Clinical Presentation
The patient is a 70-year-old man who had open surgical repair of an abdominal aortic aneurysm with bilateral aortoiliac bypass graft, bypass of the right renal, and surgical exploration of the superior mesenteric artery. The patient had a hypotensive episode during the surgical procedure. He awoke from anesthesia with flaccid paralysis of both lower extremities and diminished sensation to pinprick at the T11 level. He reported abdominal pain but no back pain or lower extremity pain. A magnetic resonance (MR) image was obtained to rule out either epidural hematoma or spinal cord infarction.
Imaging Presentation
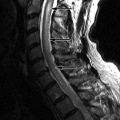
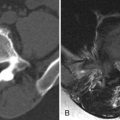
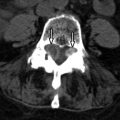
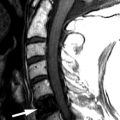
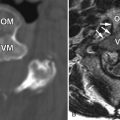
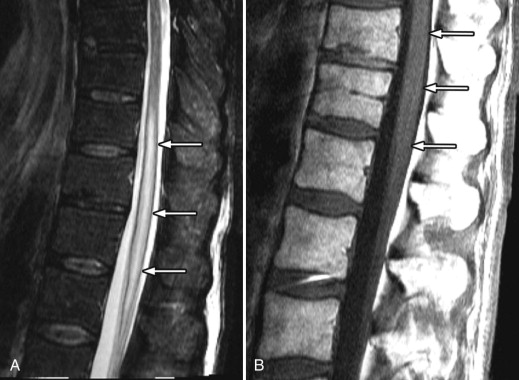
MR imaging revealed a region of abnormal T2 signal hyperintensity involving gray matter in the central portion of the lower spinal cord extending from the T9 level to the L1 level consistent with acute anterior spinal artery territory infarction ( Figs. 69-1 and 69-2 ) .

Discussion
Spinal cord transient ischemic attacks or infarction occur when the blood supply to the spinal cord is compromised. Infarction most often occurs in the lower thoracic spinal cord. The precise etiology is not always known, but a variety of conditions can predispose to spinal cord infarction including aortic or vertebral atherosclerotic disease, aortic dissection, after aortic surgery, aortic stenting, cardioembolic disease, arteritis, cocaine usage, coagulopathy, lupus erythematosus, sickle cell disease, hypotensive episode, spinal infection, sepsis, after spinal cord trauma, and decompression sickness. Smoking, diabetes, atherosclerotic disease, and hypertension are believed to be predisposing factors in many patients who develop spinal cord infarction. Cervical osteophytosis and spinal cord ischemia can also result from spinal atrioventricular (AV) fistula or AV malformation, which shunt blood from the spinal cord parenchyma. Spinal cord infarction can occur rarely after spinal surgery or during embolization of spinal AV fistulas. Spinal cord infarction rarely occurs as a complication of epidural anesthesia or epidural catheter placement. Rare cases of cord infarction may be associated with disc herniation. A case of spinal cord infarction secondary to fibrocartilaginous embolization of the nucleus pulposus has been reported. Despite the many possible causes of spinal cord infarctions, some patients with spinal cord infarction have no known cause or apparent risk factors.
Percutaneous spinal interventional procedures, including epidural injections, celiac plexus blocks, transforaminal epidural injection, or nerve blocks may predispose to AV fistula formation in some cases. Possible etiologies for iatrogenic AV fistula formation include inadvertent injection of the anesthetic and/or steroidal agent into a radiculomedullary artery, injection-related arterial dissection, or injection-induced arterial spasm. Cord infarction should be considered a possibility when cervical, thoracic, or upper lumbar injections are performed. Vertebrobasilar infarction is also a possibility after cervical injections. Even though the reported complication rate is very small with transforaminal epidural injections, to minimize the chance of complication, it is recommended that the injection be performed after digital subtraction angiography (DSA), that a nonparticulate or small particulate diameter steroidal agent be used, and that the patient receive minimal or no sedation for the procedure.
Most spinal cord infarcts involve the anterior spinal artery territory, the gray matter of the anterior horns being most susceptible to ischemia ( Figs. 69-1 to 69-7 ) . This can be understood if one considers the spinal cord arterial supply in cross section. The relatively large but solitary anterior spinal artery generally supplies the anterior two thirds of the spinal cord, whereas the paired smaller posterior spinal arteries supply the posterior one third of the spinal cord. The central portion anterior spinal artery territory is usually supplied by central sulcal arteries originating from the anterior spinal artery that extends into the anterior midline fissure of the cord. There is a rich network of tiny arteries along the surface of the spinal cord, the vasa corona, which anastomose with branches of the anterior and posterior spinal arteries. This peripheral arterial network is more prominent posterolaterally. Penetrating arteries from this network supply the periphery of the spinal cord. There are watershed zones between the peripheral and central cord arterial supply in the anterolateral aspect of the anterior horn gray matter and central portion of the dorsal horn gray matter. A rich venous network also surrounds the spinal cord that drains into the paravertebral venous plexus.





Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree