurate in classifying lung nodules as malignant or benign. Whole-body PET improves the rate of detection of mediastinal lymph node metastases as well as extrathoracic metastases when compared with CT, magnetic resonance imaging (MRI), ultrasound, and bone scanning. Since good PET scanners have a fairly high resolution (less than 6 mm), even small lesions can be detected. Limitations are that FDG is not specific for malignant tissue and that FDG-PET provides little information on the exact anatomic localization of lesions.
Our results show that integrated PET-CT provides more than the sum of PET and CT (see Fig. 38.4). In particular, it improves T staging. Due to precise CT correlation with the extent of FDG uptake, the location of the primary tumor can be exactly defined, and thereby diagnosis of chest wall infiltration and mediastinal invasion by the tumor is improved.
Classification and Staging
Non-small cell lung cancer (NSCLC) includes adenocarcinoma, squamous cell carcinoma, and large cell carcinoma. Adenocarcinomas typically develop in the periphery of the lung and are most common in women and in nonsmokers! Adenocarcinomas have a high incidence of early metastases and tend to grow more rapidly than squamous cell carcinomas. Bronchioalveolar cell carcinoma is a subtype of adenocarcinoma. It typically grows along the alveolar spaces without invasion of the stroma. It can appear as a solitary pulmonary nodule, a pneumonia-like consolidation, or multiple nodules throughout the lung. Squamous cell carcinomas are strongly associated with smoking. In general, they have the best prognosis because of their slow growth rate and their low incidence of distant metastases. They tend to become large and develop a central necrosis. Metastases to regional lymph nodes are common. Squamous
cell carcinoma is the most common cause of Pancoast tumors. These occur typically at the apex of the lung and are associated with Horner syndrome and local bone destruction. Large cell carcinomas are also strongly associated with smoking. They tend to grow rapidly and metastasize early and are associated with a poor prognosis.
cell carcinoma is the most common cause of Pancoast tumors. These occur typically at the apex of the lung and are associated with Horner syndrome and local bone destruction. Large cell carcinomas are also strongly associated with smoking. They tend to grow rapidly and metastasize early and are associated with a poor prognosis.
Table 38.1 The Tnm Staging System | |||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||||||||||||
The TNM staging system is widely used to classify NSCLC. The last revision of the staging system was approved by the American Joint Committee on Cancer and the International Union against Cancer in 2002 (1) (Table 38.1). NSCLC is also classified into four stages (1). Stage IV includes those patients with evidence of distant metastasis and those with one or more separate metastatic tumor nodules in the ipsilateral non-primary-tumor lobes of the lung. Stage III is divided in two substages, stage IIIA and stage IIIB. Only stage IIIB tumors are considered unresectable. Tumors with limited invasion of the chest wall and mediastinum are included in the operable stage IIIA (Table 38.2). Stage T4 is used for extensive invasion of the mediastinum or diaphragm (e.g., invasion of the great vessels, heart, trachea, or esophagus). N1 disease encompasses peribronchial and ipsilateral hilar metastases, including direct extension. All N1 nodes lie distal to the mediastinal pleural reflection and within the visceral pleura (2). These patients are considered resectable. However, the presence of ipsilateral hilar lymph node metastases decreases the overall survival rate. N2 disease encompasses ipsilateral paratracheal and/or subcarinal lymph node metastases. These patients have potentially resectable disease. Today, at our institution, most patients with N2 disease receive neoadjuvant chemotherapy for the reduction of the tumor mass before surgery. N3 disease encompasses contralateral mediastinal nodal metastases, contralateral hilar nodal metastases, and ipsilateral or contralateral scalene or
supraclavicular nodal metastases. N3 disease is considered unresectable.
supraclavicular nodal metastases. N3 disease is considered unresectable.
Table 38.2 Classification Into Stages | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||
Solitary Pulmonary Nodule
Lung masses have been evaluated traditionally by using planar chest x-rays, computed tomography (CT), and, more recently, magnetic resonance imaging (MRI). Some radiographic parameters such as calcifications and smooth margins of a lesion may indicate a lower likelihood that a solitary nodule is malignant, although a substantial percentage of nodules remain radiographically indeterminate. Factors that increase the probability of malignancy include size, absence of calcification, and irregular margins. Unchanged radiographic appearance of a nodule at follow-up examinations over a 2-year period imply that the lesion is benign. Definite diagnoses are established with invasive techniques such as bronchoscopy, mediastinoscopy, and biopsy. In the case of benign lesions, nondiagnostic biopsy results are not uncommon.
The ability of positron emission tomography (PET) to distinguish between benign and malignant lesions is good but not perfect. For benign lesions, Patz et al. (3) demonstrated a very high specificity for specificity for FDG-PET. Gupta et al. (4) showed that FDG-PET is highly accurate in differentiating malignant from benign solitary pulmonary nodules for sizes from 6 to 30 mm when radiographic findings are indeterminate. In a series of 61 patients, PET had a sensitivity of 93% and a specificity of 88% for detecting malignancy. PET is also effective in detecting recurrence after previous lung resection or irradiation (5). However, FDG-PET may show negative results for pulmonary carcinoids and bronchioloalveolar lung carcinoma (6,7) (Fig. 38.1).
PET is clinically useful in patients with a solitary pulmonary nodule less than 3 cm in diameter, especially where biopsy may be risky or where there is a low risk of malignancy based on the history or radiographic findings. Lesions with low FDG uptake can be considered to be benign and can be monitored with chest radiographs. Lesions with increased FDG uptake should be considered malignant, although false-positive results have been reported in cases of inflammatory and infectious processes, such as histoplasmosis, aspergillosis, and active tuberculosis. With integrated PET-CT, additional certainty regarding the presence or absence of FDG uptake in the pulmonary nodule can be achieved because morphologic CT criteria and functional CT criteria are available simultaneously.
T Staging
CT is an important imaging modality for the evaluation of the primary tumor. However, the low accuracy of chest CT in the evaluation of invasion of the chest wall or involvement of the mediastinum and the correct differentiation between tumor and peritumoral atelectasis often prevents precise T staging with CT (8). PET, on the other hand, has the disadvantage of having limited anatomical resolution, which makes the assessment of tumor extension unreliable, particularly if the tumor infiltrates the chest wall or the mediastinum.
It has been demonstrated that integrated PET-CT is a useful tool for the detection of tumor invasion into the chest wall (9,10,11). Due to the exact anatomic correlation to FDG uptake, the primary tumor can be delineated precisely (Fig. 38.2). Therefore, the diagnosis of chest wall infiltration and mediastinal invasion by the tumor is improved. Lesions with chest wall infiltration are classified as stage T3 and are potentially resectable. Surgical treatment requires en bloc resection of the primary tumor and the contiguous chest wall. Particularly in patients with poor cardiopulmonary reserve, the preoperative determination of chest wall infiltration is desirable in order to avoid extended en bloc resection.
Integrated PET-CT provides important information on mediastinal infiltration as well. Contrast-enhanced CT to evaluate infiltration of hilar and vessels has a relatively low sensitivity, specificity, and accuracy (68%, 72%, and 70%, respectively) (12). Our experience suggests that unenhanced PET-CT imaging is unable to identify direct invasion of the walls of vital mediastinal structures, and this is the scan protocol we currently use in most patients receiving an integrated PET-CT scan. Further research is necessary to evaluate the potential additional diagnostic impact of intravascular contrast agents when performing integrated PET-CT (Fig. 38.3).
It has been shown that FDG-PET is a useful tool for the differentiation of tumor and peritumoral atelectasis (Fig. 38.3). This is particularly important for the planning of radiotherapy in patients with lung cancer associated with an
atelectasis. The information provided by FDG-PET contributes to a change in the radiation field in approximately 30% to 40% of patients (13).
atelectasis. The information provided by FDG-PET contributes to a change in the radiation field in approximately 30% to 40% of patients (13).
N Staging
Morphologic Imaging
Accurate mediastinal staging is essential for the management of patients with NSCLC. Surgical resection is the treatment of choice for early stages of NSCLC. Patients with ipsilateral mediastinal lymph node metastases (N2 disease) are considered to have potentially resectable disease. If contralateral mediastinal lymph node metastases (N3 disease) are present, surgery is generally not indicated. CT and MRI have substantial limitations in depicting mediastinal lymph node metastases (Fig. 38.4). The only CT and MRI criteria for tumor involvement are morphologic; that is, the criteria rely on the size and shape of the lymph nodes. However, normal-size regional lymph nodes may prove to have metastases upon histologic examination, and nodal enlargement can be due to reactive hyperplasia or other nonmalignant conditions. The sensitivity and specificity of determining lymph node metastases for NSCLC by CT are both 60% to 70% (8,14). Thus, in 30% to 40% of cases, CT scanning will erroneously
suggest the presence of mediastinal lymph node metastases and will miss lymph node metastases in 30% to 40% of cases
suggest the presence of mediastinal lymph node metastases and will miss lymph node metastases in 30% to 40% of cases
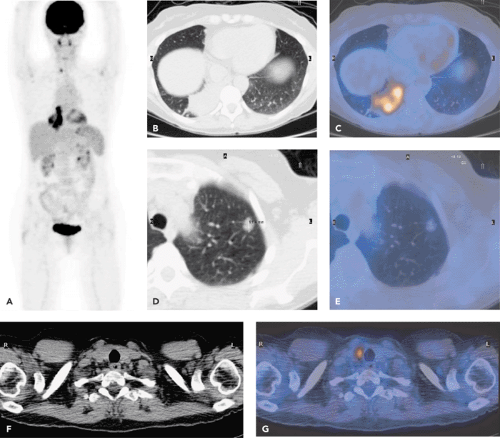
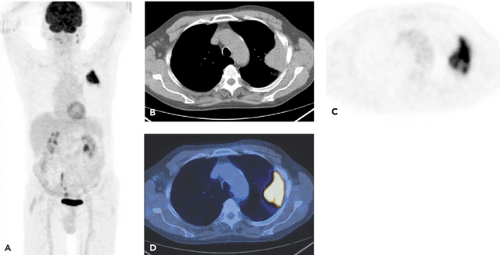 Figure 38.2 A 63-year-old man. Conventional CT demonstrated a lung cancer in the left upper lobe. Invasion of the tumor into the chest wall could not be ruled out. PET-CT was performed for staging. Images include a MIP scan (A), an axial CT scan (B), an axial attenuation-corrected PET scan (C), and an axial coregistered PET-CT scan (D). Diagnosis based on the integrated PET-CT imaging: Lung cancer in the left upper lobe. No invasion into the chest wall. No ipsilateral or contralateral mediastinal lymph node metastases. No extrathoracic metastases. Surgery confirmed that the tumor did not invade the chest wall. (See also Cases 7 and 11.)
Stay updated, free articles. Join our Telegram channel
Full access? Get Clinical Tree
 Get Clinical Tree app for offline access
Get Clinical Tree app for offline access

|