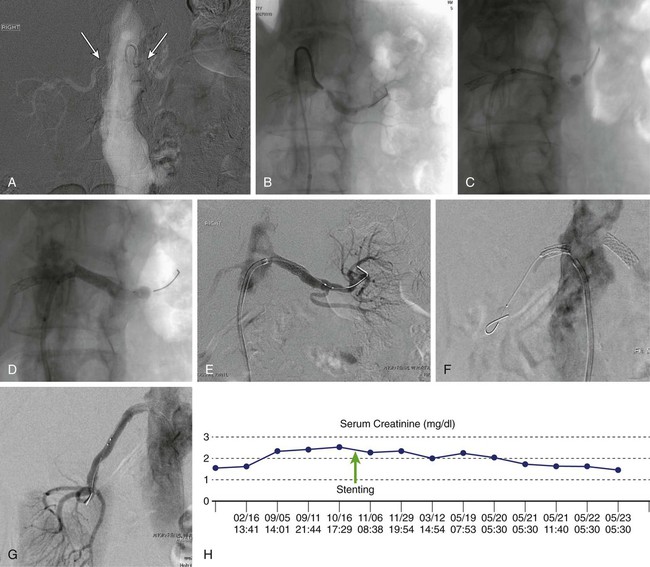
Renal artery stenosis (RAS) is a well-characterized condition caused by narrowing of one or both renal arteries due to atherosclerosis, fibromuscular dysplasia, or arteritis. Atherosclerotic disease is the predominant cause of RAS, and it is likely worldwide prevalence will continue to grow as average life expectancy rises.1 RAS is both a sequela of generalized atherosclerosis and a marker for subsequent atherosclerotic clinical events.2,3 Although some degree of RAS is extremely prevalent in elderly patients, patients with peripheral or coronary artery disease,4,5 or in autopsy samples,6 the clinical presentation is variable and often unpredictable. As a result, there is currently tremendous debate regarding the role of imaging and intervention in patients with RAS. The intrinsic physiologic alterations occurring in experimental RAS were first described by Harry Goldblatt in 1934.7,8 Over the past decade, there has been an increasing appreciation that the pathophysiology of RAS may involve activation of a much more complex neurohumoral response than originally described.8–10 RAS may be a silent “asymptomatic” finding or be associated with several related and overlapping clinical syndromes including renovascular hypertension (RVH), impaired renal function (also called ischemic nephropathy), and cardiac decompensation.11 Investigation is ongoing, but as yet, no reliable biomarkers for RVH have been identified. Measurements of elevated serum brain natriuretic peptide (BNP > 80 pg/mL) have been reported as predictive of a blood pressure response to revascularization,12 although this has not been consistently reproduced in clinical studies.13,14 RVH is the most common correctable form of hypertension and is estimated to occur in 12% to 25% of the hypertensive population.3 Clinical clues suggestive of RAS are used to define individuals warranting more detailed evaluation with noninvasive imaging or arteriography: onset of hypertension prior to age 30 (suggestive of renal artery fibromuscular dysplasia) or over age 50; accelerated, malignant, or multidrug resistant hypertension; and hypertension associated with recurrent episodes of heart failure (Table 58-1). Asymmetric renal atrophy may also be noted. Clinical prediction rules have been applied to identify a cohort of patients with approximately 30% likelihood of RAS, using an algorithmic evaluation of clinical variables that include smoking history, gender, age, presence of dyslipidemia, body mass index, serum creatinine (SCr), presence of an abdominal bruit, and existence of known atherosclerosis in other vascular beds.15 There is a linear correlation between severity of coronary artery disease (CAD) found during coronary angiography and the presence of RAS, as well as overall mortality risk.16 In patients undergoing cardiac catheterization, factors associated with concurrent RAS include peripheral arterial disease, a widened pulse pressure, dyslipidemia, older age, renal insufficiency, and more severe CAD.17 In particular, the presence of right CAD may be a marker for RAS.18 TABLE 58-1 Indications for Renal Angiography ACEI, Angiotensin-converting enzyme inhibitor; ARB, angiotensin receptor blocker. RAS is also a well-known cause of ischemic nephropathy and has been cited as a putative cause of declining renal function in 25% to 58% of patients.19 Although difficult to determine with certainty, ischemic nephropathy may also be a relatively common and treatable cause of end-stage renal disease. Earlier studies in which patients initiating renal replacement therapy were audited for renovascular disease found RAS in 27% to 45%.20–22 Characteristics suggestive of underlying RAS in this population include angiotensin inhibitor–induced renal failure, unexplained rapid declines in renal function, and an absence of proteinuria.23 Finally, RAS is a known cause of recurrent heart failure24–26 and may also contribute to poorly controlled angina in patients with nonreconstructable CAD.27 Magnetic resonance angiography has shown significant unilateral or bilateral RAS in nearly half of all patients presenting with flash pulmonary edema.28 In these individuals, the presence of predominantly diastolic dysfunction and elevated serum endothelin levels may be suggestive.29,30 Indications for imaging and intervention for RAS have been reported in a consensus document published by the Society of Interventional Radiology31 and the American College of Cardiology.32 The document also offers guidelines for appropriate use of screening renal angiography at the time of coronary angiography33 for patients in whom clinically important RAS is suspected. Incidental nonselective or selective renal angiography without a clinical indicator is not recommended. In general, three criteria should be fulfilled to support an indication for renal revascularization: a suitable clinical scenario, anatomic stenosis of at least 50% with measurement of a trans-stenotic pressure gradient of 20 mmHg in uncertain cases, and safe and reasonable anatomic conditions for access and therapy. Proper patient selection has been shown to optimize outcomes34; conversely, nonrigorous adherence to the treatment of patients with appropriate indicators has been a principal source of criticism for both cohort and randomized trials failing to show benefit from renal intervention.35,36 In particular, lack of adherence to trans-stenotic pressure measurements as a determinant for intervention in hypertensive patients is a major failing in many reported studies. In this regard, it is important to realize that the visual estimate as well as quantitative angiography of stenosis is often unreliable for evaluating the hemodynamic relevance of a lesion. While other methods have been proposed for assessing lesion severity, trans-stenotic gradients have been repeatedly validated as the simplest and most reliable technique. In a recent study comparing hyperemic systolic gradient (HSG), fractional flow reserve (FFR), quantitative angiography, and intravascular ultrasound, HSG had the best receiver operating characteristic curve for predicting blood pressure response to renal artery stenting.37 Although it is impossible in any individual to know the exact magnitude of a pressure difference across a renal artery lesion that is hemodynamically relevant and has clinical effect, a resting or hyperemic (after intrarenal vasodilator administration) trans-stenotic gradient of at least 20 mmHg has been validated in experimental and clinical trials to be associated with a greater likelihood of improved clinical outcomes.38–40 The techniques of renal artery stenting have evolved over the past decade. Modern methods incorporate specially shaped guides and sheaths that better align with the renal ostium, lower-profile wires for lesion traversal, pressure-sensing wires for measuring trans-stenotic gradients, and dedicated stents.41 Renal artery stents are balloon expandable and embedded into their delivery balloons, facilitating a low profile, and have structural variation producing proximal reinforcement to better efface resistant aorto-ostial lesions. Both over-the-wire and monorail stent delivery systems are available.42 Drug-eluting stents show trends toward reduced restenosis but have not been widely accepted for routine use,43 although they have demonstrated clinical value for small and branch RAS.44 Careful preintervention planning is necessary to assure the highest possible technical success. The best route for crossing the lesion should be carefully assessed, and flush aortography is recommended to assess the severity of aortic atherosclerosis as a determinant of both the safety of intervention (due to cholesterol or plaque embolization) and selection of technique and guiding catheters optimized to the renal artery anatomy. Arteries with a sharp caudal angulation from the aorta may be approached from an upper extremity access.45,46 For mild or equivocal lesions, trans-lesional pressure measurements should be performed to confirm the hemodynamic significance prior to intervention. Pharmacologic adjuncts are important for preventing procedure-related complications. Patients should be adequately hydrated. Oral or intravenous N-acetylcysteine or parenteral bicarbonate saline infusions may be of value for preventing contrast-induced nephropathy in patients with renal insufficiency.47 In these patients, carbon dioxide (CO2) angiography can also be used to augment or replace conventional contrast arteriography to decrease iodinated contrast exposture.48 Antiplatelet therapy, generally with clopidogrel loading, is initiated prior to the procedure. Heparin sulfate or bivalirudin should be administered to provide full anticoagulation prior to crossing the RAS, as well as periodically during prolonged catheter and device manipulation; if available, measuring activated clotting time is useful to ensure adequate levels of anticoagulation. Intraarterial nitroglycerin, verapamil, dopamine, or papaverine can be administered into the renal artery both for measurements of the hyperemic systolic gradient and to limit wire- and catheter-induced vasospasm. Verapamil (1-2.5 mg) should be used with caution in patients with first-degree heart block. There are several basic steps for aorto-ostial renal artery stenting. Once a stenosis is identified by aortography, the lesion is carefully and atraumatically crossed using a floppy-tipped 0.014- to 0.035-inch guidewire and an appropriately shaped angled-tip catheter. The goal is to have the guiding sheath or catheter aligned with the aortic ostium to prevent inadvertent scraping and dislodgment of cholesterol plaque from the aortic wall. In patients with markedly atherosclerotic or ecstatic aortas, a “no touch” technique may be used, utilizing a second stiffer wire to stabilize the guide away from the aortic wall while the working wire is passed across the RAS.49 Depending upon the balloon or stent system used, predilation or placement of a second “buddy” wire may be needed to allow the stent to pass across the stenosis. The shortest possible stent that will cover the target lesion plus 1 to 2 mm on each side and with a diameter equal to the normal width of the target renal artery is positioned across the stenosis and is slowly inflated, with care taken to avoid balloon sliding that may risk dissection. It is essential that the renal artery is imaged in the proper oblique projection so that the lesion is exactly in profile, especially if the lesion is ostial. The goal is to place the stent so that it covers the lesion and extends 1 to 2 mm past it on both sides. For ostial lesions, the proximal end should extend 1 to 2 mm into the aorta to ensure that it covers the ostium adequately. After deflation, the balloon is removed while maintaining guidewire position across the treated site. Completion angiography is performed via the guiding catheter or sheath. If there is a satisfactory angiographic appearance and no residual transstenotic gradient, the guidewire is carefully removed (Fig. 58-1). A total complication rate as high as 35% has been reported for renal interventions, although most events are minor.50 Serious complications usually occur in less than 5%,31 although major adverse events can occur in 8% to 13% of patients with advanced azotemia or severe aortic disease.51 The most common complications are puncture site related (1%-2% transfusion risk) and worsening renal insufficiency. This latter event occurs in approximately 5% with normal renal function, and up to 25% of patients with extant renal insufficiency. Progressive renal insufficiency may be due to progression of intrinsic renal pathology, contrast-induced nephropathy, or embolization. Development of overt renal infarction is rare, occurring in approximately 1% of patients. Guidewire perforations are almost always self-sealing, and peripheral perforations that do not stop bleeding can be treated by subsegmental transcather occlusion using coils or gelfoam. Renal artery rupture after balloon inflation or stenting occurs in less than 1% of cases and can potentially cause renal loss or death. Prompt recognition and reinflation of a percutaneous transluminal angioplasty balloon or an occlusion balloon across the site of rupture is critical until definitive therapy is undertaken. The availability of stent grafts allows most cases to be salvaged by endovascular therapy. There is uncertainty as to the value of routinely using distal embolic protection (DEP) devices during renal artery stenting. Embolization of both atherosclerotic debris and cholesterol crystals has been identified during all stages of renal artery interventions using both ex vivo modeling and intraprocedural duplex sonography.52,53 In one report, microembolic Doppler signals were seen in all cases of renal artery intervention and were especially prominent when stenting was performed compared with percutaneous transluminal renal angioplasty (PTRA) alone; the most striking embolization occurred with poststent dilation and flaring.53 Clinical series have described captured debris in 100% of procedures when using occlusion balloon protection systems (Guardwire [Medtronic, Santa Rosa, Calif.]) and 80% with filter-type DEPs,54 with 99% stabilization or improvement in renal function observed out to 2 years after stenting with the use of DEP. However, other reports have been less favorable, and there are considerable limitations to the regular use of DEPs. Since no DEP has been specifically designed for use in the renal arteries, the available configuration of the devices and the need for a relatively long “landing zone” for filters precludes their use in many cases. In addition, protection devices tend to have a larger crossing profile that may not easily traverse highly stenotic lesions, and they further tend to be relatively bulky and rigid, with a perceived risk of associated renal artery spasm or dissection. New devices with unique designs allowing shorter overall filter lengths and improved embolic capture have been described but are not routinely used or yet indicated for use in renal intervention.55 Although not yet validated as a clinical recommendation, there is emerging evidence that plaque characteristics identified using intravascular ultrasound-derived virtual histology (VH) may predict procedural embolization and perhaps guide decisions regarding DEP use.56–58 In these studies, the finding of a necrotic core at VH has been associated with postprocedure embolization and diminished renal function.56 In addition, measurements of renal frame rate—the number of images (at 30 frames/s) to opacify the terminal renal arteries from an ostial contrast injection—show slowed flow and diminished perfusion (renal “blush”) following stenting in arteries with VH characteristics of a necrotic core.57,58 The ability to objectively and noninvasively measure renal artery plaque characteristics and their effects on the renal parenchyma using cross-sectional imaging would offer conceptual advantages and possible clinical utility, without requiring initial passage of an intravascular ultrasound catheter across a potentially emboligenic plaque.59 Notably, there is recent evidence that a combination of both DEP and aggressive intraprocedural antiplatelet therapy with glycoprotein 2B3A receptor inhibitors is necessary to optimally preserve renal function during stent placement.60,61 These findings suggest that intrarenal platelet activation, either after primary platelet plug embolization or secondary to cholesterol/plaque embolization, may be the critical pathophysiologic sequence responsible for the observed renal functional declines in many cases. Overall, 12% of the U.S. population has treatment-resistant hypertension,62 and it is estimated that up to one quarter of these have RAS. Overall, blood pressure response after renal artery stenting (RAST) is seen in approximately two thirds of patients.63 Meta-analysis of earlier cohort series,64 as well as the five completed industry premarket approval RAST trials, have shown both early and sustained blood pressure benefit (Table 58-2).65–69 In a recent Swedish study of 234 patients undergoing RAST, both systolic and diastolic blood pressure (SBP/DBP) were improved on follow-up out to 4 years.70 Characteristics of patients experiencing hypertension benefit include higher baseline stenosis and trans-stenotic gradient,39 more elevated initial SBP and DBP, preserved parenchyma, and a shorter duration of hypertension.71,72 Better long-term blood pressure response has been described in men and patients with a glomerular filtration rate (GFR) above 40 mL/min.73 A contemporary evaluation of 149 patients followed over a course of 9 years found drug-resistant hypertension (patients receiving ≥ 4 antihypertensive medications), use of clonidine, DBP greater than 90 mmHg, and larger renal volume to be the best determinants of favorable blood pressure after treatment.74 However, randomized trials have not consistently demonstrated better blood pressure outcomes in patients treated by RAST compared with medical therapy alone.75–77 In light of this controversy, one strategy that has been proposed is to reserve intervention for patients with RAS and evidence of accelerated or malignant hypertension or end-organ injury including heart failure, stroke, or declining renal function.78 TABLE 58-2 Primary Outcomes from Prospective Clinical Trials of Renal Artery Stenting
Renovascular Interventions
Epidemiology
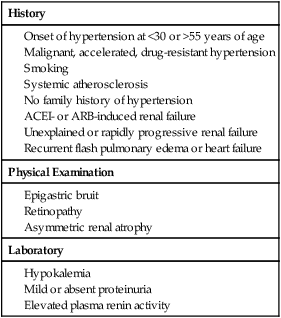
History
Physical Examination
Laboratory
Clinical Indications
Technical Details
Complications
Distal Embolic Protection
Reported Outcomes and Predictors
Hypertension
Trial
Year
Patients/Lesions
Follow-Up
Baseline (Mean Values)
Follow-Up (Mean Values)
Restenosis
SBP*
DBP*
SCr (GFR)
SBP*
DBP*
SCr (GFR)
Premarket Approval Studies
RENAISSANCE66
2008
100/117
3 yr
157
75
1.39 (51.49)
139†
71†
1.40 (53.68)
21.3%
ASPIRE-267
2005
208/244
2 yr
168
82
1.36
149†
77†
1.46
16.8%‡
SOAR68
2010
188/188
11 mo
160
77
1.15
135†§
74§
1.29§
12.6%‡
REFORM69
NP
100/115
2 yr
150
74
1.3
136†
73
N/A
8.1%‡
HERCULES70
NP
202/241
9 mo
162
78
1.2 (58)
145†
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree

 Get Clinical Tree app for offline access
Get Clinical Tree app for offline access