Fig. 1
Clinical pathologic course: prostate gland, seminal vesicles, and penis (with permissions from Rubin and Casarett 1968)
2 Anatomy and Histology
2.1 Anatomy
The distal male urogenital tract extends from the lower pelvis to the ventral portion of the body and includes the prostate gland, seminal vesicles, urethra, and penis. The testicles, which are also a part of the distal male urogenital tract, are addressed in a separate chapter. The primary roles of the distal urogenital tract are to generate components of the seminal fluid, serve as a conduit for urine and semen, and facilitate sexual intercourse. Highly detailed anatomic descriptions and atlases are available in both textbook (Gray 1995; McAninch 2008) and publication (Meyers 2001; Walz et al. 2010) form. It is increasingly clear that there can be significant variations between individuals in terms of the form and structure of the prostate gland, neurovascular bundles, external urethral sphincter, prostatic ligaments, and vascular supply to these structures.
2.1.1 Prostate Gland
The prostate gland lies within the lower pelvis immediately superior to the musculofascial floor. It is the largest accessory gland of the male reproductive tract, weighing approximately 30 grams in men without prostatic pathology. A fibrous capsule surrounds the gland, which is continuous with the surrounding connective tissue stroma, but is not present at the apex of the prostate. The fascia surrounding the prostate gland is variably adherent to the capsule. Shaped like an inverted tapered cylinder, the prostate base is immediately caudad to the bladder and its tapered apex cephalad to the urogenital diaphragm. The apex contains muscle fibers continuous with the urogenital diaphragm. The prostate lies posterior to the symphysis pubis, connected via the puboprostatic ligaments, and anterior to the rectum, separated from it by the anterior portion of Denonvilliers fascia (rectovesical septum), a thin layer of connective tissue that separates the prostate and seminal vesicles from the anterior rectal wall. The prostatic urethra passes through the gland from the base of the bladder to the membranous urethra. The ejaculatory ducts course from the convergence of the ducti deferens and the seminal vesicles obliquely, anteriorly and caudally, through the posterior prostate to communicate to join with the prostatic utricle to open into the prostatic urethra.
The blood supply of the prostate gland is from the prostatic branches of the inferior vesicle, pudendal, and middle rectal arteries. Venous drainage is shared with the penis, urethra, and lateral pelvic organs via the dorsal venous plexus (Santorini’s plexus) to the internal iliac vein. The predominant course of lymphatic drainage is to the internal iliac nodes, but alternative drainage routes to the obturator, external iliac, sacral, vesicle, and rarely the periaortic lymph nodes are common. The prostate gland has dual autonomic innervation from the prostatic nerve plexus. Parasympathetic innervation is provided by the pelvic splanchnic nerve (S2-4) and sympathetic innervation is provided by the inferior hypogastric plexus (T10-L2). The neurovascular bundle (NVB) courses between fascial layers on the posterior lateral surfaces of the prostate gland and is intimately related to the dorsal surfaces of the seminal vesicles.
The prostate has been anatomically described in terms of both lobes and zones (McNeal 1981). There are four lobes of the prostate: anterior, posterior, median, and lateral. The anterior lobe is located anterior to the urethra and contains no glandular tissue, just fibromuscular stroma. The median lobe lies in the center of the gland, with the urethra anteriorly and the ejaculatory ducts posteriorly. The paired right and left lateral lobes are the largest lobes and contain the bulk of glandular tissue, being separated into halves by the prostatic urethra. The posterior lobe lies at the dorsal portion of the gland and can be palpated via a digital rectal examination.
The zonal anatomy of the prostate (Fig. 2) was first described by McNeal in 1981 and is used more commonly. This system is based on embryonic development patterns and includes four prostatic zones described relative to the urethra: the peripheral zone, the central zone, the transition zone, and the anterior fibromuscular stroma. The peripheral zone is a horseshoe-shaped structure extending posteriorly and laterally around the inner regions of the prostate gland and contains approximately 70 % of the prostatic glandular tissue. It is embryologically derived from a double row of developing prostatic ducts that extend posterolaterally from the distal urethra. The central zone contains approximately 25 % of the glandular tissue and is formed from developing ducts that grow cephalad, posteriorly and laterally into the mesenchyme surrounding the ejaculatory ducts. It is a cone-shaped volume extending from the bladder base to the verumontanum, encompassing the ejaculatory ducts. The transition zone lays anteromedial to the central zone and surrounds the mid-prostatic urethra. Benign prostatic hypertrophy occurs most commonly in the transition zone. The fibromuscular stroma is the anterior part of the prostate and contains little to no glandular tissue but does account for one-third of the total bulk of tissue within the prostatic capsule.
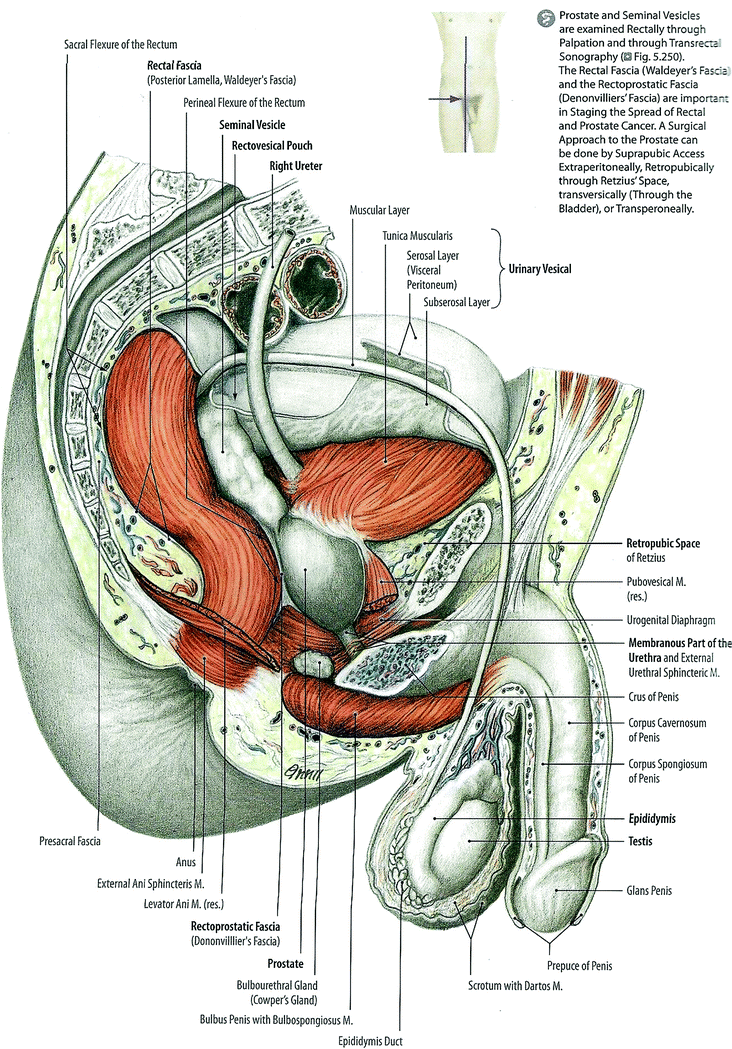

Fig. 2
Anatomy of the prostate (with permissions from Tillman 2007)
2.1.2 Seminal Vesicles
The seminal vesicles consist of two lobulated glands posterior to the bladder and prostate gland that converge medially. They are inferior and lateral to the ampulla of the ductus deferens and lie against the fundus of the bladder. The majority of these glands lie in the retroperitoneum and are enclosed by dense fascia, except for the most cephalad portion, which is intraperitoneal. Their cephalad surface abuts the posterior bladder and their caudad surface interfaces with the anterior rectum at the rectovesical (Denonvilliers’) fascia. The ureters lie medial to each seminal vesicle. The seminal vesicles converge with the vas deferens to form the ejaculatory ducts which enter the prostate gland. They produce alkaline secretions that are added to the semen. Their blood supply is from the inferior vesicle artery and vein and middle rectal artery and vein. Their main lymphatic drainage is to the internal iliac lymph nodes. Sympathetic innervation is via the superior lumbar and hypogastric nerve, which regulates rapid contraction of smooth muscle cells during ejaculation. Parasympathetic innervation is from the pelvic splanchnic nerve and from the inferior hypogastric plexus.
2.1.3 Male Urethra
The male urethra courses from the base of the bladder to the tip of the penis and is subdivided into prostatic, membranous, and spongy portions. It provides a conduit for urine and semen to be excreted from the body. The prostatic portion enters the anterior portion of the prostate gland and travels with slight anterior concavity so that it courses posteriorly toward the mid gland before resuming an anterior position at the prostatic apex. The change from the posterior to anterior course happens at a sharp, approximately 35-degree angle, at the level of the verumontanum, a midline protrusion along the posterior urethra where the ejaculatory ducts enter the prostatic urethra. The prostatic urethra is about 3-cm long and is the widest and most compliant portion of the urethra. The membranous urethra is approximately 1-cm long and extends through the urogenital diaphragm from the apex of the prostate gland to the bulb of the corpus spongiosum. The external urethral is a complex anatomic and physiologic structure closely related to the urogenital diaphragm. The innervation for the external sphincter travels near the apex of the prostate. The external sphincter is a circular muscle under partial voluntary control (innervated by the somatic nervous system) in the urogenital diaphragm and regulates the passage of urine through the urogenital diaphragm. Once the urethra enters the penile bulb, it becomes the spongy (penile) urethra which extends to the navicular fossa at the tip of the penis. The spongy urethra is about 6-cm long and is contained in the corpus spongiosum.
2.1.4 Penis
The penis is subdivided into three portions: the root, the body, and the glans. The root is anchored to the os pubis, symphysis pubis, and ischium by the crura and suspensory ligaments. The body of the penis extends between the root and the glans, a dome-shaped extension of the corpus spongiosum. The penis contains three erectile bodies, each encased in a fascial cover: two paired corpora cavernosa and the corpus spongiosum. All corpora are further encased together by the fibrous Buck’s fascia. The spongy urethra courses within the corpus spongiosum after entering this structure at the penile bulb, the most proximal portion of the spongiosum. The penile skin rests upon Colles’ fascia, which is continuous with Scarpa’s fascia of the abdominal wall.
The arterial supply of the penis and urethra is from the internal pudendal arteries which branch into the deep penile artery, the bulbourethral artery, and the dorsal artery of the penis. The deep artery supplies the corpora cavernosa, while the others supply the remainder of these organs, including the urethra and corpus spongiosum. There are multiple routes of venous drainage of the penis. The superficial dorsal vein is external to Buck’s fascia, while the deep dorsal vein lies deep into this thick fascial layer between the paired dorsal arteries. The dorsal veins join the pudendal plexus and eventually the internal pudendal vein. The penile skin and superficial fascia’s main route of lymphatic drainage is to the superficial inguinal lymph nodes. The predominant lymphatic pathway for the glans penis is through the external iliac nodes. The urethra drains to the internal and common iliac lymph nodes.
The anatomy of the pelvic floor is challenging to visualize on CT or MRI, and hence definition of the penile bulb varies. This may contribute to inconsistent reports (5–15). The penile bulb appears as an oval-shaped, hyperintense midline structure on T2-weighted MR images; on axial CT imaging it is bounded by the crura, corpora spongiosum, and the levator ani muscle. At University of California-San Francisco, the bulb is defined as the most proximal portion of the penis sitting immediately caudal to the prostate.
2.2 Histology
2.2.1 Prostate Gland
Many excellent textbook references are available with detailed descriptions of the histology of the organs of the distal male genitourinary tract (McAninch 2008; Bostwick 1998). Several cellular subtypes are evident upon histologic examination of the prostate, including basal, secretory, neuroendocrine, urothelial, and ejaculatory duct cells (Fig. 3a, b). Under the thin fibrous capsule of the prostate are smooth muscle layers and layers of collagen which also surround the prostatic urethra, forming the involuntary sphincter. Deeper to this layer is the prostatic stroma, which encases the glandular compound tubuloacinar secretory units, numbering 30–50. These units are arranged concentrically into three groups based on their relationship to the urethra. The majority of these ducts open on the floor of the prostatic urethra near the verumontanum. The main group is farthest from the urethra and constitutes the largest in number. The mucosal and submucosal glands, along with their adjacent stroma, undergo hyperplastic change with age, leading to compression of the urethra and symptoms consistent with benign prostatic hyperplasia (BPH).
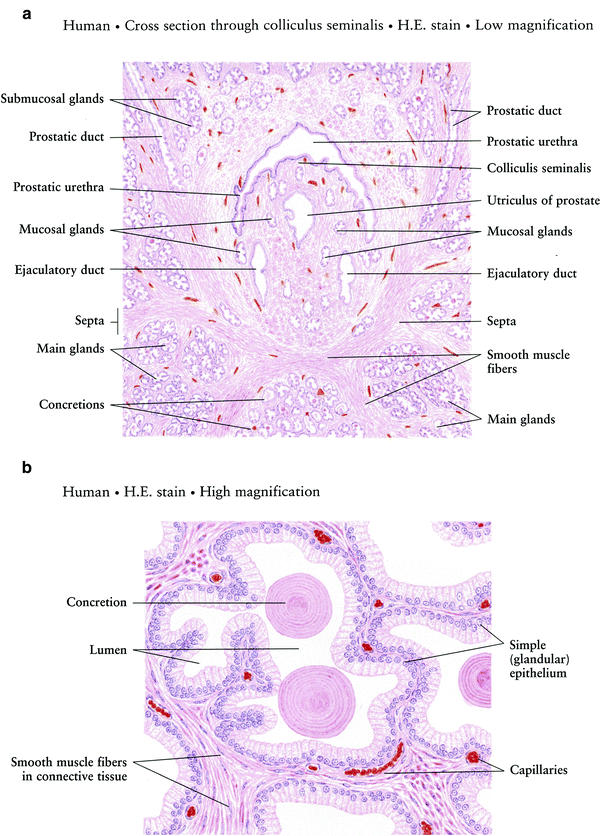
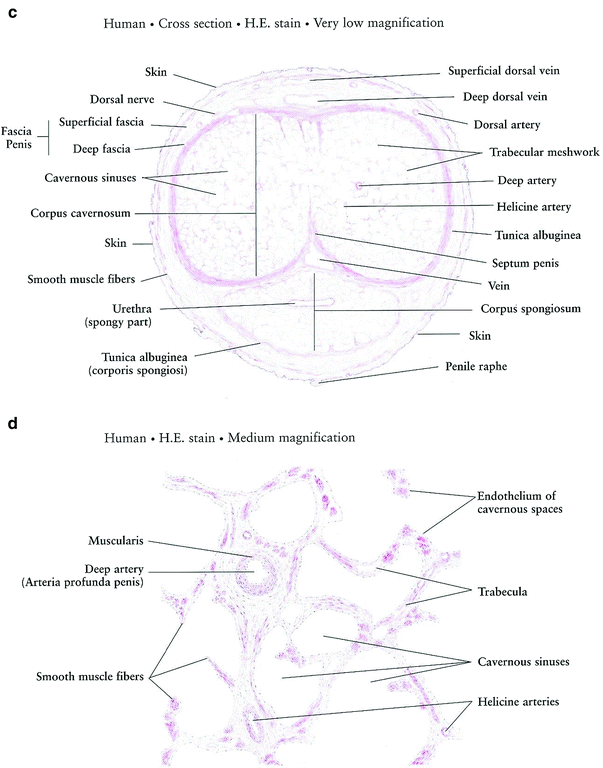


Fig. 3
Histology a Prostate gland. b Prostate gland: Acini c Penis d Penis corpus cavernosum (with permissions from Zhang 1999)
The glandular acini are highly redundant and are lined by two layers of cells: a luminal layer of tall columnar cells and a layer of cuboidal cells adherent to the underlying basement membrane. Between prostatic glands is fibromuscular stroma containing smooth muscle, collagen, and elastic fibers. In parenchymal portion, glands consist of a simple high cuboidal/low columnar epithelium.
2.2.2 Seminal Vesicles
Histologically, each seminal vesicle consists of a single coiled tubular structure that forms a large irregular lumen with many mucosal folds. The epithelium is composed of mixed columnar and pseudostratified columnar cells with mucosal crypts generated by infoldings of the mucosa. It is surrounded by two muscular layers that contract to create positive pressure to move secretions through the lumen and into the ejaculatory duct. The duct of each seminal vesicle combines with the ductus deferens to form the ejaculatory duct.
2.2.3 Urethra
Deep into the superficial urethral mucosa is the submucosa which contains smooth muscle, elastic tissue, and other stromal elements. The superficial histology of the urethra is dependent on its anatomic subsection. In the prostatic portion, it is lined with transitional epithelium. In the spongy portion, it contains stratified columnar epithelium until the navicular fossa, where a transition to non-keratinizing stratified squamous epithelium is seen. The lamina propria of the spongy portion of the urethra merges with the surrounding corpus spongiosum.
2.2.4 Penis
In the penis, cavernous bodies are irregular and lined with simple squamous endothelium and contain venous blood. The cavernous spaces of the cavernosa are larger than those of the spongiosum and have thinner stromal trabeculae, allowing the cavernosa to become more turgid than the corpus spongiosa when the penis is tumescent. (Fig. 3c, d).
2.2.5 Penile Bulb
The penile bulb consists of corpus cavernosum and responds during sexual arousal becoming distended, and is the base of the penis when erect.
3 Physiology and Biology
3.1 Physiology
The actively secreting prostate ductal areas contain pseudostratified columnar epithelium, while ducts with less secretory activity demonstrate simple columnar or high cuboidal epithelial cells. Within some ductal lumens are prostatic concretions, also called corpora amylacea, which are of uncertain physiologic significance. These concretions become more numerous with age and often calcify.
3.1.1 Prostate
The prostate gland is arranged in three concentric groups: 1. Main, one-half glandular; 2. Submucosal, one-fourth fibrous tissue; 3. Mucosal, one-fourth involuntary muscle (Fig. 4).
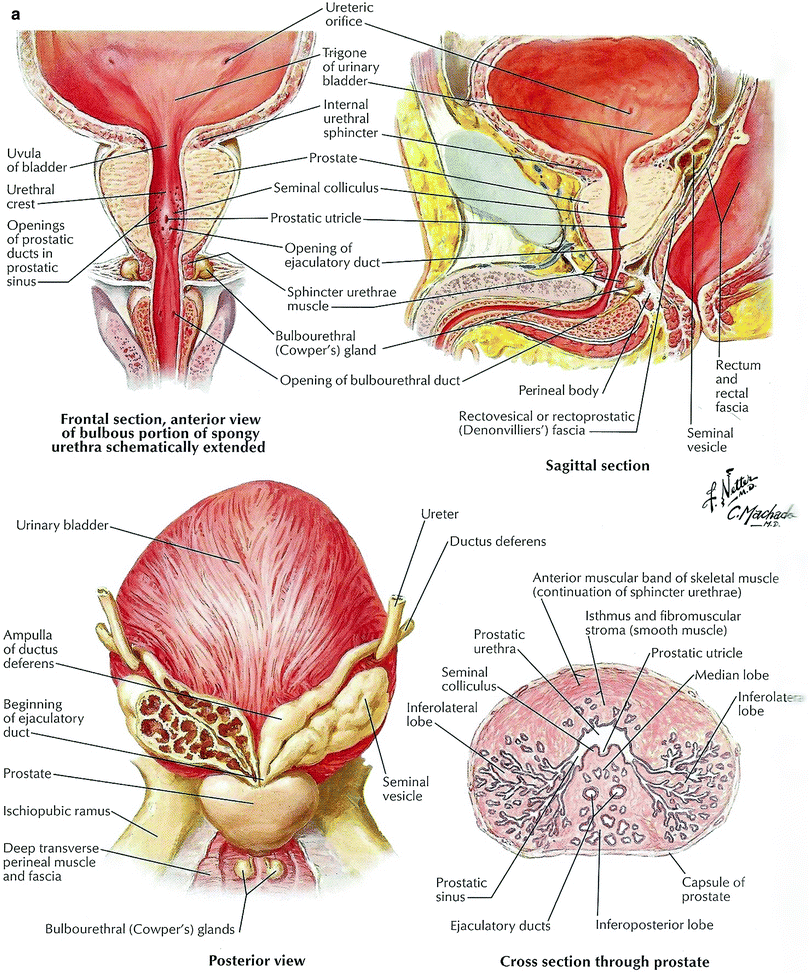
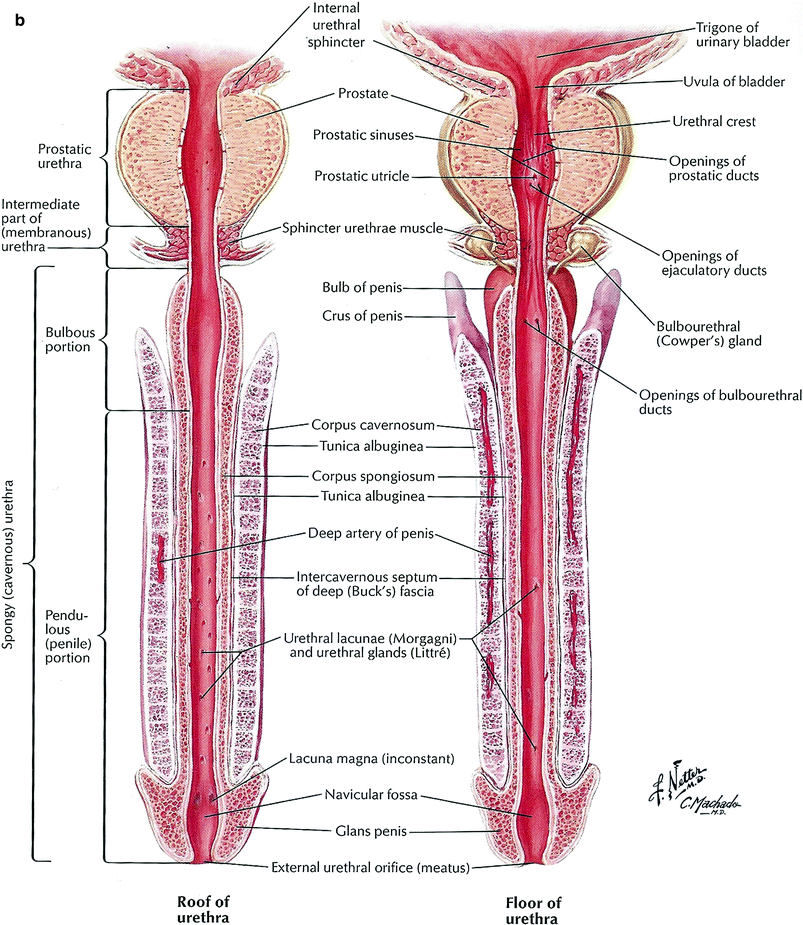
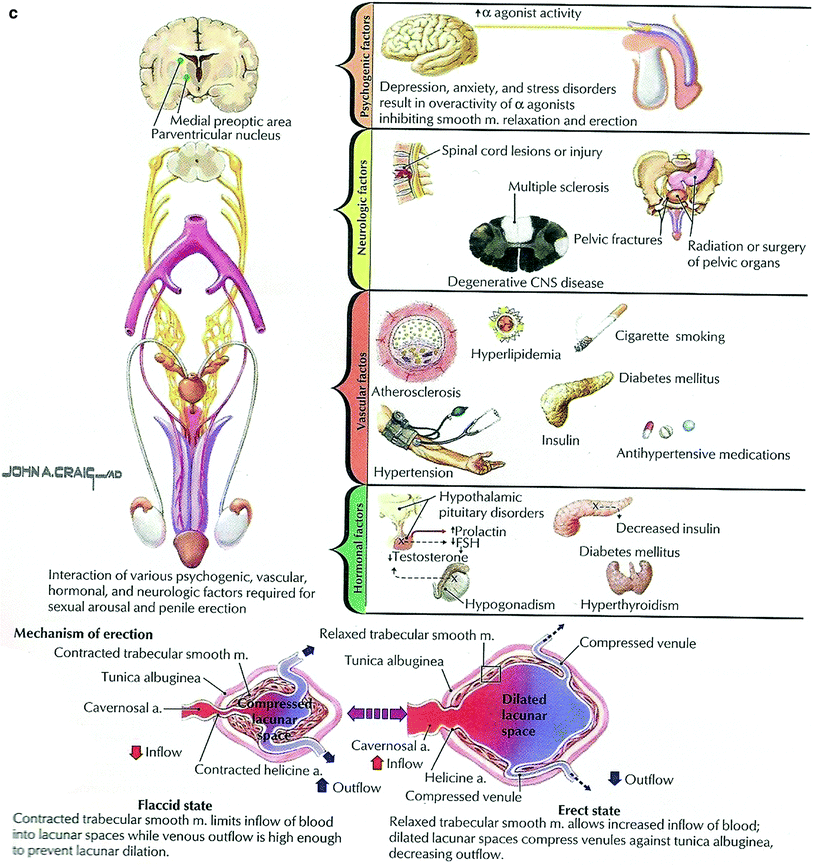



Fig. 4
Physiology of prostate (a) Urethra and penis (b) Penile Crura: sexual performance (c) (with permission from Netterimages.com)
The prostate gland is influenced by the male sex hormones, i.e., testosterone and adrenal androgens which are converted into dihydrotestosterone (DHT) which is 30 × more potent that testosterone. The DHT stimulates growth of the normal prostate glandular epithelium. The prostate gland secretes prostate acid phosphatase and prostate-specific antigen (PSA) which are incorporated into seminal fluid of which only a small fraction enters circulating blood (i.e. 4 Ng/ml). As the prostate enlarges and undergoes malignant transformation, PSA increases to >4 Ng/ml and serves as a biomarker for cancer increasing to 4.0–10.0 Ng/ml. In addition the prostate gland secretes prostatic acid phosphatase (PAP), an enzyme that regulates prostate cell growth and metabolism of the glandular epithelium. The fibronolysin in the secretions liquefies semen.
3.1.2 Seminal Vesicles
Seminal vesicle secretions consist largely of prostaglandins (accounts for the name of prostate gland) which during ejaculation discharges its secretions and assists in flushing out the semen through the urethra.
3.1.3 Urethra and Penis
During its erectile stage due to filling of its vascular spaces, allows for penetration of the vagina and its ejaculate of semen via the urethra, initiates the fertilization of the ovum (Fig. 4).
3.1.4 Penile Crura: Sexual Performance
Male sexual performance consists of specific sequence of events (Fig. 4):
Sexual arousal → libidinous desire → erection → ejaculation → orgasm → detumescence
-
Sexual arousal and libido are multifactorial and depend on the sex partner and level of testosterone. Arousal sensation via pudendal nerve S2–S4 somatic fibers).
-
Erection occurs with pelvis splanchnic (S2–S4 parasympathetic) innervations, penile vasodilatation of corpora cavernosum, and spongiosum-dependent patent inferior vesicle arteries and arterioles and mediate veno-occlusion, entrapping blood in penis to allow for vaginal penetration.
-
Ejaculation and orgasm via sympathetic via nerves.
-
Detumescence results with vasoconstriction of arterioles, diverting blood from corpora cavernosum and spongiosum into the periprostatic venous plexus via dorsal vein of the penis, via alpha-adrenergic receptor activation.
3.2 Biology
3.2.1 Prostate
The ducts of the prostate functional units empty into the prostatic urethra. The secretory elements (ducts) are surrounded by smooth muscle and connective tissue that separate the secretory units. Prostatic secretions are expelled into the urethra and join other components of the seminal fluid when these smooth muscle units contract. Prostatic secretions are alkaline (pH 6.5) and serous, containing acid phosphatase, carbohydrates, lipids, lipofuscin, hormones, citric acid, amylase, and fibrolysin, which act to liquefy the seminal fluid.
Late histologic changes in irradiated benign prostate ducts include variable ductal atrophic change, cytologic and nuclear atypia, basal cell hyperplasia, increased foreign body giant cell reaction to corpora amylacea, nuclear pleomorphism, nuclear vacuolation, hyperchromatic DNA, and presence of prominent nucleoli. Several metaplastic changes have been noted, including mucinous metaplasia, squamous metaplasia, and Paneth-like cell change. There is no significant difference in the histologic appearance of irradiated benign glands when the patients are treated with androgen deprivation therapy (Gaudin et al. 1999; Magi-Galluzzi et al. 2003). The surrounding stromal cells also undergo chronic inflammatory processes. The molecular and cellular mechanisms underlying this chronic inflammatory change are well described. Briefly, after the initial insult of radiation therapy, a cascade of inflammatory mediators is initiated including TNF-alpha, interleukin-1, and interleukin-6 (Haase 2004; Bentzen 2006). In the later phases of fibrosis, TGF-beta and PDGF, among others, simulate the proliferation of fibroblasts and the synthesis or extracellular matrix constituents and MMPs. Connective tissue growth factor (CTGF) further promotes fibrotic change (Vozenin-Brotons et al. 2003).
3.2.2 Seminal Vesicles
The seminal vesicles produce a viscous fluid containing high amount of fructose that nourishes the sperm. Other components include simple sugars, amino acids, ascorbic acids, and prostaglandins.
4 Pathophysiology
4.1 Prostate Gland
Several publications have documented the effects of various modalities of radiation therapy on prostatic tissues (Bostwick et al. 1982; Gaudin et al. 1999; Sheaff and Baithun 1997). In general, radiation changes are similar in patients receiving external beam radiation and brachytherapy, although the brachytherapy-associated changes may be more marked (Magi-Galluzzi et al. 2003).
Early histologic changes in the irradiated prostate, seen after several weeks, include nuclear contraction, signs of cytoplasmic injury, and small areas of early necrosis. These areas of injury and necrosis initiate the well described processes of acute inflammation, where polymorphonuclear cells, macrophages, and lymphocytes are recruited in a characteristic chronological pattern. As treatment progresses, a mixed acute-late inflammatory histology appearance predominates that gradually gives way to fibrotic change once radiation treatment has been completed.
Recruitment of cells typically involved in the chronic inflammatory process is also evident. Specifically, macrophages are seen in the early phase of fibrosis, which chemically recruit fibroblasts, which in turn transform to fibrocytes. Additionally, vascular changes are noted, including endothelial cell damage, intimal hyperplasia, marked arterial luminal narrowing, arterial medial thickening, cytoplasmic swelling, hyaloid changes of the capillary wall, and thinning of the capillary network (Sheaff and Baithun 1997; Herrmann 2006).
Prostate cancer cells respond differently than benign prostate cells after radiation therapy has been administered. This response, although characteristic, is quite variable, ranging from significant treatment-related changes to no apparent change after radiation therapy (Gaudin et al. 1999). Prostate cancer cells with no evident effect had an appearance similar to pre-treatment specimens. Prostate cancers with profound radiation changes demonstrated several characteristic morphologic changes including a decrease in the number of neoplastic glands, with residual glands in a more irregular morphology and some individual scattered cells not associated with glands. The cells demonstrated abundant cytoplasm with vacuolated and reticulated changes but little nuclear pleomorphism. In contrast to radiation changes in benign prostate tissues, radiation changes in prostate cancer cells were not associated with nuclear pleomorphism or prominent nucleoli. Furthermore, benign glands with radiation changes were extremely reactive to immunohistochemical stains for cytokeratin 34[beta]E12 and had a variable staining pattern to antibodies specific for prostate-specific antigen (PSA), while cancerous glands with radiation changes were not reactive to cytokeratin 34[beta]E12 were intensely immunoreactive for PSA. Additionally, while benign glands tended to maintain a lobular architecture, cancerous areas are arranged in a random, infiltrative morphology.
Part of the issue in determining the effects of irradiation of the prostate gland is the presence of a prostate cancer, which is the most common reason to treat the gland. Initially, following 60–70 Gy doses, necrosis of the cancer cells occur. Months to years later, the glandular epithelium are reduced in size and becomes atrophic and are replaced by fibrosis. The residual glands appear as “pale ghosts,” the columnar epithelium changes to cuboidal with pyrokinetic nuclear squamous metaplasia form irregular islands surrounded by dense fibrosis, foreign body giant cell reactions around corpora amylacea. Vascular changes are quite severe with obliteration of arterioles with internal forming cells.
Despite these characteristic changes, histopathologic interpretation of biopsies of a patient treated with radiation therapy is fraught with difficulty (Bostwick 1982). The risk of misinterpretation is that benign radiation changes will be mistaken for prostate cancer. In general, rebiopsy after radiation treatment (Fig. 5a–d) is reserved for the setting of a rising PSA after treatment when an isolated local recurrence is suspected and salvage brachytherapy or surgery is being considered. The presence of malignant cells in the biopsy specimen after radiation therapy should not be automatically interpreted as treatment failure. Prostate cancer cell death is a post mitotic event in a cancer with a long potential doubling time, meaning that regression of viable cancer is evident to at least 3 years after treatment (Crook et al. 2000). Furthermore, nearly 50 % of men with positive biopsies after brachytherapy and a short course of external beam treatment had viable cancer cells on planned repeat biopsy. Interestingly, only 27 % of these men experienced a biochemical failure (Goldstein 1998). Prestidge et al. reported serial post-treatment biopsies have demonstrated that a higher number of indeterminate biopsies after treatment eventually become negative after brachytherapy treatment (Prestidge et al. 1997). In general, if radiation biopsies show profound treatment effect in the adenocarcinoma, these patients are unlikely to fail therapy.
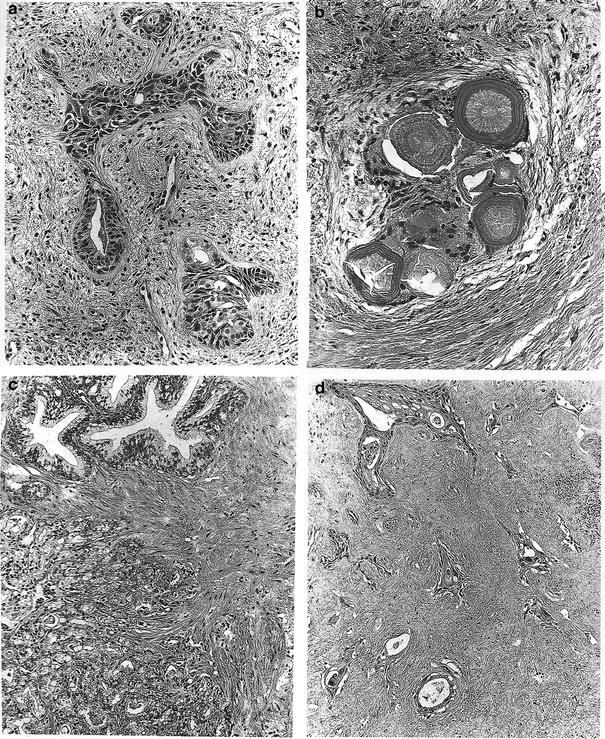

Fig. 5
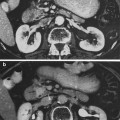
Post-RT histologic changes in the prostate gland. a Postradiation (~7000 cGy) squamous metaplasia in non-neoplasic prostatic glands with mild cytologic atypia. H&E, ×192. b Foreign body giant cell reaction around corpora amylacea 26 months after irradiation. No resudial epithelium can be recognized. H&E, ×192. c Well-differentiated adenocarcinoma in the lower field contrasting with normal glands in the upper field prior to radiation. Compare with (d), obtained 30 months after irradiation with ~7000 cGy (same magnification): there is no residual carcinoma, and the field displays extensive stromal fibrosis. The remaining, nonneoplastic glands show atrophy and extensive squamous metaplasa. H&E, ×192 (with permission from Fajardo 2001)
There are also histological changes from the use of androgen deprivation therapy alone. Androgen stimulation is an important component of normal prostate metabolism. 90–95 % of circulating testosterone is made by the testes, with the remainder produced by the adrenal glands. In the prostate gland, testosterone is converted into dihydrotestosterone by alpha-5-reductase. Dihydrotestosterone stimulates growth of both normal prostate tissue and prostate adenocarcinoma cells. When androgen deprivation is administered, consistent effects can be seen regardless if combined androgen blockade (LHRH agonist and peripheral androgen receptor blocker) or anti-androgen monotherapy (LHRH agonist alone) is used. Degenerative phenotypes are noted, including nuclear pyknosis, and vacuolization of the cytoplasm (Tetu 1991; Armas 1994). Furthermore, androgen deprivation also suppresses the histological changes commonly used to diagnose adenocarcinoma, such as increased nuclear size, nuclear pleomorphism, and prominent nucleoli. Therefore, care must be taken in the histological evaluation of patients who have received androgen deprivation therapy prior to prostate biopsy because there is a risk of underestimating both tumor extent and Gleason score. Rigorous examination of the specimen for scant individual malignant cells and special immunohistochemical stains are essential in this clinical situation (Vernon 1983).
In addition to the above commonly used strategies of androgen blockade, other agents, such as estrogens and 5-alpha reductase inhibitors also cause histological changes in normal and malignant prostate cells. The effect of estrogen administration on prostate histology is mainly of historical interest, as estrogens are not commonly used in contemporary treatment algorithms. However, estrogens induce the above changes seen with modern anti-androgen regimens and further cause a unique effect of squamous metaplasia in benign and malignant prostate cells (Schenken 1942; Franks 1960). Finasteride and dutasteride block the conversion of testosterone to dihydrotestosterone in the prostate gland by inhibiting 5-alpha reductase and are used in a variety of clinical situations including BPH and androgenetic alopecia. Their use has been found to have minimal influence on prostate cancer cells and does not typically interfere with pathologic diagnosis or the prognosis of Gleason grade (Yang et al. 1999; Carver et al. 2005; Iczkowski et al. 2005). These agents do reduce PSA values by approximately 50 % (Etzioniet al. 2005) and prostate size by about 25 % (Thompson et al. 2003). Furthermore, 5-alpha reductase inhibitors do appear to have the ability to affect the incidence and grade of prostate cancers in men who use them (Andrioleet al. 2005). The most compelling argument for this comes from the Prostate Cancer Prevention Trial, which demonstrated that healthy men treated with 5-alpha-reductase inhibitors had a 24.8 % reduction in the risk of developing prostate cancer. In this cohort, certain risk factors while on finasteride were predictive of Gleason score ≥7 disease, including higher absolute PSA values, increasing PSA values, an abnormal digital rectal examination, and older age (Thompson et al. 2003, 2007).
4.2 Seminal Vesicles
Radiation effects on the seminal vesicles are more obvious than in the prostate gland. The normal complex arborizing glands are reduced to narrow cavities with a few branches embedded in dense collagen scoring.
Radiation also causes changes in the urothelium (Antonakopoulos et al. 1982, 1984; Stewart 1986). Irradiated urothelial cells demonstrate changes including nuclear pleomorphism, swollen cytoplasm, and altered labeling indices as compared to non-irradiated urothelial cells. Loss of tight junctions is noted, allowing hypertonic urine access to the interstitial area, leading to chemical fibrotic injury and increasing the probability of bacterial infection and subsequent inflammatory damage. These morphological changes correlate clinically with the onset of irritative urinary symptoms encountered after a course of radiation therapy (Marks et al. 1995). When bulbomembranous urethral strictures are examined histologically, there is an initial ulceration of the urothelium that develops into proliferative changes of stratified squamous epithelium with interposed elongated myofibroblasts and multinucleated giant cells that produce abundant collagen (Baskin et al. 1993). The myofibroblasts are thought to be a primary causative factor for stricture formation. The ubiquitous stromal changes of radiation therapy are also noted, including obliterative endarteritis, ischemia, and fibrosis.
4.3 Penis and Urethra
Penis and urethra are incidentally irradiated in treating prostate gland cancers. In laboratory rats after single fraction doses of 1,000 and 2,000 cGy, after 5 months, the animals had impaired responses to central and peripheral stimulation, at both doses, increased with the higher dose; the number of nerve fibers positive for nitric oxide synthase.
In each penile segment decreased significantly by approximately 25 % compared to control. The mechanism for radiation-induced erectile dysfunction was attributed to defective vascularity of penile tissues as well as peroneal nerves and smooth muscle.
5 Clinical Syndromes (Endpoints)
Patients undergoing radiation to the distal male GU tract are at risk for developing both acute and late genitourinary, gastrointestinal, and sexual toxicities. Acute toxicities are attributable to effects from acute inflammation, while late toxicities are usually attributable to radiation-induced fibrosis, vascular damage, and altered patterns of vasculature. Late effects of radiation are particularly multifactorial, being affected by comorbidities, genetic factors, and other cancer treatments in addition to radiation-related variables such as total dose, dose per fraction, fractionation schedule, and dose–volume parameters. With modern teletherapy techniques such as intensity modulated radiation therapy (IMRT) and image-guided radiation therapy (IGRT), in addition to improvements in brachytherapy, the overall morbidity of external beam radiation therapy has been significantly reduced despite higher contemporary prescription doses. The addition of systemic agents, such as cytotoxic chemotherapy or androgen deprivation therapy, may alter the risk of these toxicities (Zelefsky et al. 2000; Valicenti et al. 2003; Liu et al. 2004; Feigenberg et al. 2005; Zapatero et al. 2005; Lawton et al. 2008).
5.1 Sexual Dysfunction after Radiation Therapy
The main sexual side effects of radiation therapy to the pelvis are impotence, decreased libido, decreased ejaculate, and painful ejaculation. Recent reports demonstrate the rate of radiation-related erectile dysfunction is of the order of 35–55 % (Cahlon et al. 2008; Mantz et al. 1997; Potosky et al. 2000; Hamilton et al. 2001). The process of tumescence is a complex process, depending on afferent cavernous nerves supplying the penis with nitric oxide. Relaxation of the afferent internal pudendal and accessory pudendal arterioles and cavernosal smooth muscle occurs, which allows for filling of the trabecular space. Increased venous resistance prevents outflow, causing trapping of blood, and contraction of the bulbocavernosus and bulbospongiosus muscles further increase intratrabecular space pressure. In general, radiation-induced impotence is thought to manifest within the first 2 years after treatment (van der Wielen 2007). The decline in ability to obtain and maintain erections after therapy has historically been thought to be caused by radiation exposure of tissues involved in the process of tumescence, including the afferent neurovascular bundles, the penile bulb, and the corpora cavernosa (Fisch et al. 2001; Roach et al. 2004; Wernicke 2004). However, a recent publication has demonstrated that radiation-induced erectile dysfunction is mainly due to afferent arterial insufficiency, with only a small percentage of cases due to changes in veno-occlusive capacity (Zelefsky and Eid 1998). Additionally, several publications suggest that doses to structures such as the penile bulb are not predictive of post-therapy impotence (van der Wielen et al. 2007; Hoogeman et al. 2008; Solan 2009). Regardless, modern radiation techniques are able to minimize radiation dose to these tissues that are at risk, including the penile bulb and base of the penis, possibly decreasing the rates of radiation-related erectile dysfunction (Sethi et al. 2003; Buyyounouski et al. 2004). However, sparing the neurovascular bundle with external beam techniques is not yet achievable, given its close proximity to the prostate, need for the appropriate PTV margins to account for interfraction and intrafraction motion, and continuing trends of dose escalation.
5.2 Erectile Dysfunction
The evaluation of radiation-related erectile dysfunction is complex, depending on the method of assessment, toxicity scale used, time since radiation, and radiation technique used for treatment (Rosen et al. 1997; Litwin 1998; Talcott et al. 1998). Additionally, a multitude of other factors can exacerbate post-treatment erectile dysfunction, including existing peripheral vascular disease, diabetes mellitus, smoking history, hypertension, hypercholesterolemia, administration of androgen deprivation therapy, and other medications (Hollenbeck et al. 2004; Goldstein et al. 1984). Androgen deprivation contributes to erectile dysfunction because physiologic amounts of testosterone contribute to sexual desire (O’Carroll 1984), homeostasis of the erectile apparatus and nerves (Saad et al. 2007), and cavernous vasodilation (Aversa et al. 2000).
The social situation of the patient also has a significant role on sexual function, including the presence or absence of a willing partner and the physiological and emotional impact of cancer diagnosis and treatment on the patient (Goldstein et al. 1984; Fiorino et al. 2009). Furthermore, independent of a diagnosis of cancer, loss of erectile function is common in men between 40- and 69-years old, with up to 26 out of every 1,000 men developing ED each year (Johannes et al. 2000). It is therefore difficult, if not impossible, to define the radiation parameters clearly causing sexual dysfunction and the time frame in which they manifest after treatment.
It is recommended by Quantec authors that patients undergo pre- and post-RT assessment of ED using the IIEF. Patients can be grouped into five groups according to their scores; for example, in none (D’Amico et al. 2004; Chen et al. 2001; van der Wielen et al. 2007; Goldstein et al. 1984), mild (Macdonald et al. 2005; Merrick et al. 2002; Zelefsky et al. 1999; Weber et al. 1999; Rosen et al. 1999), mild to moderate (Wallner et al. 2002; Zelefsky et al. 2006; Wernicke et al. 2004; van der Wielen et al. 2008; Skala et al. 2007), moderate (Selek et al. 2004; Roach et al. 2004; Pinkawa et al. 2009a, b; Mangar et al. 2006), and severe (Fisch et al. 2001; Cahlon et al. 2008; Brown et al. 2007). It is important that the evaluation of ED is performed with a detailed history including sexual, medical, and psychosocial status and other laboratory tests (Rosen et al. 1997, 1999; Kratzik et al. 2005; Rosenberg 2007). Further clinical studies may be needed to validate the IIEF for the assessment of ED after RT.
5.3 Acute and Late Effects from Penile Radiation
The main acute toxicity of irradiation of the penis is the skin reaction (see “Thyroid”). However, some relevant radiation-specific literature is available on this topic and merits discussion (Crook et al. 2009, 2010). After interstitial penile brachytherapy, moist desquamation appears to be the only significant acute toxicity, peaking 2–3 weeks after treatment and taking several months to heal. Patients are also at risk for acute post-treatment adhesions which usually present with a split or deviated urine flow from the meatus. Common late complications include penile soft tissue necrosis and urethral stenosis, which occurs in about 10 % of men treated with interstitial penile brachytherapy for squamous cell carcinoma (Crook et al. 2009). Soft tissue necrosis usually appears as an area of progressive ulceration that typically takes place 6–18 months after brachytherapy (Delannes et al. 1992). Ulcerations leading to necrosis can be exacerbated by thermal injury or traumatic episodes to the penis, including biopsy. Radiation-induced penile urethral strictures occur between 1 and 3 years after treatment and can present with altered urodynamics, divergent stream, pain, or hematuria. The skin can also undergo chronic atrophic change after treatment, including thinning of the dermis and epidermis, formation of irregular pigmentation patterns with gain or loss of natural pigment, increased skin sensitivity or pain, and formation of teleangectatic vessels.
5.4 Radiation-Induced Urinary Incontinence
Following RT for prostate cancer, the rate of RT-induced urinary incontinence is of the order of 1–4 % in men who have no history of invasive prostatic procedures (Talcott et al. 1998; Potosky et al. 2000; Hamilton et al. 2001). This rate increases when the patient undergoes surgical procedures to the prostate gland before or after radiation treatment. Furthermore, a history of intercurrent illness, especially diabetes mellitus, increases the risk of late GU toxicity. Investigators at Fox Chase Cancer Center found that diabetics treated with three-dimensional conformal radiation therapy for prostate cancer had increased rates of late Grade 2 GU toxicity (Herold et al. 1999). Additionally, they reported that diabetics had an early onset of late toxicity (median 10 months versus 20 months for non-diabetics).
In limited circumstances, radiation therapy can cause chronic improvement in urinary function. Specifically, reports of improved urinary function and quality of life have been noted in patients with significant pre-treatment irritative or obstructive symptoms (Chen et al. 2009). The proposed mechanism for these improvements is a radiation-induced reduction in prostate volume (Coia et al. 1995).
5.5 Fecal Incontinence
Although rare, radiation-induced fecal incontinence profoundly effects quality of life. Two large, modern studies have demonstrated that the chance of using pads of fecal incontinence at any time after treatment is approximately 10 % and the probability of needing regular use of pads after 2 years of treatment is approximately 3 % (Peeters et al. 2006c; Fiorino et al. 2008). Profound incontinence is even rarer, with the chance of needing multiple pads per week years after treatment is less than 1 %. Fecal incontinence is thought to be multifactorial. Contributing factors probably include decreased absorptive capacity of irradiated rectal mucosa, chronic inflammatory changes leading to urge incontinence, neurovascular damage to the afferent nerves controlling the anal sphincter, and direct muscle damage to the sphincter itself. Prior abdominal or anorectal surgery or trauma increases the probability of fecal incontinence after treatment (Peeters et al. 2006c; Fiorino et al. 2008).
When the endpoint of fecal incontinence is considered, there is also a strong correlation of clinical endpoint with dose–volume parameters (Peeters et al. 2006c; Fiorino et al. 2008). Unlike late rectal bleeding, where the high dose regions are most predictive, it appears that a large volume of rectum receiving an intermediate dose is most predictive of late fecal incontinence. Fiorino prospectively studied a cohort of over 500 patients and found that there was less than 1.5 % probability of late fecal incontinence requiring pads when V40 < 65 %. Peeters essentially confirmed this finding. Furthermore, the location of radiation exposure also predicts for late fecal incontinence. Several authors have reported that inclusion of large portions of the distal rectum, including the anorectum, predisposes that patient to late rectal bleeding (Vordermark et al. 2003; al-Abany et al. 2004; Heemsbergen et al. 2005). Modern IMRT and 3D-CRT techniques have obviated the need to include large portions of the anorectum in the treatment field and should yield decreases in incontinence rates. See “Radiation Induced Rectal Toxicity” for further discussion of this issue.
5.6 Diagnosis
5.6.1 Atrophy
Irradiation of the prostate gland leads to interstitial fibrosis. Atrophy and calcifications can be seen on CT and MR images (Fig. 6a, b). Radiation changes are also seen in the seminal vesicles, which lose volume and demonstrate low T2-weighted signal intensity on MR images after radiation. The prostatic and membranous urethras are very sensitive to radiation. The irradiated urethral tissue often develops strictures.
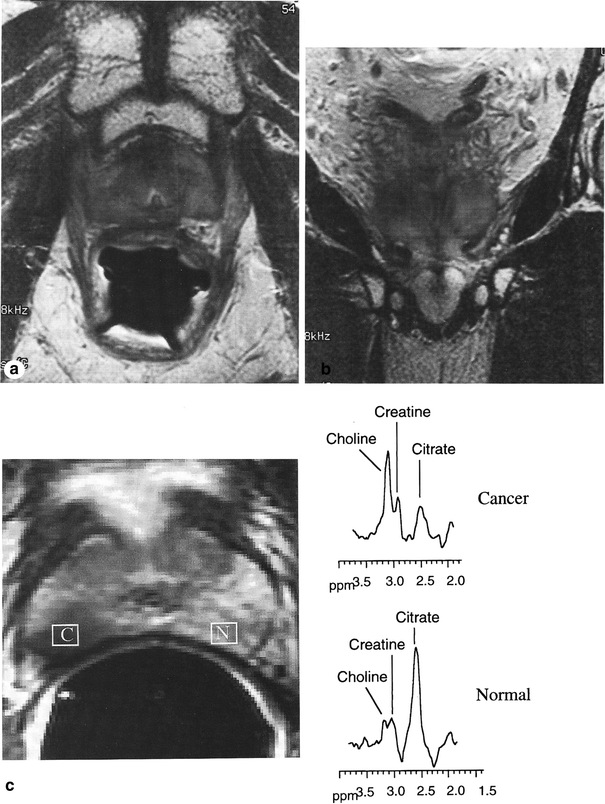

Fig. 6
Imaging changes in the prostate gland. a, b T2-weighted MR images of the prostate. The zonal anatomy is indistinct (compared to a normal untreated gland), and the peripheral zone shows low signal intensity. c Images from a patient with stage T2 adenocarcinoma of the prostate: fast spin-echo T2-weighted axial endorectal magnetic resonance (MR) spectroscopic image through the midgland. Prostate carcinoma appears as an area of abnormal low signal intensity (C) in the normally high signal intensity peripheral zone (N). MR spectra from the area of carcinoma (C) in the right peripheral zone demonstrates abnormal elevation of the choline peak and an abnormally low citrate peak. In comparison, MR spectra from an area of normal peripheral zone (N) demonstrates a normal high citrate peak and a normal low choline peak. Tumor in the right peripheral zone demonstrates contact with a smooth and apparently intact prostatic capsule. Postradiation changes in the prostrate. This patient underwent radiation therapy for prostate cancer. The axial (a) and the coronal (b) T2-weighted fast spin echo magnetic resonance images show a small, feature gland with a dark peripheral zone indicative of radiation changes in the prostate (with permission from Bragg et al. 2002)
5.6.2 Persistence of Cancer
As aforementioned in the pathophysiology section, a vexing issue is persistence of cancer cells, the same is true for follow-up assessment by serial MRIs in follow-up. Comparing pre- to post-RT MRIs might be helpful in this regard, as is MR spectroscopy. Figure 6c illustrates an example of the utility of MR spectroscopy in defining tumor in the prostate.
6 Dose, Time, Fractionation: Radiation Tolerance, Predicting RT-Induced Injury and Recommended Dose–Volume Constraints
Since Burman et al. (1991) reported on the modeling of dose–volume effects based on clinical outcomes from the 2D era, the advent of increasingly advanced radiation technologies has led to careful study of the effect of dose–volume relationships on normal tissues during radiation treatment. A great deal of literature is available examining toxicities from and recommended treatment parameters for radiotherapeutic treatment of pelvic malignancies, including those of the distal male GU tract.
6.1 Rectal Constraints
6.1.1 General Rectal Constraints
Although the rectum is covered in a separate chapter, a discussion of this organ is merited here, as the appropriate dose–volume rectal constraints for prostate cancer radiotherapy is the most published area of normal tissue tolerance in this malignancy. The reason for this interest in rectal tolerance is clear. Unlike bladder toxicity, late rectal toxicity has been found to be directly and consistently correlated with physical treatment parameters such as dose and volume. Rectal complications are the main late toxicities that limit dose escalation in prostate cancer. General trends can be drawn from the available clinical literature regarding these constraints for conventionally fractionated radiation therapy despite institutional differences in prescribed dose, extent of contouring, radiation techniques, and outcomes measurement. Moderate to severe late rectal effects are dose dependent, occurring in around 25 % of men treated with 78 Gy and 13 % in men treated with 70 Gy (Pollack et al. 2002; Peeters et al. 2006a, b; Zietman et al. 2005). Despite this dose dependency, several reports indicate that careful planning techniques and intensity modulation can mitigate this toxicity and allow dose escalation without excess late GI toxicity (Zelefsky et al. 2002; Beckendorf et al. 2004; Peeters et al. 2006a, b).
Numerous studies have been published with late rectal bleeding as an isolated clinical endpoint with consistent contouring of the rectum. Jackson et al. from MSKCC reported on 171 patients treated with 3D conformal radiation therapy to a dose of 70.2 or 75.6 Gy. They found a significant dose–volume correlation with RTOG > 2 late bleeding and recommended, for the techniques and doses used, V40 < 60 % and, for patients treated with 75.6 Gy, a V77 < 14 % (Jackson et al. 2001). An Italian intergroup published two papers using RTOG > 2 late bleeding endpoints in patients treated in the range of 70–78 Gy and ultimately suggested the following dose–volume constraints: V50 < 60 %, V60 < 45 %, and V70 < 25 % (Fiorino et al. 2002, 2003). The recommendation of V70 < 25 %, using the same endpoint, was independently confirmed by the group at William Beaumont Hospital using adaptive image-guided radiation with prescription doses between 70.2 and 79.2 Gy (Vargas et al. 2005). Peeters et al. found that a V65 > 30 % was highly predictive for late bleeding requiring transfusions or interventional laser coagulation in patients treated between 68 and 78 Gy (Peeters et al. 2006c). When SOMA-LENT Grade > 2 late bleeding criteria was used as the clinical endpoint, recommendations are similar: V50 < 55 %, V60 < 40 %, V70 < 25 %, and V75 < 5 % (Fiorino et al. 2008; Fellin et al. 2009). In addition to these straightforward dose–volume constraints, several groups have generated normal tissue complication probability models and nomograms for late rectal bleeding that are generally consistent with the above studies (Rancati et al. 2004; Söhn et al. 2007; Valdagni Rancati et al. 2008).
Dose–volume constraint recommendations are only slightly altered when all forms of late rectal toxicity are included in outcomes analysis. The MD Anderson group reported on rectal complications from their randomized dose escalation trial and found that the rates of rectal complications were similar in both arms (70 and 78 Gy) but found a significant increase in late rectal complications when V70 > 25 % (Storey et al. 2000a, b; Kuban et al. 2008). When a different subset of patients treated in the same general dose range was evaluated from the same institution, the investigators found that the percentage of the rectum exposed to a certain dose, rather than an absolute volume, was predictive of late rectal toxicities. In this paper, the following recommendations were made: V60 < 40 %, V70 < 25 %, V75.6 < 15 %, and V78 < 5 % (Huang et al. 2002). Analysis of the high dose arm (74 Gy) of RTOG 94-06 yielded a recommendation to keep the V65 < 50 % to keep late rectal toxicity Grade I or less (Michalski et al. 2004). A recent report from MSKCC analyzing 478 patients treated to 86.4 Gy found a 4 % rate of CTCAE 3.0 late rectal toxicity with V47 < 53 % and V75.6 < 30 % at a median follow-up of 53 months (Cahlon et al. 2008). Numerous studies have found results similar to the results seen in these trials (Fonteyne et al. 2007; Karlsdóttir et al. 2008).
Taking these individual publications into account, there is general agreement that multiple dose–volume constraints in the intermediate (30–50 Gy) and high range (>70 Gy) regions of a histogram are prudent to shape the dose–volume histogram appropriately and to minimize the probability of late rectal bleeding to rates <10 %. It is also clear that while the volume of the rectum receiving a high dose is more predictive of late rectal toxicity, the amount of rectum receiving lower doses also predicts rectal toxicity, particularly with non-IMRT techniques. The distribution of dose on the rectum is also important, as increasing doses to the posterior rectal wall and upper rectum are associated with increase rates of late rectal toxicity (Skwarchuk et al. 2000; Fiorino et al. 2002; Heemsbergen et al. 2005; Peeters et al. 2006d; Munbodh 2008). A summary of recommended dose/volume constraints for late rectal injury is provided in Table 1. See “Radiation Induced Rectal Toxicity” for additional discussion.
Table 1
Recommended dose–volume constraints from large studies intended to minimize the risks of LATE rectal toxicity for patients with treated with modern radiation techniques and doses for prostate cancer
|
References
|
Prescription Doses (Gy)
|
Suggested constraints
|
Comments
|
|---|---|---|---|
|
Jackson et al (2001)
|
70.2 or 75.6
|
V40 Gy < 60 %
V77 Gy < 14 % (for patients treated at 75.6 Gy)
|
Bleeding endpoint
RTOG Grade ≥ 2
Rectum solid structure contoured with the caudal limit = first CT slice above the anal verge, cranial limit = first CT slice below the sigmoid flexure
|
|
Fiorino et al. (2002)
|
70–76
|
V50 Gy < 60 %
V60 Gy < 50 %
|
Bleeding endpoint
Modified RTOG Grade ≥2
Rectum solid structure contoured with the caudal limit = first CT slice above the anal verge, cranial limit = first CT slice below the sigmoid flexure
Included non-conformal patients; excluded pts with rectal volume >100 cc
|
|
Fiorino et al. (2003)
|
70–78
|
V50 Gy < 60 %
V60 Gy < 45 %
V70 Gy < 25 %
|
Bleeding endpoint
Modified RTOG Grade ≥ 2
Rectum solid structure contoured with the caudal limit = first CT slice above the anal verge, cranial limit = first CT slice below the sigmoid flexure
V70 more predictive for Grade 3 bleeding
|
|
Vargas et al. (2005)
|
70.2–79.2
|
V70 Gy < 25 %
|
Bleeding endpoint
CTC 2.0 Grade ≥ 2
Rectum solid structure contoured with the caudal limit = first CT slice above the anal verge, cranial limit = first CT slice below the sigmoid flexure
Chronic rectal toxicity, mostly bleeding V50 also correlated (no specific constraints suggested)
|
|
Peeters et al. (2006c)
|
68 or 78
|
V65 Gy < 30 %
|
Bleeding endpoint
Bleeding requiring lasers/transfusions
Rectum solid structure contoured with the caudal limit = first CT slice above the anal verge, cranial limit = first CT slice below the
Sigmoid flexure
V55-V65 correlated with V65 most predictive; independent impact of abdominal/pelvic surgery
|
|
Fiorino et al. (2008)
|
70–78
|
V50 Gy < 55 %
V60 Gy < 40 %
V70 Gy < 25 %
V75 Gy < 5 %
|
Bleeding endpoint
SOMA/LENT Grade ≥ 2
Rectum solid structure contoured
With the caudal limit = first CT slice above the anal verge, cranial limit = first CT slice below the sigmoid flexure
Prospectively scored patients; previous abdominal/pelvic surgery independently predictor of bleeding (suggested V70 < 15 %); V75 best predictor of Grade 3 bleeding
|
|
Fellin et al. (2009)
|
70–80
|
V75 Gy < 5 %
|
Bleeding endpoint
SOMA/LENT Grade ≥ 2
Rectum solid structure contoured with the caudal limit = first CT slice above the anal verge, cranial limit = first CT slice below the sigmoid flexure
Prospectively scored patients; previous abdominal/pelvic surgery independently predictor of bleeding (suggested V70 < 15 %) and best predictor of Grade 3 bleeding
|
|
70 or 78
|
V70 Gy < 25 %
|
General toxicity endpoint
GI RTOG Grade ≥ 2
Rectum 11 cm long starting 2 cm below ischial tuberosities
|
|
|
Huang et al. (2002)
|
70 or 78
|
V60 Gy < 40 %
V70 Gy < 25 %
V75.6 Gy < 15 %
V78 Gy < 5 %
|
General toxicity endpoint
GI RTOG Grade ≥ 2
Rectum 11 cm long starting 2 cm below ischial tuberosities
|
|
Michalski et al. (2004)
|
74
|
V65 Gy < 50 %
|
General toxicity endpoint
GI RTOG Grade ≥ 2
|
|
Fonteyne et al. (2007)
|
74–80
|
V40 Gy < 84 %
V50 Gy < 68 %
V60 Gy < 59 %
V65 Gy < 48 %
|
General toxicity endpoint
GI RTOG Grade ≥ 2
All patients were treated with
IMRT technique
|
|
Karlsdóttir et al. (2008)
|
70
|
V40 Gy < 70 %
|
General toxicity endpoint
GI RTOG Grade ≥ 2
A number of cut-offs predictive of toxicity; V40–V43 most predictive
|
|
Kuban et al. (2008)
|
70 or 78
|
V70 Gy < 25 %
|
General toxicity endpoint
GI RTOG Grade ≥ 2
Rectum 11 cm long starting 2 cm below ischial tuberosities
|
6.1.2 Stool Frequency
Stool frequency has also been correlated with treatment-related dose–volume parameters. Fonteyne et al. reported that the rate of mild diarrhea was correlated with the volume of the rectum receiving 40 Gy, and chronic rectal urgency was correlated with the volume of the rectum receiving 70 Gy (Fonteyne et al. 2007). Similarly, Peeters reported that both the V40 and the mean rectal dose were predictive of stool frequency (Peeters et al. 2006c). See “Biophysiopathology of the Microvasculature and Microcirculation” for additional discussion.
6.1.3 Brachytherapy
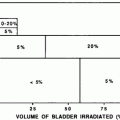
The prostate brachytherapy literature has consistently demonstrated that very small volumes of the rectum can tolerate very high doses of radiation, which is an important consideration in this era of highly conformal dose escalation. Snyder et al. found that when the rectum was contoured as a hollow structure from 9 mm below the prostate apex to 9 mm above the top of the seminal vesicles, the rate of grade 2 or greater proctitis was dependent on the volume of rectum receiving the prescription dose of 160 Gy. The rates of proctitis per absolute volume of rectal wall receiving prescription dose were: 0 % if < 0.8 cc, 7–8 % between 0.8 and 1.8 cc, and 25 %, and 24–25 % if > than 1.8 cc (Snyder et al. 2001). D’Amico’s group also found that the absolute volume of rectum receiving 100 Gy was predictive of needing argon plasma coagulation for late proctitis after brachytherapy, with no patients needing intervention when the volume of rectum receiving 100 Gy was below 8 cc, whereas 20 % of men needed intervention with argon plasma coagulation when the volume of rectum receiving 100 Gy was greater that 8 cc (Albert et al. 2008). Han et al. found a similar outcome when endoscopically proven radiation proctitis was used as the endpoint and the rectum was contoured as a solid structure, with the volume of the rectum receiving at least 100 % of the prescription dose being 2.5 cc in patients with endoscopically-confirmed proctitis and 0.6 cc in those with no evidence of bleeding (Han and Wallner 2001). The maximum point dose to the rectum after brachytherapy is also predictive of RTOG > 2 bleeding, with a 0.4 % toxicity rate for a maximum point dose of 150 Gy, a 1.2 % rate for a maximum point dose of 200 Gy, and a 4.7 % rate for a maximum point dose of 300 Gy (Waterman and Dicker 2003). Therefore, it is clear that small volumes of the rectum can receive very high doses, leading investigators to embrace more precise brachytherapy strategies, and by extrapolation, external beam treatments in combination with increasingly effective real-time image guidance solutions in an effort to dose escalate the prostate safely while sparing more of the rectum from the very high dose regions. It is evident that the smaller the amount of rectum irradiated, the less likely that late rectal bleeding will occur.
6.1.4 Acute GI Toxicities
Dose volume constraints and their effect on acute GI toxicities from pelvic radiation has been studied less. Nonetheless, these appear also to be highly dependent on dose–volume parameters, typically with mean rectal dose and the volume of rectum receiving greater than 60 Gy. Peeters examined GI effects in the first 6 weeks of treatment and found that the mean rectal dose as well absolute and the relative amount of rectum receiving 5, 15, and 30 Gy were predictive of acute GI toxicity (Peeters et al. 2005a). Vavassori and Cheng also reported DVH correlations with rates of acute toxicity, including the mean dose, and the minimal dose to 10, 20, and 50 % of the rectum (Vavassori et al. 2007; Cheng et al. 2008). Other authors have offered more specific recommendations. Nuyttens’ group found that a V75 < 11 cc and a mean dose < 38 Gy minimized acute rectal toxicity (Nuyttens et al. 2002). Another report looking at patients treated with 78 Gy found that a V65 Gy < 20 % was protective against any grade I or higher acute GI toxicity (Karlsdttir et al. 2004). Of note, most of the above studies looking at acute side effects contoured the rectum as a solid structure, including the luminal contents. A summary of recommended dose/volume constraints for acute rectal effects is provided in Table 2.
Table 2
Recommended Dose–Volume Constraints From Large Studies Intended to Minimize the Risks of ACUTE Rectal Toxicity for Patients with Treated with modern radiation techniques and doses for prostate cancer
|
References
|
Prescription Dose range
|
Suggested constraints
|
Comments
|
|---|---|---|---|
|
Nuyttens et al. (2002)
|
72–80 Gy 2 Gy/fr
|
V75 Gy < 11 cc
Mean dose < 38 Gy
|
≥G2 modified RTOG toxicity; acute toxicity during treatment retrospectively assessed; solid rectum including filling
|
|
Karlsdóttir et al. (2008)
|
70 Gy 2 Gy/fr
|
V40 Gy, V70 Gy
|
≥G2 modified RTOG toxicity; acute toxicity during treatment; solid rectum including filling
|
|
Peeters et al. (2005b)
|
68–78 Gy 2 Gy/fr
|
Mean dose V30 Gy, V35 Gy, V60 Gy, V65 Gy absolute V50 Gy, V60 Gy, V65 Gy % rectum length > 5 Gy, > 30 Gy absolute rectum length > 5 Gy, > 15 Gy, > 30 Gy
|
≥G2 modified RTOG toxicity; acute toxicity during treatment prospectively assessed; rectal wall; DVH of the first 6 weeks of treatment
|
|
Michalski et al. (2004)
|
78 Gy 2 Gy/fr
|
V65 Gy < 20 %
|
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree