Chapter 13 Quality assurance and performance testing
INTRODUCTION
This chapter provides the reader with a general overview of quality assurance (QA) and performance testing of diagnostic ultrasound equipment. Performance testing of pulse-echo imaging systems will be covered, however because the evaluation of Doppler systems is technically more difficult and is normally performed by specialized medical physicists, it will not be included in this chapter.
QUALITY ASSURANCE
QA is the process of ensuring that all aspects of an ultrasound service meet and perform to agreed standards. The aim of QA is to maintain standards, and to seek to improve the performance of all aspects of an ultrasound service. QA is an essential aspect of clinical governance as it is a risk management tool for ensuring minimum standards of practice and performance are attained. This provides reassurance that all patients have access to high-quality ultrasound services wherever they are.
A comprehensive quality assurance programme should consider:
QUALITY ASSURANCE TESTING
Equipment Performance Testing
The performance assessment and QA of diagnostic ultrasound scanners can also help to:
In the UK, the Institute of Physics and Engineering in Medicine (IPEM) has produced guidelines for the routine QA of ultrasound imaging systems (Price 1995). The IPEM report advocates three levels of QA testing:
User tests
These are to be carried out at frequent intervals (between 1 to 4 weeks) by the operator, in order to discover any significant changes in scanner performance over time. It includes a number of simple tests which require a minimum amount of time to carry out. These tests relate to the aspects of the scanner function on which users depend for clinically meaningful results and include simple checks, such as testing caliper accuracy.
Equipment Required for Image Performance Testing
Tissue equivalent (TE) phantoms
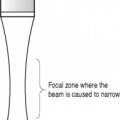
Commercially available test phantoms are normally filled with an aqueous gel or are formed from urethane materials. They contain various targets and structures designed for testing a number of parameters. Examples of some commercially available test phantoms are illustrated in Figure 13.1.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree