Etiology, Prevalence, and Epidemiology
Pleural effusion is the accumulation of fluid in the pleural space resulting from disruption of the homeostatic forces responsible for the movement of pleural fluid. Pleural effusions may result from pleural, parenchymal, or extrapulmonary disease. Approximately 1 million people develop this abnormality each year in the United States.
Clinical Presentation
The oncotic pressures on both sides of the parietal and visceral pleura are normally equal, but an inequality exists in the hydrostatic pressures between the systemic circulation that supplies the parietal pleura and the low-pressure pulmonary circulation that supplies the visceral pleura. One fundamental component of the lymphatic anatomy is the existence of naturally occurring pores (stomas) in the caudal portion of the peripheral parietal pleura and lower mediastinal pleura. These pores are capable of transferring particulate matter and cells directly into lymphatic channels for removal. Most of the fluid that accumulates abnormally in the pleural space is derived from the lung through the visceral pleura and absorbed primarily through the parietal pleura.
Transudative effusions are mostly caused by an imbalance of hydrostatic and oncotic forces ( Table 72.1 ). It can also occur because of increased negative pleural pressure (atelectasis) or movement of intraabdominal fluid into the pleural cavity (mostly on the right side) through diaphragmatic pores. These effusions have low protein levels and show low specific gravity. Exudative effusions usually result from pleural diseases or from injury in the adjacent lung and can be due to an increase in capillary permeability, as in pneumonia, or decreased lymphatic drainage, as in mediastinal carcinomatosis ( Table 72.2 ). These effusions typically display high protein and lactate dehydrogenase (LDH) levels and an elevated specific gravity.
| Common | Less Common | Rare |
|---|---|---|
| Left heart failure | Nephrotic syndrome | Constrictive pericarditis |
| Cirrhosis | Mitral stenosis | Superior vena cava obstruction |
| Hypoalbuminemia | Hypothyroidism | Urinothorax |
| Meigs syndrome | ||
| Ovarian hyperstimulation syndrome |
| Common | Less Common | Rare |
|---|---|---|
| Malignancy | Pulmonary embolism | Drugs |
| Parapneumonic effusions | Rheumatoid arthritis | Yellow nail syndrome |
| Benign asbestos effusion | ||
| Dressler syndrome | ||
| Pancreatitis |
Traditionally, exudates are distinguished from transudates by measuring the pleural fluid protein concentration. Exudates have a protein level greater than 30 g/L, and transudates have a protein level less than 30 g/L. Care should be taken in interpreting the pleural protein concentration if the serum total protein level is abnormal. In these cases the distinction is based on the ratio of pleural to serum protein levels and measurement of serum and pleural fluid LDH. The effusion is considered to be an exudate when the pleural fluid protein level divided by serum protein level is greater than 0.5, when pleural fluid LDH divided by serum LDH is greater than 0.6, and when pleural fluid LDH is more than two-thirds the upper limit of normal serum LDH.
Manifestations of the Disease
Radiography
Small pleural effusions are not readily identified on conventional chest radiographs. When approximately 175 mL of fluid accumulates in the pleural space, there may be a noticeable blunting of the lateral costophrenic angle on erect frontal views; however, 525 mL of fluid occasionally may be required before the blunting of the angle is apparent ( Box 72.1 ). A retrospective analysis of 71 patients who had chest computed tomography (CT) scans and posteroanterior and lateral chest radiographs showed that pleural fluid becomes visible on upright lateral radiographs at approximately 50 mL. This occurs in the form of a meniscus in the posterior costophrenic sulcus on the lateral view, whereas in the posteroanterior projection, the meniscus becomes visible at about 200 mL. It is estimated that at least 500 mL of pleural fluid is necessary to obliterate the hemidiaphragm.
Radiographic Signs
Meniscus sign on frontal radiograph (>200 mL)
Blunting of costophrenic sulcus on lateral view (>50 mL)
Subpulmonic effusion: lateral displacement of the apex of the pseudodiaphragm
Computed Tomography Signs
Sickle-shaped opacity in most dependent part of the thorax, the posterior costophrenic sulci in supine patient
Fluid attenuation (0–20 Hounsfield units)
Pleural effusions can be entirely overlooked on supine radiographs or can be misdiagnosed as pulmonary consolidation or atelectasis. Pleural fluid is often represented as a hazy opacity of one hemithorax with preserved vascular shadows. Other signs are loss of sharp silhouette of the ipsilateral hemidiaphragm and thickening of the minor fissure. The supine radiograph often underestimates the volume of pleural fluid. A prospective study of supine radiographs in 40 patients with pleural effusions showed that effusions with less than 175 mL are unlikely to be detected by this technique.
Lateral decubitus chest radiographs can be used to detect small, free-flowing pleural effusions. As little as 5 mL of pleural fluid can be detected by elevating the patient’s hips and aiming the central beam at the lateral chest wall parallel to the expected fluid level.
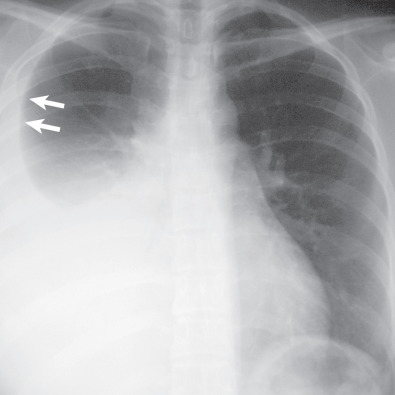
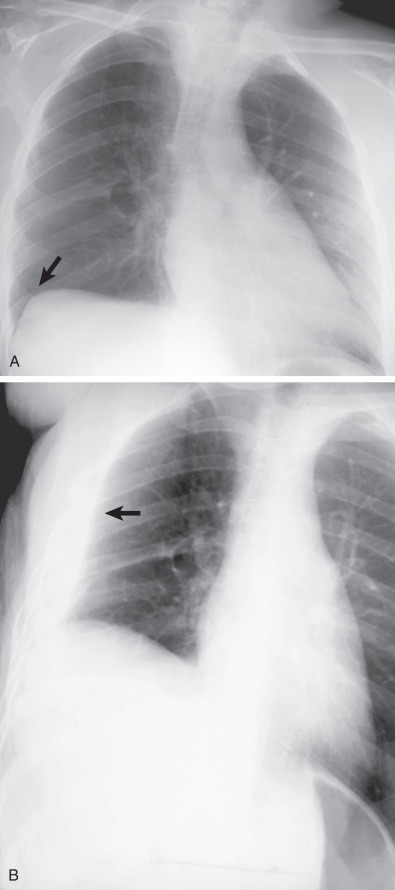
The appearance of free pleural fluid as a homogeneous opacity with a concave upper border, higher laterally than medially, on erect frontal and lateral radiographs is called the pleural meniscus ( Fig. 72.1 ). The meniscus appearance can be explained by the configuration of free fluid within the pleural space and its relationship to the x-ray beam. As a result of its elasticity, the lung tends to preserve its shape while recoiling from the chest wall. This property of the lung causes the fluid to rise against gravity and surround the lower part of the lung. Because x-ray beams have to traverse a greater depth of fluid at the periphery of the thorax, the upper margin appears higher in the form of a meniscus. The superior margin of the fluid located more medially does not produce a layer of sufficient depth to be visible on the radiograph.
Subpulmonic Effusion
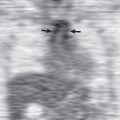
Significant amounts of pleural fluid can accumulate in the subpulmonic region, causing flattening and inversion of the hemidiaphragm without significant blunting of the lateral costophrenic angle. In effect, the lung “floats” on a layer of fluid. These effusions are often transudates and can be difficult to diagnose on the posteroanterior radiograph and may require a lateral decubitus view or ultrasound. On erect frontal radiographs the apex of the pseudodiaphragmatic contour in patients with subpulmonic effusion is more lateral than the apex of the normal hemidiaphragm in about 50% of the cases ( Fig. 72.2 ). This appearance is accentuated on expiratory views. Decubitus view with the involved side dependent results in the accumulation of the fluid along the lateral wall (see Fig. 72.2 ). Nonvisualization of the intrapulmonary blood vessels below the level of the hemidiaphragm may be the only evidence of a subpulmonic effusion, but this sign is nonspecific and can be caused by lower lobe airspace disease or ascites. The normal distance between the left hemidiaphragm and gastric air bubble is usually less than 1 cm, and an abnormal separation of these two has been described as a helpful sign in the diagnosis of left-sided subpulmonic effusions. In the lateral view, typically the pseudodiaphragmatic contour presents a flat superior surface that slants sharply downward at the major fissure.
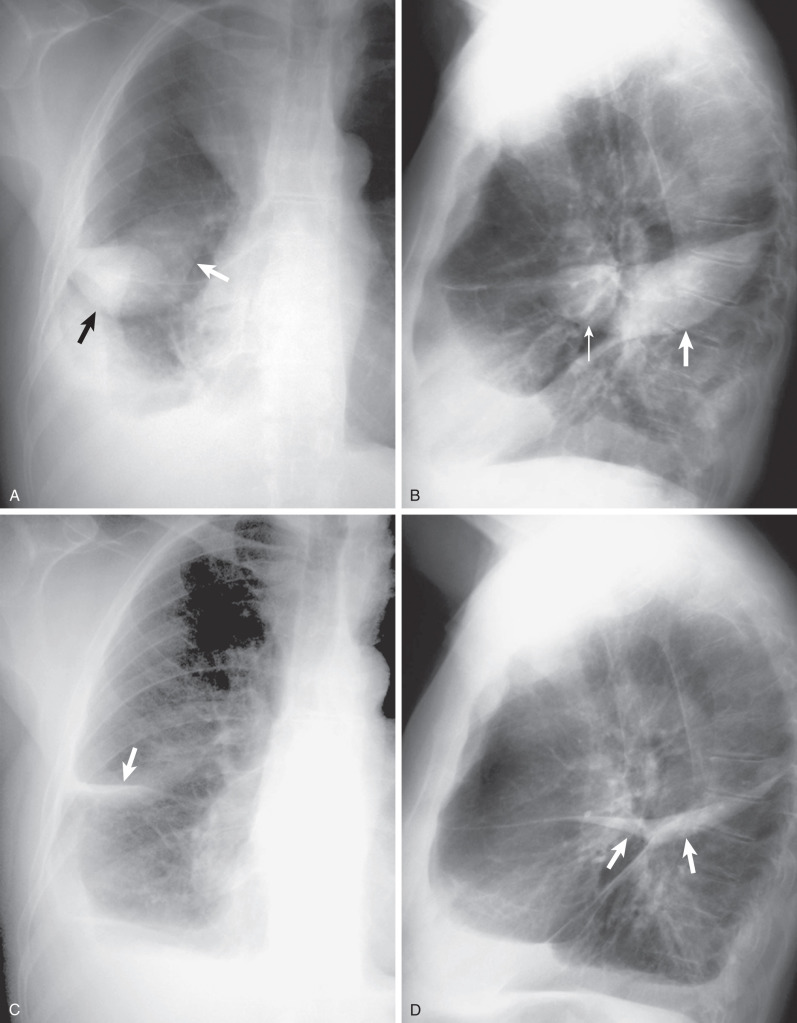
The radiographic appearance of interlobar fluid is multifactorial and depends on the shape and orientation of the fissure, location of fluid within the fissure, and direction of the x-ray beam. Fluid in the major fissure usually appears as a sharp curvilinear interface that is lucent medially and more dense laterally in erect frontal radiographs. It has been classically described as the “incomplete fissure sign,” implying that the effusion is bound by the edge of the incompleteness of the fissure. This sign may be seen even when the major fissure is complete when the effusion is bound by the lateral border of the lower lobe. An interface lucent laterally and more dense medially is usually due to parenchymal disease in the lower lobe. On the lateral chest radiograph, fluid within the upper part of the major fissures is seen as an arcuate opacity with a “fading off” anterior concave margin and sharp posterior interface. This is due to the usual concave surface and lateral facing of the upper part of major fissures so that fluid in the lateral aspect of the fissure is seen in profile, whereas fluid located medially is seen obliquely. Another interesting appearance is the “middle lobe step,” which occurs when fluid accumulates lateral to the middle or lower lobes and extends to the undersurface of the upper lobes. Because incompleteness of the major and minor fissures is a common finding, it is likely that the middle lobe step is due to fluid within the incomplete major and minor fissures.
Pleural pseudotumor is due to accumulation of pleural fluid within an interlobar fissure. This can mimic a pleural or parenchymal mass and create diagnostic confusion. It is most often encountered with minor fissure encapsulation in patients with resolving left heart failure. On frontal views the location of the opacity at the site of the fissure and slight tapering of the medial and lateral margins suggest minor fissure encapsulation ( Fig. 72.3 ). Lateral views are usually confirmatory because the long axis of the opacity confirms the position of the distended fissure, and the tapering extremities are seen more clearly in this projection (see Fig. 72.3 ). These effusions disappear on treatment of the primary cause and are also termed vanishing tumors (see Fig. 72.3 ).
On supine radiographs free pleural effusions produce a hazy density over the hemithorax, caused by the posterior layering of the fluid. This results in an increased homogeneous density superimposed over the lungs that does not obscure normal bronchovascular markings or show air bronchograms. The pleural fluid can cap the apex of the lungs on a supine view ( Fig. 72.4 ) because of the relatively small capacity of the apex compared with the base of the thorax and because the superior and lateral aspects of the apex are the most dependent parts of the thorax tangential to a frontal x-ray beam. Apical capping, seen in about 50% of large effusions, is usually not seen in small- or moderate-sized effusions. Blunting of the lateral costophrenic angle can be seen in the supine position when there is sufficient pleural fluid to fill the posterior hemithorax up to or near the level of the lateral costophrenic angle and is seen in 25% of moderate-sized and 41% of large effusions.

Computed Tomography
CT has been shown to be superior to chest radiographs in distinguishing a peripheral parenchymal lesion from a pleural process. On CT a free-flowing pleural effusion produces a sickle-shaped opacity in the most dependent part of the thorax. Free pleural fluid in a supine patient first accumulates in the posterior costophrenic sulci, the most dependent portion of the chest. Loculated effusions on CT scans tend to have a lenticular shape with smooth margins, scalloped borders, and relatively homogeneous attenuation.
Pleural effusions are characterized on CT by attenuation values between those of water (0 Hounsfield units [HU]) and soft tissue (approximately 100 HU), typically in the order of 10 to 20 HU. CT density measurements alone are considered unreliable in differentiating transudates from exudates or in the diagnosis of chylous pleural effusions. The presence of pleural thickening, pleural nodules, loculation of pleural fluid, or increased attenuation of extrapleural fat can be detected using CT, and these ancillary findings occur most commonly with exudative effusions. CT scans for pleural effusion should be performed with contrast enhancement because pleural abnormalities are better visualized and more easily distinguished from adjacent parenchymal abnormalities. In our experience, CT in systemic venous phase rather than pulmonary arterial phase is better for assessing for pleural enhancement, thickening, and nodularity. It is also often easier to appreciate pleural thickening and nodularity by reviewing the 5-mm sections rather than the thin-section images.
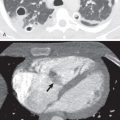
Distinguishing small pleural effusions from ascites on CT is facilitated by analysis of four signs: the diaphragm sign, the displaced-crus sign, the interface sign, and the bare-area sign (see Box 72.1 ). Ascites can be readily differentiated from the pleural effusion if the diaphragm is identifiable adjacent to an abnormal fluid collection in the right upper quadrant. On CT fluid “inside” the diaphragm is ascites, whereas fluid “outside” the diaphragm is pleural effusion (diaphragm sign). Pleural effusions displace the crus anteriorly and laterally away from the spine (displaced-crus sign) ( Fig. 72.5 ). The presence of the diaphragm produces an indistinct interface between the pleural effusion and liver, whereas its absence in ascites results in a sharp, well-defined interface (interface sign) ( Fig. 72.6 ). The right lobe of the liver is directly attached to the posterior abdominal wall and the diaphragm without intervening peritoneum. This direct attachment that lacks a peritoneal covering is called the bare area. Ascites cannot extend behind the liver at the level of the bare area, whereas a pleural effusion can because the posterior sulcus of the right pleural space extends behind the liver (bare-area sign). Fluid behind the liver at the level of the bare area is in the pleural space. When these four signs were compared in a retrospective blinded analysis of 52 patients with right pleural effusion, ascites, or both, it was observed that when effusion and ascites were present, none of the four signs alone could reliably differentiate the fluid collections; however, combined use of the four signs led to correct identification of all the abnormal fluid collections.


Magnetic Resonance Imaging
Magnetic resonance imaging (MRI) plays a limited role in the evaluation of pleural effusion. Fluid collections in the pleural cavity show low signal intensity on T1-weighted images and a high relative signal intensity on T2-weighted images because of their water content. Differentiation of transudates from exudates is unreliable on conventional MRI; however, a study using diffusion-weighted MRI showed that application of diffusion gradients to analyze pleural fluid can differentiate between these two with a sensitivity of 91% and specificity of 85%. Subacute or chronic hemorrhage in the pleural space can be recognized by very high signal intensity on T1-weighted and T2-weighted images. In subacute or chronic hematomas, a concentric ring sign, consisting of an outer dark rim composed of hemosiderin and bright signal intensity in the center because of the T1 shortening effects of methemoglobin, can be observed.
Ultrasonography
Ultrasound is a reliable method for detecting small pleural effusions and for guiding thoracentesis. Most pleural effusions, whether free flowing or loculated, are hypoechoic with a sharp echogenic line that delineates the visceral pleura and lung. An anechoic effusion can be a transudate or exudate ( Fig. 72.7 ). Complex septated, complex nonseptated, or homogeneously echogenic effusions are always exudates ( Fig. 72.8 ). Fibrinous septations are better visualized on ultrasound than on CT scans. Ultrasound has the added advantage of being portable, allowing bedside imaging with the patient in a sitting or recumbent position.


Positron Emission Tomography/Computed Tomography
Positron emission tomography (PET) using fluorodeoxyglucose (FDG) has been shown to be an accurate diagnostic tool in differentiating benign from malignant pleural effusions in patients with malignant disease with sensitivity of 88% to 100% and specificity of 67% to 94%. A retrospective review of PET-CT studies in 32 patients with primary extrapleural malignancy and pleural effusion revealed that PET was more sensitive than CT alone in identifying pleural involvement, and the presence of concomitant pleural abnormalities is the most accurate sign of malignancy when assessing the nature of pleural effusion ( Fig. 72.9 ). Focal increased uptake within the posterior portion of the fluid, presumably resulting from an uptake in malignant cells accumulating in the dependent location, a relatively infrequent finding in patients with malignant pleural effusion, is considered highly specific for malignancy.

False-positive uptake of FDG by the pleura can result from pleural infection or from inflammation after talc pleurodesis; interpretation of the PET findings should take into account the results of pleural fluid analysis and the patient’s recent medical history. A negative PET result can be useful, however, by confirming the absence of pleural metastatic disease, particularly when the results of thoracentesis also are negative.
Differential Diagnosis
Differential diagnoses from clinical data and other supportive diagnostic techniques are listed in Box 72.2 .
Pleuropulmonary infection
Mycobacterium tuberculosis
Nontuberculous bacteria
Actinomyces and Nocardia species
Fungi
Parasites
Viruses and Mycoplasma
Pleuropulmonary malignancy
Lung cancer
Metastatic disease—to pleura and mediastinal lymph nodes
Lymphoma
Leukemia
Connective tissue disease and vasculitis
Systemic lupus erythematosus
Rheumatoid disease
Granulomatosis with polyangiitis
Pulmonary embolism and infarction
Asbestos exposure
Drugs
Heart failure
Metabolic and endocrine disease
Myxedema
Diabetes mellitus
Amyloidosis
Skeletal disease
Gorham disease (chylothorax)
Langerhans cell histiocytosis
Liver disease
Kidney disease
Dialysis
Urinoma
Nephrotic syndrome
Acute glomerulonephritis
Uremia
Pancreatic disease
Acute pancreatitis
Chronic pancreatitis with pleuropancreatic fistula
Gynecologic tumor
Ovary, uterus, fallopian tube
Ovarian hyperstimulation syndrome
Gastrointestinal tract
Gastric/duodenal-pleural fistula
Diaphragmatic hernia
Idiopathic inflammatory bowel disease
Miscellaneous causes
Subphrenic abscess
Lymphatic hypoplasia
Yellow nail syndrome
Dressler syndrome
Familial paroxysmal polyserositis
Systemic cholesterol embolization
Extramedullary hematopoiesis
Idiopathic causes
Transudative Effusion
Left heart failure accounts for most transudative pleural effusions. Effusions in a hepatic hydrothorax, hypoalbuminemia, nephrotic syndrome, constrictive pericarditis, or urinothorax and during peritoneal dialysis also can have the characteristics of a transudate. The development of a transudative effusion indicates that the pleural membrane is normal, and if the underlying problem is corrected, the fluid is reabsorbed without sequelae. Thoracentesis should not be performed for bilateral effusions in a clinical setting strongly suggestive of a pleural transudate unless there are atypical features or they fail to respond to therapy.
Exudative Effusion
Pneumonia, malignancy, thromboembolism, connective tissue diseases, and drugs account for most exudative effusions. Exudative pleural effusions develop when the pleural surface or the local capillary permeability or both are altered. A diagnostic pleural fluid sample should be gathered with a fine-bore (22-gauge) needle and a 50-mL syringe. The sample should be placed in sterile vials and blood culture bottles and analyzed for protein, glucose, LDH, pH, Gram stain, acid-fast bacillus stain, cytology, and microbiologic culture.
Infection
Most empyemas occur as a complication of pneumonia or lung abscess, after thoracic surgery, or in association with an intraabdominal infection. Bacteria such as Staphylococcus aureus, Streptococcus pneumoniae, and enteric gram-negative bacilli are the most common etiologic agents. In an infected effusion, a pH <7.2 indicates the need for tube drainage. Closed-needle biopsy of the pleura for histologic examination has been recommended for undiagnosed exudative effusions when tuberculosis is a concern. The combination of histology and culture establishes the diagnosis of tuberculosis in 90% of patients.
Malignancy
Malignancy is the second most common cause of pleural effusion after left heart failure in elderly patients. The most frequent causes are pulmonary, breast, ovarian, and gastric carcinomas. Malignant effusions can be diagnosed by pleural fluid cytology alone in about 60% of cases. According to the most recent British Thoracic Society (BTS) guidelines, as much fluid as possible should be sent for cytology from the available diagnostic sample (likely to be 20–40 mL) and, when the initial result is negative but malignancy is suspected, the sending of a higher-volume sample after a second aspiration should be considered. If the initial aspiration is both therapeutic and diagnostic, greater than 60 mL should be sent for cytologic examination. If the first pleural cytology specimen is negative, the test should be repeated. The yield depends on the skill of the cytologist and on tumor type, with a higher diagnostic rate for adenocarcinoma than for mesothelioma, lymphoma, and sarcoma. Percutaneous pleural biopsies are recommended when cytology findings are not diagnostic. The diagnostic yield from pleural biopsy is higher under image guidance, which can identify areas of pleural thickening or nodularity.
Connective Tissue Diseases and Vasculitis
Connective tissue disorders and vasculitis may produce pleural effusion with varying degrees of pleural inflammation. The collagen vascular diseases and vasculitides most commonly associated with pleural effusions are rheumatoid arthritis (RA), systemic lupus erythematosus (SLE), Sjögren syndrome, and granulomatosis with polyangiitis (formerly Wegener granulomatosis). RA is unlikely to be the cause of an effusion if the glucose level in the fluid is greater than 1.6 mmol/L (29 mg/dL).
Chylothorax
Approximately 50% of cases of chylothorax are due to malignancy (particularly lymphoma); 25% are related to trauma; and the rest are due to tuberculosis, sarcoidosis, and amyloidosis. Pseudochylothorax or cholesterol pleurisy results from the accumulation of cholesterol crystals in a long-standing pleural effusion. It can occur in chronic rheumatoid pleurisy and tuberculosis. If a chylothorax or pseudochylothorax is suspected, pleural fluid should be sent to measure triglyceride and cholesterol levels and to determine the presence of cholesterol crystals and chylomicrons. A true chylothorax shows the presence of chylomicrons and has a high triglyceride level (>1.24 mmol/L). In a pseudochylothorax the cholesterol level is elevated (>5.18 mmol/L), and cholesterol crystals are often seen at microscopy.
Pleural Effusion Caused by Infection
Infectious pleural effusions, most caused by bacterial infections, tend to be exudative in nature. The formation of pleural fluid is attributed to increased capillary permeability and protein leakage as a result of the inflammatory process. In addition, inflammatory exudate on the pleural surface and fibrous thickening of the pleura impede reabsorption of fluid by the lymphatics.
Mycobacteria
Pleural involvement is one of the most common extrapulmonary manifestations of Mycobacterium tuberculosis infection. Pleural involvement may be seen at any time in the natural course of the disease but occurs most commonly 3 to 7 months after the primary infection. About 80% of patients with tuberculous pleural effusion have concomitant pulmonary tuberculosis.
Epidemiology
The frequency of tuberculous effusion depends on the prevalence of tuberculosis in the population studied. In developed countries, such as the United States and United Kingdom, pleural involvement is seen in less than 10% of cases of infection with M. tuberculosis . In countries where tuberculosis is common, it is the most frequent cause of exudative pleural effusion or empyema.
Emergence of the human immunodeficiency virus (HIV) pandemic has had a profound effect on the epidemiology of tuberculosis. One meta-analysis showed significant association between presence of HIV and extrapulmonary tuberculosis. In a prospective study of 1000 patients with pleural effusion in a tertiary care hospital in Spain, of the 42 patients with HIV infection, the most common cause of pleural effusion was tuberculosis. The rate of pleural involvement in HIV-positive patients with tuberculosis can be 40%. Tubercular pleural effusions in HIV-positive patients are also more likely to be bilateral and have associated parenchymal involvement and positive bacteriology when compared with HIV-negative patients.
Pathophysiology
Tuberculous pleural effusions usually occur after a latent period of 3 to 6 months after primary infection and are due to the rupture of the subpleural Ghon focus into the pleural cavity. These primary tuberculous effusions are the result of a delayed-hypersensitivity reaction to bacilli entering the pleural space rather than direct infection. An effusion in reactivation disease is due to direct infection of the pleural space, rather than an immunologic phenomenon, and occurs as a result of the direct spread of bacilli from the cavitary lung lesions, bloodstream, and lymphatic system. In contrast to primary tuberculous effusions, numerous bacilli are secreted into the pleural cavity. Chronic active infection may lead to the formation of a tuberculous empyema. Rupture of a parenchymal cavity into the pleural space can result in a bronchopleural fistula and pyopneumothorax.
Clinical Presentation
The clinical presentation of tuberculous pleural effusion may be acute or insidious. Symptoms include dyspnea, cough, pleuritic chest pain, and malaise.
Manifestations of the Disease
Radiography.
Tuberculous pleural effusion in non-HIV–infected patients is almost invariably unilateral and seldom massive. In one retrospective study, only 19 of 113 (17%) tuberculous pleural effusions occupied more than two-thirds of the hemithorax on posteroanterior upright chest radiographs.
Apart from detecting pleural fluid, routine radiographs have a limited role in establishing the diagnosis because parenchymal lesions most often go undetected. It is prudent to keep these patients in isolation until a thorough evaluation for the presence of active pulmonary tuberculosis is negative.
Computed Tomography.
Patients may develop new pulmonary lesions while on medications for tuberculous pleural effusions. A paradoxical response, affecting 16% of patients in one study, generally occurs 3 to 12 weeks after the beginning of antituberculous therapy, usually regressing without a change in the initial drug regimen. In a retrospective review of 16 patients with radiologic evidence of paradoxical response to antituberculous therapy, CT revealed the development of new peripheral pulmonary nodules or masses, which mostly abutted the normal or thickened pleura, subsequently decreasing in size and finally disappearing on continuation of the same antituberculous medication.
Tuberculous pleuritis may progress to chronic persistent pleural effusion or empyema ( Figs. 72.10 and 72.11 ), which may be defined as persistent, grossly purulent pleural fluid containing numerous tubercle bacilli. Tuberculous empyema may manifest as a loculated pleural fluid collection with parenchymal disease and cavitation. In most cases of tuberculous empyema, the final outcome is extensive fibrothorax, easily detected as a thick calcified pleural rind on chest radiographs and CT.


Chronic persistent effusion should be suspected if pleural thickening is greater than 2 cm or when more than one linear shadow is visible running parallel to the inner chest wall on chest radiographs. CT shows a loculated pleural fluid collection in association with pleural thickening and calcification. Pleural thickening caused by tuberculosis can mimic malignancy and can involve pleura in circumferential fashion with involvement of mediastinal pleura, parietal pleural thickness of greater than 1 cm, and nodularity. Chronic tuberculous empyema may decompress through the chest wall, in which case it is called empyema necessitatis, a rare but well-recognized complication of tuberculous empyema. CT findings of empyema necessitatis include well-demarcated, thick-walled fluid collections in intrathoracic and extrathoracic locations ( Fig. 72.12 ). Air-fluid levels in the pleural space indicate bronchopleural fistula.

The occurrence of malignant neoplasm is a relatively rare but important complication of chronic tuberculous empyema. Pathologic cell types of malignancy associated with long-standing empyema include malignant lymphoma, squamous cell carcinoma, mesothelioma, malignant fibrous histiocytoma, sarcoma, and hemangioendothelioma.
Tuberculous pleurisy can result in chyliform (pseudochylous or cholesterol effusion) pleural effusion. Fat-fluid or fat-calcium level at CT was described as pathognomonic of tuberculous chyliform effusion, and associated findings included parietal pleural thickening of 4 to 10 mm and curvilinear or nodular pleural calcifications.
Investigations and Differential Diagnosis
Thoracentesis of a primary tuberculous pleural effusion characteristically yields a clear, straw-colored exudate with total pleural fluid protein greater than 3 g/100 mL, pleural fluid LDH greater than 200 U, and pleural fluid–serum LDH ratio greater than 0.5. The fluid is lymphocyte predominant, often exceeding 70% or more of all cellular material, and so during cytologic examination may appear as the only cell type, resulting in an erroneous diagnosis of lymphoma. The glucose content is often low in tuberculous effusions. This finding is not specific for tuberculosis, however; low pleural fluid glucose content also is seen in bacterial pneumonia, rheumatoid disease, and pulmonary carcinoma.
Several studies have shown that a level of adenosine deaminase (ADA) in pleural fluid greater than 40 IU/L can indicate pleural tuberculosis with a sensitivity of 81% to 100% and specificity of 83% to 100%. Analysis and determination of the isoenzymes have shown that increases in ADA with tuberculous pleurisy are due to increase in ADA 2 . Estimation of pleural fluid ADA 2 levels has a similar sensitivity and a slightly better specificity compared with the pleural ADA in the diagnosis of tuberculous pleuritis. False-positive results can occur in other lymphocytic effusions, such as malignancies and parapneumonic effusions, but are rare.
An elevated pleural fluid interferon-γ level also is valuable in diagnosing tuberculous pleural effusions. In a prospective study of 595 pleural effusions, of which 82 (14%) were due to tuberculosis, a cutoff of 3.7 international units (IU)/mL yielded a sensitivity of 98% and a specificity of 98% for diagnosing tuberculosis. In a study comparing the efficacy of different biologic markers for the diagnosis of tuberculous pleuritis, interferon-γ proved to be the most sensitive and specific biologic marker.
Definitive diagnosis of pleural tuberculosis requires the identification of mycobacteria by microscopy or culture of the pleural fluid or tissue. Mycobacterial culture has a higher sensitivity than direct smear for acid-fast bacilli because direct examination requires a concentration of 10,000 bacilli/mL, whereas culture requires the presence of only 10 to 100 organisms/mL. Microscopy and culture of the pleural fluid are negative in many cases and may not exclude tuberculous pleurisy. The incidence of positive culture from pleural fluid is low, having been reported in 15% to 30% of proven cases.
In patients with tuberculous effusions, culture of pleural biopsy specimens is positive in 55% to 80% of cases, and in one series a diagnostic sensitivity of 79% and specificity of 100% were achieved when histology and culture were combined. In a study to determine the optimal number of pleural biopsy specimens for the diagnosis of tuberculous pleurisy, it was shown that the sensitivity of percutaneous needle biopsy for diagnosis of tuberculous pleurisy was greatest when more than six specimens were obtained; on average, these contained more than two specimens of parietal pleura. Image-guided pleural biopsy can be useful in obtaining diagnostic tissue in patients who have small effusions. Thoracoscopy can be performed under local anesthesia in patients with pleural effusion and provides a means to visualize directly the pleural abnormalities. Thoracoscopic pleural biopsy has a high diagnostic rate with sensitivity greater than 90% and can be performed easily and safely.
The potential of pleural fluid polymerase chain reaction in the diagnosis of tuberculous pleural effusion has been explored, but sensitivities range from 20% to 81%, and specificities range from 78% to 100%, implying a low clinical utility of the test.
Treatment
Tuberculous empyema should be managed with intercostal drainage of pus, antituberculous therapy, and the consideration of surgical interventions such as decortication when conservative measures are unsuccessful.
Bacteria Other Than Mycobacteria
A parapneumonic effusion is an accumulation of exudative pleural fluid secondary to an ipsilateral pneumonia or lung abscess. A complicated parapneumonic effusion or an effusion in which the bacterial cultures are positive requires an invasive procedure, such as tube thoracostomy, for its resolution.
Prevalence and Epidemiology
Parapneumonic effusions occur in some 20% to 40% of the approximately 1 million patients hospitalized yearly with pneumonia in the United States. These effusions are usually serous exudates that resolve spontaneously without specific therapy; approximately 10% become complicated or progress to empyema. Delays in drainage of clinically significant effusions have been associated with increased morbidity and mortality. In a multicenter prospective study of 1906 patients with clinical and radiographic evidence of community-acquired pneumonia, the mortality risk was 6.5 times higher when the effusions were bilateral and 3.7 times higher when the effusion was unilateral.
Bacteriology
The bacteriology of parapneumonic effusions and empyemas is diverse, and significant differences are noted between community-acquired and nosocomial infections. Regardless of the setting, most culture-positive effusions are due to gram-positive aerobic bacteria, most commonly streptococcal species, followed by staphylococci. In the nosocomial setting, gram-negative aerobes, such as Haemophilus influenzae, Escherichia coli, Pseudomonas, and Klebsiella, also are common causes of pleural infections. The incidence of anaerobic bacterial pleural infection is reported to range from 12% to 34%. The most frequent anaerobic isolates are Bacteroides species and Peptostreptococcus. On occasion, Actinomyces, Nocardia, or fungi such as Aspergillus may be the cause of empyema.
Clinical Presentation
The clinical picture in patients with pneumonia is similar whether or not they have pleural infection and parapneumonic effusion. Pleural infection should be suspected in any patient being treated with antibiotics who has nonresolving pneumonia. In contrast to aerobic pleuropulmonary infections that usually manifest as an acute febrile illness, anaerobic infections have a more subacute or chronic presentation, with associated weight loss and anemia.
On occasion, a purulent effusion can occur without pneumonia (primary empyema). This may represent a postpneumonic empyema in which pulmonary consolidations have already resolved or a nonpneumonic empyema, most often iatrogenic and related to thoracic surgery. It also can be secondary to thoracic trauma, esophageal perforation, or transdiaphragmatic spread of abdominal infections.
Rarely, bilateral pleural effusions are present, with one side representing empyema and the other being a noninfected pleural effusion ( Fig. 72.13 ). The Contarini condition is characterized by empyema on one side and contralateral pleural effusion secondary to fluid overload or congestive cardiac failure; the condition was named after the 95th doge of Venice, who died in 1625 with orthopnea, foul-smelling sputum, cardiac arrhythmia, and clear pleural fluid on one side and pus on the other.

Pathophysiology
The progression of parapneumonic effusion can be divided into three stages. The first stage is the exudative stage, in which the accumulation of fluid in the pleural space is most likely the combined result of the pulmonary interstitial fluid traversing the pleura to enter the pleural space and increased permeability of the capillaries in the pleural space. This effusion is free flowing; is not infected; and has normal biochemical characteristics, such as a pH greater than 7.30, glucose level greater than 60 mg/dL, and a slightly elevated LDH level. At this stage, this sterile exudate is also referred to as “simple parapneumonic effusion,” and most do not require chest tube drainage.
This effusion may progress to the second stage, which is the fibropurulent stage, in patients with no or ineffective antibiotic therapy. This stage is characterized by the accumulation of fibrin clots and fibrin membranes, resulting in a heavily loculated pleural space. Gram stains and bacterial cultures are usually positive, with the cytology of the pleural fluid usually showing neutrophils greater than 10,000/mL. The metabolic and cytolytic activities in this stage are high, as reflected by low pH values (<7.20), low glucose level (<35 mg/dL), and high LDH value (>1000 IU/L), consistent with “complicated parapneumonic effusion.”
If the effusion is not drained at the fibropurulent stage, it can progress to the final organizational stage characterized by fibroblast proliferation, leading to transformation of interpleural fibrin membranes into a web of thick and nonelastic pleural peels. The peel over the visceral pleura prevents the lung from expanding. The further course after such an organization varies. Some individuals exhibit spontaneous healing with resolution of pleural thickening, whereas others progress to lung function deficit and chronic infection of the pleural space, making way for further complications, such as lung abscess, bronchopleural fistula, or empyema necessitatis.
Manifestations of the Disease
Radiography.
The evaluation of patients with pneumonia also must include assessment for a parapneumonic effusion. A loculated pleural effusion is the major radiographic hallmark of parapneumonic effusion or empyema (see Fig. 72.11 ). No other specific radiographic features are seen except, rarely, the presence of gas in the pleural space (pyopneumothorax) or chest wall owing to gas-forming bacteria, such as Clostridium perfringens and Bacteroides fragilis, or more likely from bronchopleural fistula or recent thoracentesis .
Prompt recognition and precise characterization of a parapneumonic effusion is important because delays in drainage of clinically significant effusions have been associated with increased morbidity and mortality. The detection of pleural fluid on standard radiographs is affected by patient position, which alters the distribution of pleural effusions as previously discussed.
The lateral decubitus view often reveals small effusions that have not been demonstrated by erect chest radiographs. Bilateral decubitus chest radiographs also may help to separate dense consolidation from free-flowing pleural fluid. A decubitus chest radiograph also can guide decision making for thoracentesis. Small free-flowing pleural effusions (<10-mm thickness) in the lateral decubitus view usually resolve with antibiotic therapy alone, and thoracentesis of these effusions is often unnecessary.
Computed Tomography.
CT is often used to evaluate pleural or parenchymal complications of pneumonia. It is sensitive in the detection of parapneumonic effusions, depicting multiple loculations in these effusions, and has proved helpful in revealing poor chest tube placement. Empyemas typically have an oval or lentiform shape, exhibit obtuse margins with the pleura, have scalloped contours, and have mass effect on the airways and adjacent lung, displacing them away from the effusion. These help to distinguish them from the more rounded appearance of pulmonary abscesses, which typically form acute angles with the chest wall. There may be several loculated effusions, requiring more than one chest tube for adequate drainage.
A contrast-enhanced chest CT scan that shows parietal and visceral pleural enhancement, and thickening with increased attenuation of the extrapleural fat is highly suggestive of an empyema in the appropriate clinical setting. Enhancement of the pleura in empyema is thought to be caused by the increased vascularity of the inflamed pleura. The presence of fluid between the enhancing thickened pleural layers has been termed the split pleura sign (see Fig. 72.11 ). Although best seen with contrast-enhanced CT, it may be present on noncontrast CT (see Fig. 72.10 ). If no interventional procedure has been performed, gas bubbles within a pleural fluid collection are virtually diagnostic of an empyema and indicate either infection caused by gas-forming bacteria or more likely the presence of a bronchopleural fistula. Enhancing pleura and thickened parietal pleura are strongly associated with empyema. In a study of empyema, pleural enhancement was present in every patient, and pleural thickening was seen in 92% of patients. In another study a retrospective review of the CT findings in 30 children with parapneumonic effusions in which 21 met the clinical criteria for empyema, pleural enhancement was noted in all children with empyema and 8 children with parapneumonic effusions (89%). Pleural thickening is also more commonly, and to greater extent, seen in patients with empyema than in patients with parapneumonic effusion. Absence of pleural thickening suggests simple parapneumonic effusion.
When parapneumonic effusion or empyema complicates bacterial pneumonia, mediastinal lymph node enlargement can occur. In a retrospective study of 50 patients with parapneumonic effusion or empyema, enlargement of mediastinal nodes greater than 1 cm was present in 18 patients (36%), whereas 13 patients had nodes measuring less than 1 cm (26%), indicating that intrathoracic lymphadenopathy less than 2 cm could be regarded as a normal finding in empyema. In this study no correlation could be established between the presence of enlarged nodes with the length of the illness, stage of the effusion, presence of frank purulence, extent of consolidation, or the size of the effusion. In another study intrathoracic lymphadenopathy occurred in 48% of cases with empyema, suggesting that in patients with smooth pleural thickening and pleural effusion, lymphadenopathy alone should not be used as a criterion to differentiate empyema from malignant or tuberculous pleural effusion.
- •
Split pleura sign
- •
Oblong configuration
- •
Obtuse angle with adjacent pleura
- •
Displacement of adjacent lung and airways from pleura
Ultrasonography.
A clear-cut distinction between an uncomplicated parapneumonic effusion and an infected effusion cannot be made with confidence using transthoracic sonography alone. Infected fluid may be completely sonolucent without internal echoes. A retrospective study of 50 patients with parapneumonic effusions using sonography found that most of these effusions were septated and echogenic (see Fig. 72.8 ), and there was no relationship between sonographic features and biochemical parameters or outcome measures. This study also showed that septa may be found in stage I collections, which are frequently known to resolve without tube drainage. When the effusions are small in size, transthoracic ultrasound is useful for guiding thoracentesis or for inserting chest tubes and is especially useful in circumstances that require precise targeting, such as loculated effusions.
Chest ultrasonography is useful for showing early fibrin membranes and septations in the pleural cavity in patients with empyema. In the absence of clear septations on ultrasonography, simple chest tube drainage with or without fibrinolysis is adequate, whereas septated effusions often require mechanical adhesiolysis.
Management
Parapneumonic effusions are classified on the basis of anatomic characteristics of the fluid, the bacteriology of the pleural fluid, and the chemistry of the fluid. Category 1 pleural fluid is a small free-flowing effusion less than 10 mm thick on a decubitus radiograph, CT, or ultrasound. Thoracentesis is not done because the risk of a poor outcome with a category 1 effusion is low. Observation is an acceptable treatment option for this category of effusion.
Category 2 effusion is free flowing and small to moderate in size (>10 mm thick and less than half of the hemithorax). Gram stain and culture of the pleural fluid are negative, and the pleural fluid pH is greater than 7.20. The risk of a poor outcome with category 2 effusions is low. It is recommended that the pleural fluid be analyzed within a short time to determine whether any poor prognostic factors develop that would indicate the need for aggressive drainage.
Category 3 effusions occupy more than half the hemithorax, are loculated, or are associated with thickened parietal pleura. Gram stain or culture is positive. The pleural fluid pH is less than 7.20, or the pleural fluid glucose is less than 60 mg/dL. The risk of a poor outcome with a category 3 effusion is moderate. Category 4 effusions are characterized by pleural fluid that consists of pus, and the risk of a poor outcome is high.
As it is impossible clinically to differentiate simple from complicated parapneumonic effusion in patients with possible pleural infection, BTS guidelines recommend diagnostic pleural fluid sampling under imaging guidance in all patients with a pleural effusion greater than 10-mm depth in association with a pneumonic illness or recent chest trauma or surgery and who have features of ongoing sepsis. If a patient has sufficient pleural fluid to warrant a thoracentesis, a therapeutic rather than diagnostic thoracentesis is recommended. If the fluid reaccumulates and there was no poor prognostic factor at the time of initial thoracentesis, no additional therapy is indicated as long as the patient is doing well. If the fluid reaccumulates, and there were poor prognostic factors present at the time of the initial thoracentesis, a second therapeutic thoracentesis should be performed. If the fluid reaccumulates a second time, a tube thoracostomy should be performed. Chest tube drainage is recommended by BTS in patients with frankly purulent fluid aspirated, identification of organisms on Gram stain and/or culture, pleural fluid pH less than 7.2, or loculated pleural fluid. Along with poor prognostic factors, if the pleural fluid is loculated, more aggressive therapy is indicated, such as tube thoracotomy with fibrinolytics. If complete drainage is not achieved with one or two administrations of fibrinolytics, thoracoscopy should be done. Decortication should be performed without delay if the lung does not reexpand completely with thoracoscopy alone.
Fungi
Pleural effusions are uncommon in immunocompetent hosts with acute or disseminated histoplasmosis. In a series of 50 patients with acquired immune deficiency syndrome (AIDS) and disseminated histoplasmosis, 5 (10%) patients showed pleural effusions on chest radiographs.
Small pleural effusions may occur in about 20% of patients with acute coccidioidomycosis and is thought to be secondary to direct spread of contiguous parenchymal infection. Spontaneous rupture of a coccidioidal pulmonary cavity into the pleural space is known to occur in 1% to 5% of patients with chronic cavitary disease, resulting in a pyopneumothorax.
Effusions caused by Blastomyces dermatitidis usually are associated with acute airspace pneumonia. Of a series of 26 patients with pulmonary blastomycosis, 23 (88%) showed radiographic evidence of blastomycotic pleural disease; of these, only 4 patients had pleural effusions ( Fig. 72.14 ), and the patients responded well to amphotericin B therapy.

Pleural effusions are more common in immunosuppressed patients with thoracic cryptococcosis. A retrospective study comparing 16 immunocompromised patients with 13 immunocompetent patients, all with pulmonary cryptococcosis, reported effusions only in 3 (19%) immunocompromised hosts, whereas none of the immunocompetent patients showed effusions.
Fungal thoracic empyema, most often hospital acquired, is caused mainly by Candida species. The major causes of fungal empyema include abdominal infection, bronchopulmonary infection, surgical intervention, and repeated thoracentesis. Aspergillus thoracic empyema is uncommon and can have a radiologic appearance of marked pleural thickening bordered by a shaggy inner margin. It is caused by rupture of an aspergilloma cavity or as a complication of a preexisting chronic empyema.
Parasites
Pleuropulmonary amebiasis is a relatively common condition in developing countries. The liver is the most common extraintestinal site of amebic involvement, followed by the lung. Pulmonary involvement is usually by a direct transdiaphragmatic extension of a hepatic abscess across an obliterated pleural space, resulting in consolidation, abscesses, or bronchohepatic fistula. About 6% to 40% of patients with amebic liver abscesses are known to develop pleuropulmonary complications; a right-sided pleural effusion occurs in about 60% of cases. In addition to a right-sided pleural effusion, radiographs commonly reveal an elevated right hemidiaphragm with an upward hump-like prominence on the right lateral view and a triangular right lower lobe consolidation. The effusions usually are serofibrinous. In contrast to bacterial empyemas, which progress from an early exudative stage to the final organizing stage, amebic empyemas do not show temporal progression, and loculations and septations are only rarely detected on sonography. The pulmonary consolidation may cavitate, providing communication between the bronchial tree and the liver abscess. Expectoration of a fluid having an “anchovy sauce” appearance indicates the development of a bronchohepatic fistula.
Echinococcus Granulosus
Pleural effusion is uncommon in hydatid disease but occurs when a pulmonary hydatid cyst ruptures into the pleural space, resulting in a hydropneumothorax. Daughter cysts floating on the surface of the fluid produce irregularities of the fluid surface, creating the water lily sign or the “sign of the camalote.”
Pleural Effusions in Patients With Acquired Immunodeficiency Syndrome
Pleural effusions are seen in 7% to 27% of hospitalized patients with HIV infection. The three most common causes of effusions are bacterial pneumonia, tuberculosis, and Kaposi sarcoma. Bacterial pneumonia is the most common condition associated with pleural effusion in HIV-infected patients in the United States, whereas in underdeveloped countries it is tuberculosis. Noninfectious pleural effusion in hospitalized patients with AIDS is most often caused by hypoalbuminemia. Patients with HIV usually have small effusions. Bilateral effusions are more common in patients with AIDS-related Kaposi sarcoma and lymphoma.
Asbestos-Related Pleural Effusion
Etiology, Prevalence, and Epidemiology
Benign asbestos-related pleural effusions are a significant and underrecognized manifestation of asbestos exposure with important sequelae, such as diffuse pleural thickening, which may be associated with functional impairment and for which compensation may be sought.
Clinical Presentation
Benign effusion is the most common asbestos-related abnormality during the first 20 years after exposure; typically, there is a latency period of 10 years. The majority of patients are asymptomatic (44%–66%), but some can present with symptoms of acute pleuritis with pain and fever. Effusions are mostly unilateral but can sometimes be bilateral, recurrences are frequent, and can be on the same or the contralateral side.
Pathophysiology
Benign asbestos-related pleural effusions are exudative but have a variable composition; often the pleural fluid is hemorrhagic, of mixed cellularity, and may have an increased eosinophil count. Asbestos fibers are not usually found in the effusion. Epler and colleagues reported a dose-response relationship with an increased incidence of effusions in individuals with the most severe exposure; prevalence was 7% with severe, 3.7% with indirect, and 0.2% with peripheral exposure. These investigators also suggested an occupational relationship, with the effusions seen most commonly in asbestos pipe coverers and least commonly among shipfitters and welders. The effusions usually resolve spontaneously after 3 to 4 months. They may persist or recur, however, over several years. In Epler’s study, most effusions were small and recurred in 29% of cases.
Differential Diagnosis
Benign asbestos-related effusion is defined by (1) exposure to asbestos, (2) confirmation by radiographs or thoracentesis, (3) no other disease related to pleural effusion, and (4) no malignant tumor within 3 years after the onset of the effusion. One case of mesothelioma has been reported 6 years after benign effusion. Asbestos exposure should be sought carefully in patients with “idiopathic” pleural effusion. The presence of pleural plaques is helpful to identify benign asbestos-related pleural effusion, but pleural plaques have a much longer latency period than pleural effusions and are most commonly not present ( Fig. 72.15 ). The presence of “crow feet”—linear parenchymal structures converging on the pleura—and rounded atelectasis is helpful in suggesting the diagnosis; they are seen less frequently in exudative pleural effusions of other causes. The absence of nodular, circumferential, or mediastinal pleural thickening is helpful in suggesting a benign diagnosis.