Figure 10.1
Embryologic origin of parathyroid glands can be suspected from the positioning of the enlarged parathyroid relative to the inferior thyroid (ITA) and superior thyroid (STA) arteries. This type of ultrasound detail also provides helpful orientation to surgeons
Despite the high yield and usefulness of US in parathyroid disease, there are inherent drawbacks to successful evaluation. These include decreased detection secondary to thyroid nodular disease and thyroiditis, increased body mass index, the presence of multiglandular or ectopic parathyroid disease, and the presence of a small parathyroid adenoma [4, 14]. In addition to these factors, thyroid nodules situated in the posterior portion of the thyroid, or benign central compartment lymph nodes, may inadvertently be mistaken for a parathyroid gland and vice versa [4]. Although the minimal amount of tissue between the US probe and the thyroid and parathyroid glands makes US ideal for scanning the neck, it is difficult to visualize a parathyroid gland located behind the trachea, esophagus, in the superior mediastinum, or in patients with thyroiditis who may have significant reactive adenopathy [4]. With the use of US to direct the exposure of the parathyroid glands at surgery, the time to locate the glands is decreased, thereby decreasing the operative time and extent of dissection. Generally speaking, shorter operative times are expected to have quicker recoveries, shorter hospital stays, and increased patient satisfaction.
Thyroid Cancer and Ultrasound Applications Relevant to Surgeons
When presented with a patient with cytology diagnostic of or suspicious for thyroid cancer , the surgeon’s primary concern becomes not so much whether to perform surgery but rather what operation is best indicated. The American Thyroid Association’s 2015 guidelines provide a framework for this decision; however, in order to follow these recommendations, the patient must first be fully assessed for the information that is the basis of those guidelines. As previously mentioned, the report from an US performed by an outside facility may be lacking in relevant content, even if the prior images are obtained and reviewed by the surgeon personally. A thorough, clinician-performed neck US is ideal in every patient diagnosed with thyroid cancer to evaluate for lymph node involvement and contralateral disease. Preoperative evaluation of the neck changes a surgeon’s management in up to 66% of the cases [2]. The most frequent indication for a change in surgical plan was metastatic cervical lymphadenopathy [2]. A partial or near total thyroidectomy may have been the initial plan for a patient as the results of the outside US are discussed in clinic; however, upon the clinician-performed exam, new findings such as additional suspicious nodules in the contralateral lobe or lateral lymphadenopathy may lead to an indication of a total thyroidectomy or a lateral or central neck dissection. Just as surgical management can increase in invasiveness based upon results of the surgeon-directed US, a shift toward avoiding unnecessary surgeries can also occur. Surgeon-performed US is a valuable resource for evaluating for central lymphadenopathy and thoracic extension of the thyroid and diseased lymph nodes. For central neck lymph node metastases, preoperative US performed prior to the initial surgery provides 95% sensitivity, 90% specificity, 97% negative predictive value and 83% positive predictive value [15]. Surveillance after thyroid surgery in the case of thyroid cancer is pivotal in early diagnosis of recurrence. Radioactive iodine scans and cross-sectional imaging studies are costly, time-consuming, and accompanied by radiation. Standard laboratory studies such as thyroglobulin can also be negative in the presence of antibodies or poorly differentiated recurrent disease [2]. Clinician-performed US can detect recurrent disease in patients with otherwise negative or inconclusive standard surveillance methods [2] (see Chap. 8, Ultrasound and Mapping of the Neck Lymph Nodes).
Perioperative Assessment of Vocal Cords
Damage to the recurrent laryngeal nerve is a known complication of thyroid and parathyroid surgery and can carry significant morbidity to the patient. Therefore, both preoperative and postoperative assessment of the vocal cords can inform the surgeon about their functionality. This is especially relevant in the case of a reoperation for recurrent disease, history of other prior neck surgery such as tracheostomy, history of prior neck radiation, or physical exam findings that would suggest possible nerve dysfunction preoperatively [16]. The traditional approach to evaluating the vocal cords is direct laryngoscopy which allows visual examination of vocal cord appearance and movement. However, using US to assess the vocal cords can be almost as effective with an 85–100% visualization rate [17, 18]. Vocal cord US brings with it the benefit of being noninvasive, therefore avoiding the risks and expense of direct laryngoscopy. In the event of a suboptimal US exam, the patient may still proceed with the standard direct laryngoscopy without having sacrificed significant time and resources. Despite the high rate of visualization as well as the benefits of laryngeal US, direct laryngoscopy is still advisable for those patients at higher risk of having a prior injury or showing signs of dysfunction preoperatively (Fig. 10.2).
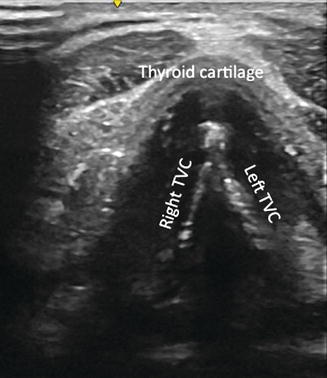

Fig. 10.2
Appearance of true vocal cords (TVC) on laryngeal ultrasound as the brightest white hyperechoic lines in V shape. Laryngeal ultrasound is an application that surgeons are increasingly using for initial assessment of TVC both pre- and postoperatively. Select patients are able to proceed to direct laryngoscopy if appropriate, but many are spared the additional cost and invasiveness of this procedure
Intraoperative US
Not only is surgeon-performed US exam essential in the preoperative clinic setting, but it is also extremely valuable in the operating room for several applications. The use of US imaging once the patient is anesthetized is becoming more popular among endocrine surgeons. There are a number of advantages to this practice, and, aside from the small amount of time to perform the exam, there are no disadvantages to its use. One advantage is aiding optimal placement of the surgical incision (Fig. 10.3). It is the authors’ practice to assess the patient in the preoperative area and look for a natural skin crease for best cosmetic results in the sitting and supine positions. Based on the location of the pathology and the skin creases, more than one crease may be marked, and the most appropriate one is selected based on intraoperative US evaluation. Once the patient is under anesthesia, the head and neck are positioned in the ideal location for the operation. At this point, prior to the sterile skin prep, the neck is examined with the US probe. This study may reveal additional pathology prompting a change in the surgical procedure or extent of the surgical procedure. This view also allows the surgeon to plan their surgical incision more appropriately. In the event there is thoracic extension or newly found lymphadenopathy, the incision may need to be placed further inferior or extended slightly laterally. In patients with parathyroid disease, greater compression of the neck with the transducer probe often elicits deeply situated enlarged glands that were not apparent with office-based US (Fig. 10.4). Once the operation has begun, occasionally despite extensive dissection, a parathyroid gland may not be found and parathyroid hormone levels may still be elevated. At this time, a sterile US probe can be used to evaluate the surrounding tissue for possible ectopic locations of the missing gland. If the gland cannot be located and there appears to be a suspicious nodule within the thyroid tissue on intraoperative US, an aspirate can be sent for parathyroid hormone levels. This saves both the time of dissecting the thyroid and avoids the possibilities of unnecessary thyroid lobectomy or further unrevealing parathyroid exploration.
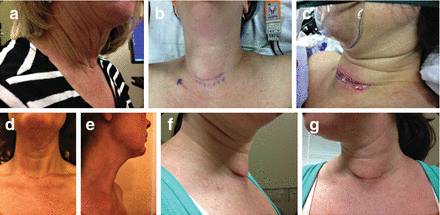
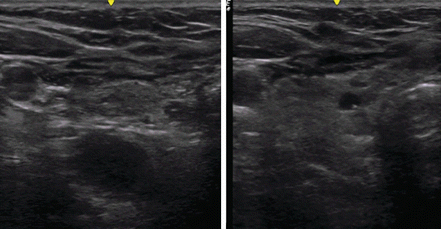

Fig. 10.3
A well-healed cervical incision is barely visible several years after surgery (a). Marking of potential skin incision along existing natural creases is ideally started while the patient is awake and in the preoperative area (b). Surgeon preference varies for using higher- vs lower-situated incisions (c). Two weeks after surgery, this patient was pleased with her scar (d, e). Three months after surgery, this patient (f, g) took her own photos appreciating the cosmetic position of her scar. Ultrasound done after anesthesia and prior to surgery can help select incision placement relative to underlying pathology as well

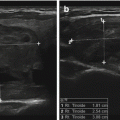
Fig. 10.4
Application of greater transducer pressure (left panel) brings out a parathyroid adenoma (large hypoechoic oval) that with light pressure may stay unrecognized deeper in the tracheoesophageal groove (right panel). A small thyroid cyst is evident in the right thyroid lobe. Surgeons may be more motivated to apply such maneuvers in order to localize the abnormality and thus make surgery more efficient
Robotic and Remote Access Surgery
Although thyroid surgery has become less invasive with smaller incisions, these are nonetheless in a location on the anterior neck that is not easily concealed. This drawback to conventional surgery has led to developments in remote access thyroid surgery allowing for more aesthetically pleasing results. Surgical treatment of thyroid disease via nontraditional approaches is now possible with the use of endoscopic assistance through an axillary, chest, or postauricular incision; the two most frequently used are the robotic axillary thyroidectomy (RAT) and the robotic facelift thyroidectomy (RFT) [4, 19, 20]. Eligibility criteria for such surgeries include benign nodules ≤5 cm or malignant lesions ≤2 cm for the axillary approach and benign nodules <4 cm for the facelift approach [4, 19–21]. While these guidelines exist, there have been studies showing successful RAT and RFT total thyroidectomy as well as total thyroidectomy with bilateral modified neck dissection [4, 22, 23]. These studies, however, are small in size and lack the long-term follow-up needed to fully assess outcomes. The use of US is instrumental in determining eligibility for RAT and RFT because it evaluates the overall size and appearance of the thyroid gland preoperatively and allows the surgeon to establish characteristics of the individual’s disease that influence the type of surgery offered [4, 17]. For both the RAT and RFT approaches, US findings of Hashimoto’s thyroiditis or extrathyroidal, substernal, or retropharyngeal extension are exclusion criteria [4, 24, 25]. The specific location of the nodule within the thyroid gland is also taken into account, with the ideal position in the midportion of the thyroid lobe [4, 26]. This location enables the surgeon to grasp the superior pole for manipulation without damaging the capsule of the cancer and permitting an easier, thus, quicker operation [4]. Despite the appealing nature of nontraditional approaches for cosmetic reasons, they have not been widely adopted and are only performed at a small number of facilities in the United States.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree