- •
Look for signs of fetal hydrops such as pericardial effusion, pleural effusion, ascites, or scalp edema.
- •
Assess heart size through cardiothoracic ratio measures.
- •
Evaluate ventricular systolic function.
- •
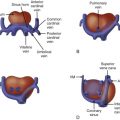
Assess size of the inferior vena cava (which may enlarge due to lower body increased venous return).
- •
Evaluate for tricuspid and mitral valve regurgitation.
- •
Assess umbilical arterial Doppler flow pattern, with particular attention to the diastolic component.
- •
Evaluate ductus venosus flow pattern.
- •
Evaluate umbilical venous flow pattern.
- •
Calculate the combined cardiac output measures and follow serially at regular intervals.
Anatomy, Frequency, and Development
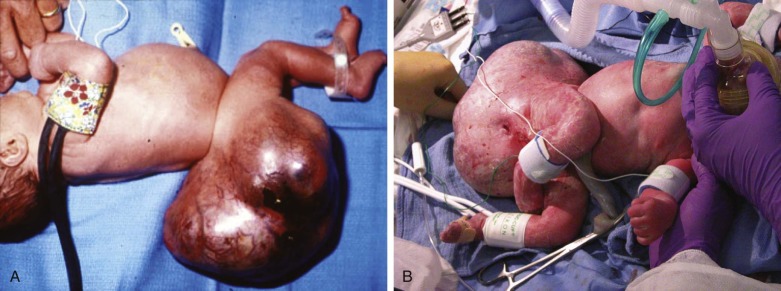
Sacrococcygeal teratoma (SCT) is a tumor originating from the sacral-coccygeal region of the body. It is defined as a neoplasm composed of all three primordial tissue germ layers or a neoplasm formed from multiple tissues that are foreign to that part of the body, lacking in any organ specificity. SCT is believed to arise from totipotent somatic cells in the caudal region of the developing embryo, which in some manner, escape the normal inductive influences of controlled differentiation and evolve into a growing mass of tissue. These tumors can grow wildly into enormously large structures with potential for invasion into the pelvic and abdominal cavities.
Although rare, SCT is the most common tumor seen in the fetus and neonate. The reported incidence is 1 in 35,000 to 40,000 live births. The natural history of prenatally diagnosed SCT differs from that of postnatally diagnosed SCT. Malignant degeneration is the primary cause of death in the postnatal SCT, which is a rare phenomenon in the fetus. The high mortality rate of fetal SCT is attributed to the effects of tumor mass on dystocia, preterm labor due to secondary polyhydramnios, and fetal hydrops and placentomegaly related to the cardiovascular effects of high-output failure. There are also reports of spontaneous rupture or hemorrhage with subsequent fetal anemia.
Prenatal Physiology and Diagnosis
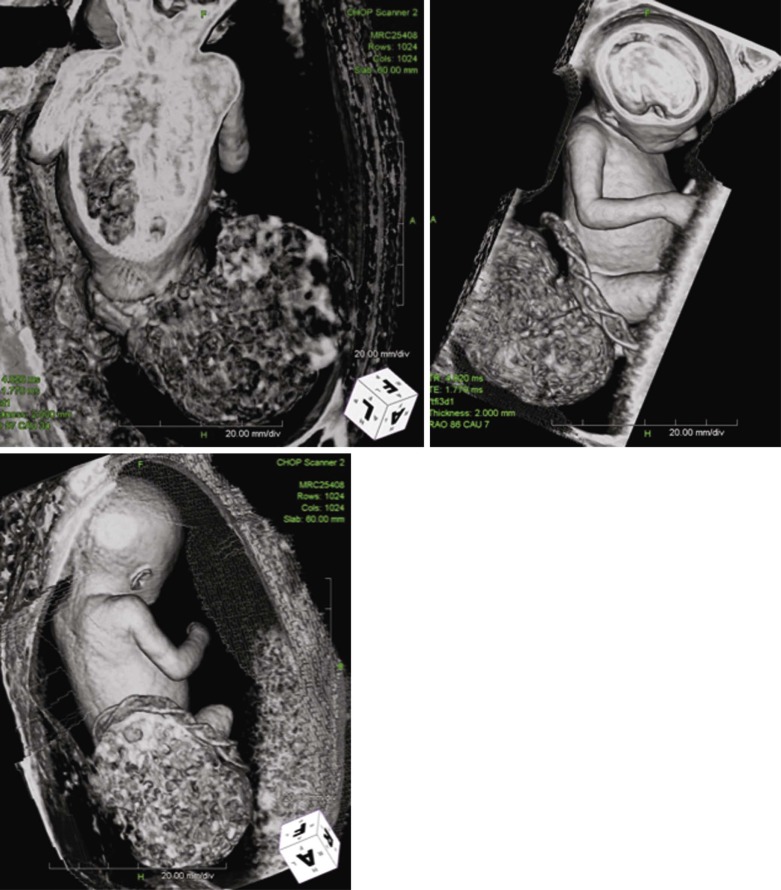
SCT can grow substantially and achieve a massive size ( Figure 40-1 ). The impact of SCT on the fetal cardiovascular system is due to the highly vascularized nature of the tumor. As the tumor grows, so does its vascular supply. In addition, the tumor mass itself can be inherently highly vascularized with multiple arterial to venous connections, effectively creating a giant arteriovenous malformation (AVM).
Fetal SCT influences prenatal cardiovascular physiology in a number of ways. Perfusion of the vascularized mass results in an increase in total fetal circulating blood volume and increased preload to the heart. The heart accommodates the volume load by compensatory dilation and increase in myocardial mass. Furthermore, the SCT mass can act as a low vascular resistance “sink.” Because the arterial blood supply originates from the iliac region of the descending aorta, the SCT may compete with blood flow to the placenta. Placental vascular resistance is normally very low; however, if the SCT mass is large and highly vascularized, its resistance may be lower than that of the placenta, creating a “steal” phenomenon, shunting blood away from the placenta.
Overall, the deleterious impact of SCT is in creating a state of high-output heart failure, because the fetal heart tries to compensate for the massively increased volume load. Ventricular dilation can lead to atrioventricular annular dilation and induce atrioventricular valve regurgitation, further exacerbating the volume load. As the heart fails, atrial pressures rise and hydrops ensues. When present, hydrops in a fetus with SCT typically leads to in utero demise, unless prenatal intervention is undertaken.
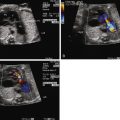
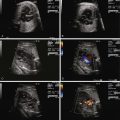
SCT size and makeup can be assessed by ultrasound and magnetic resonance imaging techniques ( Figure 40-2 ). Large tumors may consist mostly of cystic structures, which may have little vascularization or the degree of vascularization may be quite high, heralding the possibility of high-output heart failure.
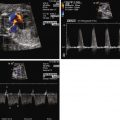
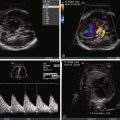
Fetal echocardiography plays a key role in the evaluation and serial surveillance of the fetus with SCT. Heart size should be assessed by measurement of cardiothoracic ratio. Overall ventricular systolic function should be noted. Inferior vena cava dilation is common, because lower body venous return is increased. Flow patterns in the ductus venosus and umbilical vein may reflect increased atrial pressure and worsening heart failure. Doppler evaluation of the umbilical artery will reveal a decrease in diastolic flow, or perhaps even reversal of flow, if there is a substantial competitive steal away from the placenta into the SCT. Because the essence of the pathophysiology of SCT relates to high-output failure, Doppler derived measures of cardiac output are very important. By measuring the diameter across the aortic and pulmonary valves and sampling blood flow using Doppler interrogation, one may calculate the individual ventricular and combined cardiac outputs by the formula: cardiac output = valvar cross-sectional area (3.14 × radius 2 ) × velocity-time integral of flow across the valve × heart rate. Indexed combined cardiac output in the normal fetus is relatively stable throughout gestation and is approximately 400 to 500 mL/min/kg.
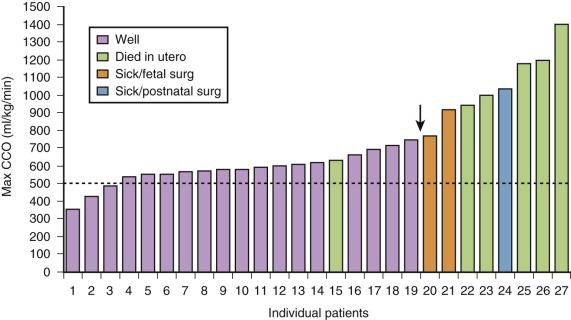
In a series of fetuses with SCT and potential for high-output failure, we evaluated combined cardiac output measures and found that most fetuses tolerate increased combined cardiac output to levels that approach 750 to 800 mL/min/kg, nearly twice the normal. When values much above 800 mL/min/kg are reached, the likelihood of cardiovascular instability, heart failure, and hydrops increases ( Figure 40-3 ). Serial evaluation of fetal echocardiographic parameters is part of a sound protocol for monitoring the fetus with SCT and determining the need for fetal intervention.
Prenatal Management
A number of therapies are available for treating SCT in the fetus. Amnioreduction in cases of polyhydramnios may improve maternal comfort and prevent premature labor. If the mass consists of sizable cystic structures, the tumor size can be reduced through cyst aspiration. Percutaneous laparoscopic laser ablation and interruption of vascular supply to the SCT is feasible. Alcohol sclerosis of the tumor mass has also been reported.
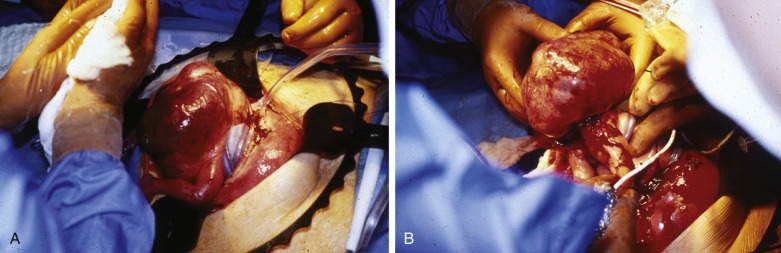
Surgical debulking through the techniques of open fetal surgery is one possible form of therapy. Fetal surgery for this anomaly is indicated only if the life of the fetus is threatened by the tumor and other options are not available. We performed fetal surgery in a series of four fetuses with giant SCT and evidence of high output failure at 21 to 26 weeks’ gestation. The procedure included hysterotomy with manipulation of the fetus to bring the SCT up to the uterine incision for removal ( Figure 40-4 ). Continuous intraoperative fetal echocardiography was performed to monitor the cardiovascular system during these procedures. Figure 40-5 shows the Doppler-derived values for combined cardiac output before surgery (time #1) and at induction of anesthesia (time #2), after SCT removal (time #3), and after surgery (time #4). Before surgery, the mean combined cardiac output was markedly elevated at over 800 mL/kg/min. With induction of anesthesia for the surgery, there was a slight decrease in combined cardiac output likely related to the depressive effect of the agents administered; however, values still remained abnormally high. Immediately after removal of the mass, there was a dramatic drop in cardiac output to levels below normal, which persisted into the postoperative period.