Abstract
Prune-belly syndrome is characterized by urologic abnormalities, flaccidity, or dysplasia in abdominal wall musculature, and cryptorchidism (in males). Because of the poorly developed abdominal wall muscles, the infant’s abdomen has a wrinkled, prune-like appearance, thus giving origins of the syndrome’s name. It occurs in 1 : 29,000 to 1 : 50,000 births and is more common in males than females and blacks compared with whites. The most common observation on prenatal ultrasound is bladder distention, which may be accompanied by hydronephrosis, dysplastic kidneys, or unilateral renal agenesis. Oligohydramnios is frequently seen. Prenatal interventions exist and may improve postnatal survival and function. At this time, no specific genetic etiology has been linked definitively to prune-belly syndrome. Recurrence risk is thought to be low, though this may change as genetic etiologies are identified and genetic testing improves.
Key Words
Prune-belly syndrome, obstructive uropathy, Potter sequence, vesicoamniotic shunt
Introduction
Prune-belly syndrome (PBS), also known as Eagle-Barrett syndrome, is a triad of anomalies composed of abdominal wall flaccidity, urologic abnormalities, and bilateral cryptorchidism. The typical urologic findings are distended bladder, hydronephrosis, and renal dysplasia. A patent urachus is a common finding. In some infants, urinary obstruction may lead to oligohydramnios or anhydramnios with a resulting Potter sequence (pulmonary hypoplasia, dysmorphic facies). Concomitant cardiac, orthopedic, and gastrointestinal anomalies are frequent and are reported in approximately 75% of PBS cases.
Disorder
Definition
PBS is characterized by urologic abnormalities, dysplasia, or flaccidity of the abdominal wall musculature, and bilateral cryptorchidism (in males). The poorly developed abdominal wall results in a wrinkled appearance of the infant’s abdomen, giving the syndrome its name.
Prevalence and Epidemiology
The reported incidence ranges from 1 : 29,000 to 1 : 50,000 live births with a significant male preponderance; 95% of cases occur in males. The syndrome is twice as common in blacks compared to whites. Also the incidence in twins has been reported to be four times higher than in singletons. An apparent increase in incidence of PBS in children of younger mothers has also been observed.
Etiology and Pathophysiology
Two theories exist as to the etiology of PBS. The first suggests that the sequence is initiated by an obstructive uropathy. This theory posits that a primary urinary tract obstruction leads to distention of the bladder, prohibiting formation of the anterior abdominal wall muscles and descent of the testis. The resulting oligohydramnios then results in a Potter sequence, leading to contractures and pulmonary hypoplasia. However, this theory does not fully explain the associated cardiac and gastrointestinal defects associated with PBS; furthermore, it does not explain why some urinary tract obstructions, such as posterior urethral valves, do not lead to the spectrum of findings found in PBS. Last, most infants born with PBS do not actually have a urinary tract obstruction.
The second theory focuses on embryogenesis; an aberration of mesenchymal development between the sixth and 10th weeks of gestation could lead to the spectrum of findings in PBS. A mesenchymal defect in the sixth to seventh week could explain not only the typical triad of findings, but also the commonly found orthopedic and gastrointestinal findings.
The male preponderance has suggested an X-linked recessive or perhaps a sex-influenced autosomal recessive mode of inheritance. A few case reports exist of PBS in association with a trisomy or a chromosomal deletion, but the loci of the abnormality is inconsistent across these reports. At this time, no chromosomal abnormality has been linked definitively to PBS. However, an abnormality of hepatocyte nuclear factor 1β (HNF1β), which is expressed on chromosome 17, has been demonstrated in patients with sporadic PBS. This transcription factor is found on numerous tissues in the body including mesonephric duct derivatives, renal tubules of the metanephros, and the developing prostate. Other potential genetic factors have been suggested including hypomethylation on chromosome 6q24 and 11p15 and ACTG2 mutations, which encodes for enteric actin.
The risk of recurrence of PBS is considered low ; however, considering the genetic factors/influences of PBS are not well understood, this may change as technology/genetic testing options improve.
Manifestations of Disease
Clinical Presentation
Abdominal Wall.
The defect in the development of the abdominal wall musculature gives the infant’s abdomen a wrinkled, prune-like appearance. In severe defects, the muscle may be completely absent ventral to the midaxillary line.
Urogenital System.
Distention of the bladder leads to thickening of the bladder wall, displacement of the ureteral orifices laterally, and vesicoureteric reflux. Urinary obstruction is present in approximately 25% of infants, and most infants demonstrate urinary retention, in part caused by defects in the abdominal wall musculature. Imaging of the upper urinary tract is necessary: hydronephrosis is a common finding, and polycystic kidneys, renal atrophy, and renal agenesis have all been documented.
A patent urachus is present in 20%–50% of cases. This can be diagnosed on physical exam by noting the presence of a clear discharge from the umbilical cord; specific gravity can be done on the liquid to confirm that it is urine. The significance of this finding is unclear; some have suggested that a patent urachus is associated with an increased mortality rate, whereas others have suggested that a patent urachus may allow urinary drainage in utero , thereby preventing oligohydramnios and pulmonary hypoplasia.
Bilateral cryptorchidism is present in affected males, possibly caused by interruption of testicular descent by a distended bladder. In most cases both testes are present intraabdominally and viable. Affected females are rare, but may present as pseudohermaphrodites with an abnormally located urethral meatus, fused labia, and clitoral hypertrophy.
Pulmonary System.
Oligohydramnios in utero frequently leads to pulmonary hypoplasia. The degree of pulmonary hypoplasia varies and typically determines outcomes in these infants. In addition, recurrent pulmonary infections occur, likely caused by impaired ventilation as a result of abdominal wall laxity.
Cardiovascular System.
The incidence of cardiovascular anomalies has been estimated at 10%. Anomalies described include patent foramen ovale, atrial and ventricular septal defects, and tetralogy of Fallot.
Gastrointestinal System.
Anomalies in the gastrointestinal system seem to affect approximately 30% of infants with PBS. Commonly reported findings include malrotation, mesenteric anomalies, atresia (particularly colonic atresia), fistula, imperforate anus, and diverticula.
Musculoskeletal System.
Abnormalities of the musculoskeletal system have been reported in up to 63% of patients. This may be a consequence of a mesenchymal insult during embryogenesis or a result of oligohydramnios, as in Potter sequence. Common findings include congenital hip dysplasia, scoliosis, club foot, torticollis, and contractures.
Prognosis.
PBS can be classified into three prognostic groups. The first group comprises 20% of cases; these infants are typically stillborn or die shortly after birth because of significant pulmonary hypoplasia and renal dysplasia. Infants in group 2, representing 40% of cases, have adequate renal function at birth but require intervention; without intervention, one-third of these patients will die from renal failure or sepsis. Patients in group 3, making up the remaining 40% of cases, have mild urinary tract abnormalities and most will survive.
Imaging Technique and Findings
Ultrasound.
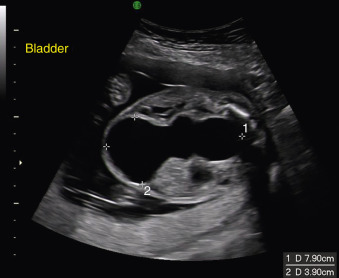
The earliest time of diagnosis of PBS reported in the literature was made in the 12th week of gestation. The most frequent ultrasound finding in cases of PBS is a distended bladder ( Figs. 139.1 and 139.2 ). This may be accompanied by hydronephrosis, dysplastic kidneys, or unilateral renal agenesis. Oligohydramnios is a frequent finding. Cryptorchidism can be detected prenatally; however, this diagnosis should not occur before 25 weeks as testicular descent is not observed until after 25 weeks; 97% of normal males will have bilateral testicular descent at 32 weeks. Cardiac and musculoskeletal anomalies may be seen, but oligohydramnios may limit the examination of these and other structures.