- •
CAV remains a major limitation for the long-term success of cardiac allografts.
- •
The high prevalence of renal dysfunction has limited the use of invasive coronary angiography.
- •
The evaluation of asymptomatic and symptomatic individuals with suspected or known CAV is now frequently carried out with various noninvasive imaging modalities.
- •
DSE is a highly specific, nonexpensive, radiation-free, contrast-free modality but has limited sensitivity to detect CAV.
- •
CCTA is very sensitive and has modest specificity but, like ICA, its use may be limited to patients with preserved renal function.
- •
MPI with CMR, SPECT, and PET appears to have modest sensitivity and relatively good specificity. The sensitivity of PET is significantly increased, however, when coupled with absolute quantification of MBF.
- •
Multiparametric PET, which includes myocardial perfusion, flow quantification, and an LV function assessment, appears to be particularly suited for CAV detection.
- •
Serial assessment of CAV with PET may detect patients in whom screening may be spaced out to every 2 to 3 years.
Introduction
Despite significant improvements in the management of heart transplant recipients, cardiac allograft vasculopathy (CAV) remains a major limitation for the long-term success of orthotopic heart transplantation (OHT) and is one of the leading causes of morbidity and mortality beyond the first year after OHT. CAV is manifested by progressive and diffuse narrowing of the coronary lumen, leading to the development of myocardial ischemia, fibrosis, diastolic dysfunction, and, eventually, graft failure and death.
Annual surveillance with invasive coronary angiography (ICA) to assess the presence and severity of CAV remains standard care at most transplant centers. Nevertheless, because of its costs and inherent risks and complications, noninvasive testing using echocardiography, cardiac computed tomography (CT) angiography (CCTA), cardiac magnetic resonance (CMR) imaging, single photon emission computed tomography (SPECT), or positron emission tomography (PET) has emerged as an important clinical alternative to ICA. In this chapter, we will review the advantages and limitations of different approaches for CAV screening. We will focus on radionuclide techniques and discuss the role of alternative approaches used alone or in combination.
Clinical aspects of cardiac allograft vasculopathy
Pathophysiology
CAV is a condition characterized by panarterial derangement, including adventitial fibrosis, diffuse concentric intimal proliferation and hyperplasia of the epicardial coronary arteries, and medial thickening of the smaller intramyocardial coronary vessels, which results in diffuse vessel narrowing, focal epicardial coronary stenoses, and obstructive microvasculopathy. , This is in contrast to the more focal lesions of traditional atherosclerosis, which tend to be noncircumferential and commonly involve the proximal segments of the epicardial coronary arteries. Nevertheless, superimposed atherosclerotic coronary lesions can coexist with CAV and have some overlap in their angiographic appearance. Histopathologically, atherosclerotic plaques are usually heterogenous and contain a distinctive fibrous cap (composed of macrophages, cholesterol clefts, and calcium deposits), and the lumen is eccentric, whereas allograft lesions tend to be more homogenous and symmetrically located with a composition mainly of fibrous tissue, and the lumen is more centrally located.
The pathogenesis of CAV is multifactorial and appears to be triggered by both immune-related mechanisms and nonimmunologic vascular risk factors that ultimately result in inflammatory vascular injury and perivascular fibrosis. Some of the risk factors associated with the development of CAV include cellular and antibody-mediated rejection, donor-specific antihuman leukocyte antigen (HLA) antibodies, cytomegalovirus infection, dyslipidemia, hypertension, recipient pretransplant diagnosis of ischemic cardiomyopathy, and older age.
Definition, epidemiology, diagnosis, and prognosis
In 2010, the International Society for Heart and Lung Transplantation (ISHLT) established a nomenclature for the definition of CAV based on information derived from ICA, in combination with markers of cardiac allograft function ( Table 22.1 ). With this system, patients are divided into four groups:
- •
ISHLT CAV 0 includes patients with angiographically normal coronary arteries;
- •
ISHLT CAV 1 refers to patients with nonobstructive disease (<70% luminal narrowing);
- •
ISHLT CAV 2 includes patients with single-vessel obstructive CAV (≥70%); and
- •
ISHLT CAV 3 includes patients with multivessel obstructive CAV and those with ISHLT CAV 1 or CAV 2 who also have graft dysfunction as evidenced by either left ventricular systolic dysfunction or restrictive physiology on right heart catheterization or echocardiography.
| Author, Year | Test | N | ISHLT Reference | Prevalence | Sensitivity (%) | Specificity (%) | PPV (%) | NPV (%) |
|---|---|---|---|---|---|---|---|---|
| Clerkin 2016 | DSE | 221 | CAV 1–3 | 13% | 0 | 99 | 0 | 82 |
| Sade 2014 | DSE | 23 | CAV 1–3 | 39% | 56 | 64 | 50 | 69 |
| Chirakarnjanakorn 2015 | DSE | 497 | CAV 1–3 | 38% | 7 | 98 | 82 | 41 |
| Chirakarnjanakorn 2015 | DSE | 497 | CAV 2–3 | 8.6% | 28 | 98 | 71 | 89 |
| Clemmensen 2015 | GLS | 178 | CAV 1–3 | 39% | 42 | 93 | 78 | 72 |
| Wever-Pinzon 2014 (Meta-analysis) | CTA | 203 | CAV 1–3 | 43% | 97 | 81 | 78 | 97 |
| Günther 2018 | CAC | 133 | CAV 2–3 | 12% | 88 | 50 | 19 | 97 |
| Colvin-Adams 2011 | MPI with CMR | 68 | CAV 1–3 | 34% | 41 | 74 | 45 | 63 |
| Manrique 2010 | MPI with SPECT | 110 | CAV 1–3 | 42% | 63 | 78 | 67 | 75 |
| Manrique 2010 | MPI with SPECT | 110 | CAV 2–3 | 17% | 84 | 70 | 37 | 96 |
| Wenning 2012 | MPI with SPECT | 162 | CAV 2–3 | 13% | 62 | 81 | 33 | 93 |
| Bravo 2018 | MPI with PET | 66 | CAV 2–3 | 18% | 83 | 82 | 50 | 96 |
| Miller 2020 | MPI with PET | 99 | CAV 2–3 | 14% | 64 | 93 | 60 | 94 |
Some centers may also perform an intravascular ultrasound (IVUS) if the angiographic findings are inconclusive or insufficient to explain the clinical presentation. The general consensus is that detection of maximal intimal thickness of at least 0.5 mm, particularly in the left ascending anterior coronary artery, is consistent with CAV.
A recent report from the ISHLT Thoracic Organ Transplant Registry, which included 146,975 OHT survivors of all ages, found an overall CAV prevalence of 7.7%, 29%, and 46.8% within 1, 5, and 10 years post-OHT, respectively. Similar findings were described in a single-center study that used the recommended ISHLT CAV nomenclature in 501 adult recipients, with a prevalence for any CAV 1-3 of 9.8% at 1 year, 22% at 4 years, 44% at 10 years, 56% at 15 years, and 59% at 20 years. Of note, CAV 2-3 was rare within the first year (0.6%) and relatively uncommon at 4 years (3.9%), but its occurrence increased significantly at 10 years (13.7%), 15 years (18.7%), and 20 years (23.6%).
The distinction between CAV groups is clinically relevant and has important therapeutic and prognostic implications. For example, compared with CAV 0 , the relative risk for death or retransplantation was 1.22 (0.85 to 1.76; p = .28) for CAV 1 , 1.86 (1.08 to 3.22; p = .03) for CAV 2 , and 5.71 (3.64 to 8.94; p < .001) for CAV 3 among 501 patients with a mean follow-up of 11.9 ± 6.4 years. In a different study, 169 OHT recipients were followed for 5.6 ± 2.8 years for a composite of major adverse cardiovascular events (MACEs) and compared with CAV 0 ; the relative risk for MACE was 1.1 (0.3 to 4.1; p = NS) for CAV 1 , 3.6 (0.8 to 16.0; p = .088) for CAV 2 , and 9.2 (3.1 to 5.27; p <.01) for CAV 3 . Similar results have been observed in the pediatric population. Overall, these data highlight the high prevalence of CAV and the enhanced prognostic value of the ISHLT CAV grading system.
Clinical manifestations and treatment
CAV can affect people of all ages, including children, and is characterized by a heterogeneous clinical expression and diverse clinical course. For example, angina pectoris, the classic symptom of coronary obstruction leading to myocardial ischemia, is uncommon in OHT recipients because of the surgical transection of the presynaptic sympathetic nerve terminals that occurs after transplantation. Although myocardial re-innervation does eventually occur, this process is almost invariably incomplete. Thus, most individuals remain asymptomatic or report nonspecific symptoms, such as weakness, shortness of breath, and/or palpitations, whereas some patients may have more ominous clinical presentations related to more advanced CAV, including acute coronary syndrome, heart failure, and sudden death. In fact, CAV is the most common cause of graft failure and sudden death beyond the first year post-OHT. , ,
Management of CAV is aimed at preventing and treating established disease. In the former case, there is evidence that statin therapy improves OHT survival and may reduce the incidence of CAV. Once CAV is established, however, the options for treatment are more limited; nevertheless, observational data suggest that sirolimus, an inhibitor of mammalian target of rapamycin (mTOR), may slow down the progression of CAV. , Percutaneous and surgical coronary revascularization remain palliative options but can be considered in suitable patients with focal or multifocal epicardial stenoses. There is no evidence, however, that such procedures improve long-term outcomes. , Retransplantation appears to be the only viable option for the group of CAV patients who develop graft failure, with some data suggesting acceptable outcomes that are superior to those undergoing retransplantation for other causes.
Rationale for screening
Because the clinical manifestations of CAV can be subtle or absent, especially early in the disease course, periodic (usually annual) surveillance to evaluate for the presence and severity of CAV is recommended. ICA, with or without IVUS, is considered the cornerstone and accepted standard of care for establishing the diagnosis; however, ICA is an invasive procedure that is not free of complications. Moreover, the prevalence of renal dysfunction increases significantly over time in this population, with serum creatinine levels greater than 2.5 mg/dL documented in 5.1% and 12.2% of patients at 1 and 5 years post-OHT, respectively. Therefore, annual surveillance with noninvasive testing has become an alternative to ICA, especially after the first 5 years and/or in patients with a glomerular filtration rate (GFR) less than 30 mL/min/1.73m 2 .
Noninvasive imaging approaches for screening of cardiac allograft vasculopathy
Radionuclide imaging
Myocardial perfusion imaging (MPI) techniques with SPECT or PET, especially when coupled with absolute myocardial blood flow (MBF) quantification, are increasingly used for CAV detection. The ability to quantify MBF and myocardial flow reserve (MFR) represents a unique advantage of PET, which is well suited to interrogate the biology of diffuse transplant vasculopathy. Please refer to Chapters 2 , 3 , and 4 for a more detailed discussion regarding the principles and tools needed for quantitative perfusion imaging.
Historically, MPI with PET has demonstrated better performance compared with SPECT for the diagnosis of flow-limiting coronary artery disease in the nontransplant literature. , There are emerging data suggesting that PET, especially when combined with absolute MBF quantification, may also be superior for detection of CAV in OHT recipients.
The use of SPECT MPI had modest sensitivity (56% to 63%) and relatively good specificity (78% to 89%) to detect any CAV in studies totaling 350 OHT patients, , , with slightly higher sensitivity (62% to 84%) but lower specificity (70% to 78%) for the detection of moderate to severe CAV. Importantly, patients with abnormal SPECT MPI had a fivefold increase in the risk for cardiac death and retransplantation (relative risk 5.6, p =.0053). In contrast, patients with normal gated SPECT MPI had a low risk for future cardiac events.
PET MPI appears to have significantly higher specificity (82% to 93%) than but comparable sensitivity (64% to 83%) to SPECT for the detection of moderate to severe CAV. , Higher specificity and fewer false-positive results is a critical performance characteristic because many of these patients have marginal renal function and avoiding exposure to iodinated contrast media is of paramount importance. Likewise, abnormal PET MPI is predictive of adverse events. Miller et al. followed 99 OHT patients for a median of 3.4 years and found that a summed stress score (SSS) ≥4 was associated with all-cause mortality (log-rank p =.017). Similarly, Bravo et al. followed 94 patients for a median of 2.3 years and found that an SSS ≥2 was a univariate predictor of adverse outcomes, including death (hazard ratio [HR] 3.11 [1.26 to 7.66]; p =.014).
Screening with non-nuclear imaging modalities
Table 22.1 summarizes the evidence for all noninvasive tests available for CAV screening (using the ISHLT nomenclature as a reference). Among these, dobutamine stress echocardiography (DSE) is likely the most widely used modality and the noninvasive test with the greatest experience for CAV screening. DSE is radiation- and contrast-free and therefore is ideal for this patient population from a practical and safety perspective. The external validity of DSE was established more than 20 years ago, , but several recent studies have actually shown that DSE has a limited sensitivity (range 0% to 56%) to detect ISHTL CAV 1-3 , albeit an excellent specificity (range 64% to 98%). For example, one study observed a very low incidence of abnormal DSE, with only 1.8% of 1243 studies showing evidence of dobutamine-induced wall motion abnormalities consistent with ischemia, despite a CAV 2-3 prevalence of 8.6%. Additionally, DSE failed to predict future adverse events, a finding that highlights the insufficient sensitivity of DSE to detect CAV and provide incremental prognostic value. Similar findings have been reported for speckle-tracking peak systolic global longitudinal strain.
In contrast, and in resemblance to the literature of native coronary artery disease, CCTA is a more sensitive (range 70% to 100%, weighted average 97%) test with good specificity (weighted mean 81%) to detect ISHTL CAV 1-3 . , In addition, there is a significant association between coronary artery calcium (CAC) and CAV severity. However, in approximately 12% of cases with moderate to severe CAV, coronary calcium may be absent. Therefore, CAC alone cannot be used to exclude the presence of significant CAV. Like with ICA IV contrast use CCTA may not be feasible in patients with renal dysfunction, which according to some estimates represents 52% and 68% of the OHT population at 5 and 10 years, respectively, limiting the clinical utility of CTA as a screening tool. In addition, the clinical utility of CTA for serial assessment of CAV is unknown.
CMR imaging has also been used in this population; however, the literature of stress CMR is currently limited to a handful of studies that have shown a low sensitivity (41%) and only moderate specificity (74%) for detecting CAV. Moreover, subendocardial or transmural late gadolinium enhancement (LGE) in a pattern typical of coronary disease–related myocardial infarction (MI) is not uncommon in OHT patients (18% to 22%). , For instance, a study showed that this LGE pattern was present in 19%, 10%, 36%, and 71% of OHT patients classified angiographically as CAV 0 , CAV 1 , CAV 2 , and CAV 3 , respectively. These findings highlight some of the limitations of ICA for the detection of CAV, although it is also conceivable that alternative mechanisms of myocardial injury (resulting in LGE) other than CAV may exist (e.g., perioperative ischemic damage). Not surprisingly, the presence and extent of myocardial scarring are also independently associated with all-cause mortality and MACEs after transplantation.
Patient-centered applications of radionuclide imaging and multimodality imaging in patients after heart transplantation
Case vignette 1: Evaluation of patient with suspected clinical progression of cardiac allograft vasculopathy
A 63-year-old male with a history of ischemic cardiomyopathy underwent successful OHT at age 50. Except for hypertension, obesity, and sleep apnea, the patient had no major transplant-related complications, including preserved renal function and angiographically normal coronary arteries until his seventh annual evaluation when he had a 50% lesion in the mid left anterior descending (LAD) coronary artery (ISHTL CAV 1 ) and worsening renal function (GFR 50 mL/min/1.73 m 2 ) during his eighth annual screening visit. The patient subsequently developed low-effort dyspnea on exertion and worsening peripheral edema, yet a DSE performed during his ninth annual visit was unremarkable. The patient’s renal dysfunction (GFR 37 mL/min/1.73 m 2 ) and symptoms worsened over the next year. At that point, the patient underwent vasodilator stress 13 N-ammonia PET/CT MPI including flow quantification. MPI showed a medium-sized, moderately severe, fully reversible defect in the inferolateral wall, suggestive of flow-limiting coronary stenosis in the left circumflex (LCx) distribution ( Fig. 22.1 A and B). Quantitative flow demonstrated globally reduced stress MBF (0.96 mL/min/g) and MFR (1.53), indicative of an associated diffuse global CAV. Left ventricular (LV) systolic function was preserved (LV ejection fraction [LVEF] 50%). Overall, the PET findings were consistent with disease progression to moderate to severe CAV. This was confirmed on a subsequent ICA, which revealed subtotal occlusion of the first obtuse marginal (see Fig. 22.1 C) and severe distal tapering of the LAD, LCx, and right coronary artery (RCA) (see Fig. 22.1 C), all consistent with moderate to severe ISHTL CAV.
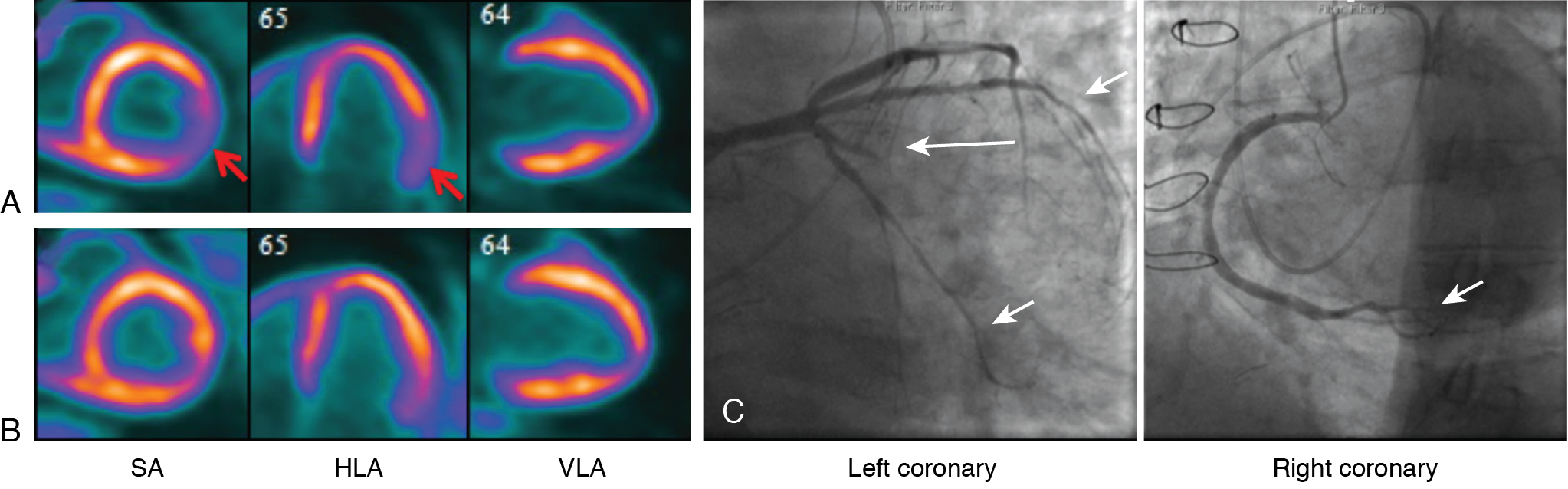
Multiparametric PET provides the unique opportunity to quantify regional and global MBF (complementary to semiquantitative visual analysis) noninvasively, potentially serving as an indicator of the integrity and function of the large and small coronary allograft vessels, which more closely reflect the pathobiology of CAV. In this regard, recent studies have consistently observed that MBF quantification by PET is a very sensitive marker to exclude significant CAV, albeit at the expense of reduced specificity ( Table 22.2 ). For example, Miller, Chih, and Bravo, using stress MBF cutoffs of 3.70, 2.30, and 1.70 (mL/min/g), found a sensitivity of 93%, 100%, and 92% and a specificity of 22%, 42%, and 77%, respectively, to diagnose CAV. The optimal cutoff in Miller’s study, however, was ultimately estimated at less than 2.83 (sensitivity 73% and specificity 56%). Interestingly, stress MBF also showed a trend for higher area under the curve (AUC) for the detection of moderate to severe CAV compared with MFR in Chih’s study (0.83 [0.70 to 0.97] vs. 0.71 [0.48 to 0.93]) and Bravo’s series (0.89 [0.78 to 0.99] vs. 0.80 [0.66 to 0.95]), suggesting that stress MBF may potentially be a better marker for CAV diagnosis than MFR. These data also underscore the challenges in developing unified MBF cut points, particularly for the diagnosis of CAV.
| Author, Year PET Tracer Study Size | Parameters (Single or Combined) | Reference | Prevalence | Sensitivity | Specificity | PPV | NPV |
|---|---|---|---|---|---|---|---|
| Miller 2020 82 rubidium (Rb) N = 99 | MFR <2.00 | CAV 2–3 | 14% | 71 | 72 | 29 | 94 |
| MFR <1.75 | CAV 2–3 | 14% | 57 | 78 | 30 | 92 | |
| Stress MBF <3.7 | CAV 2–3 | 14% | 93 | 22 | 17 | 95 | |
| Stress MBF <1.70 | CAV 2–3 | 14% | 43 | 95 | 60 | 91 | |
| SSS >1 | CAV 2–3 | 14% | 64 | 73 | 28 | 93 | |
| SSS >3 | CAV 2–3 | 14% | 64 | 93 | 60 | 94 | |
| MBF <1.7 and SSS >1 or LVEF ≤45% | CAV 2–3 | 14% | 43 | 98 | 75 | 91 | |
| SSS ≥4, EF ≤45%, or MFR <1.75 | CAV 2–3 | 14% | 71 | 69 | 28 | 94 | |
| LVEF ≤45% and MFR <1.75 | CAV 2–3 | 14% | 36 | 100 | 100 | 90 | |
| Chih 2018 82 Rb N = 40 | cMFR <2.9 a | CAV 1–3 | 35% | 71 | 54 | 45 | 78 |
| Stress MBF <2.3 | CAV 1–3 | 35% | 100 | 42 | 48 | 100 | |
| cMFR <2.9 a and stress MBF <2.3 | CAV 1–3 | 35% | 58 | 97 | 89 | 81 | |
| cMFR <2.9 a or stress MBF <2.3 | CAV 1–3 | 35% | 95 | 66 | 59 | 94 | |
| cMFR <2.9 a | MIT ≥0.5 | 80% | 69 | 88 | 96 | 41 | |
| Stress MBF <2.3 | MIT ≥0.5 | 80% | 84 | 75 | 93 | 55 | |
| cMFR <2.9 a and stress MBF <2.3 | MIT ≥0.5 | 80% | 58 | 97 | 99 | 38 | |
| cMFR <2.9 a or stress MBF <2.3 | MIT ≥0.5 | 80% | 95 | 66 | 91 | 71 | |
| Bravo 2018 13 N ammonia N = 66 | Stress MBF <1.70 | CAV 2–3 | 18% | 92 | 70 | 41 | 97 |
| MFR <1.70 | CAV 2–3 | 18% | 75 | 65 | 32 | 92 | |
| MFR <2.0 | CAV 2–3 | 18% | 83 | 48 | 26 | 93 | |
| SSS >1 | CAV 2–3 | 18% | 83 | 82 | 50 | 96 | |
| PET CAV 2/3 score b | CAV 2–3 | 18% | 83 | 93 | 71 | 96 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree