- •
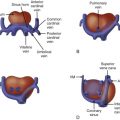
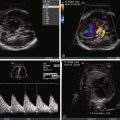
Monochorionic pregnancy with size discrepancy between twins.
- •
For the smaller donor, heart function appears ostensibly normal with small ventricular cavity volumes and hyperdynamic systolic function.
- •
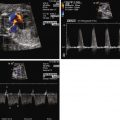
Umbilical artery impedance (pulsatility index) can be higher than normal.
- •
For the larger recipient, a number of cardiovascular findings are common including:
- •
Cardiomegaly.
- •
Ventricular hypertrophy.
- •
Poor systolic function.
- •
Atrioventricular valve regurgitation.
- •
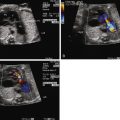
Abnormalities of Doppler flow suggesting poor right ventricular compliance and diastolic dysfunction such as reversal in the ductus venosus and umbilical venous pulsations.
- •
Abnormalities of Doppler inflow across the tricuspid and mitral valves, namely formation of a single-peak inflow.
- •
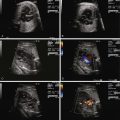
Pulmonary insufficiency.
- •
Right ventricular outflow tract obstruction manifested as a smaller than normal pulmonary artery relative to the aorta, “functional” pulmonary atresia, or frank anatomical pulmonary atresia.
- •
Reversal of flow in the ductus arteriosus in the presence of right ventricular outflow tract obstruction.
- •
- •
Look for signs of hydrops in the recipient.
Anatomy and Anatomical Associations
The twin-twin transfusion syndrome (TTTS) is a disorder that occurs in monochorionic multiple pregnancies. Placental vascular connections provide the substrate for exchange of volume and mediators between twin fetuses, which leads to a host of abnormal findings for both. One twin, the recipient, is larger, manifests polyhydramnios, and develops cardiovascular changes reflective of volume load and ventricular hypertrophy leading to cardiomyoapthy. The other twin, the donor, is smaller, manifests oligohydramnios, and develops hypovolemia and increased vascular tone. The term “transfusion syndrome” is a bit of a misnomer and an oversimplification of the pathophysiological process because hemoglobin levels measured in the twin partners are usually not different from each other. However, volume exchange between the twins prompts a cascade of events that results in profound physiological changes for both.
Overall, TTTS manifests significant morbidity and mortality and is fatal to one or both twins in over 90% of cases if not treated in some manner.
Frequency, Genetics, and Development
TTTS is seen in approximately 15% of monochorionic twin pregnancy. With increasing use of reproductive technologies, twinning in general is occurring more frequently in the human population. Hence, screening for TTTS, the means of accurate detection, an improved understanding, and the actual number of patients with this disorder are all on the rise.
TTTS is a “placental vasculopathy” and is a disorder that can be considered primarily of cardiovascular origins. In the normal monochorionic pregnancy, multiple placental vascular connections naturally allow for exchange between the twin partner circulations. Exchange takes place at arterial-to-venous connections with equilibration of net flow at arterial-to-arterial connections. The arterial-to-arterial connections compensate for any net flow from one twin to the other and keep the relative circulatory volumes stable. However, if there are an inadequate number of such arterial-to-arterial connections, equilibration is hampered and flow due to arterial-to-venous connections predominates, resulting in volume load to one twin and volume depletion to the other. TTTS is presumed due to an inadequate number of such arterial-to-arterial connections. Furthermore, abnormalities of donor cord insertion have been described as a possible precursor to TTTS. Insertion of the donor umbilical cord onto the edge of the placenta with reduction in vascular territory or the velamentous insertion of the cord onto the placenta (velamentous insertion means that the veins, unprotected by Wharton’s jelly, traverse the membranes before they come together into the umbilical cord) may predispose to creating disparity in blood flow between the twins.
There are no current data to suggest a genetic basis to TTTS. In a recent large review of karyotype of fetuses undergoing therapy for TTTS, only 1% of 600 fetuses were found to have abnormalities including Turner’s syndrome (46,XO) and 47, XYY. The authors concluded that abnormal karyotype in TTTS is no different than what is seen in the general population.
Prenatal Physiology
The pathophysiology of TTTS and its cardiovascular manifestations are not fully understood. However, based on observable findings, a working plausible hypothesis has been generated. With net transfer of volume between the donor twin and the recipient, a cascade of events takes place ( Figure 38-1 ). First, the donor twin experiences progressive hypovolemia. The normal response to hypovolemia is an increase in systemic vascular resistance, in order to preserve perfusion pressure. In order to achieve this goal, a host of mediators are released, such as endothelin-1. The renin-angiotensin system is up-regulated with release of angiotensin II. Observation of the donor twin reveals a small heart size, with underfilled ventricular cavities, and a hyperdynamic state with ostensibly good systolic ventricular function. Urine production is diminished, visualization of the bladder may be limited, and oligohydramnios occurs. The recipient twin experiences an increase in volume load, but in addition to a transfer of volume, it also receives the various vasoactive mediators released by the donor. Recent data also suggests renin-angiotensin release from a portion of the recipient placenta may also contribute. In essence, these mediators compound the pathological response of the recipient’s cardiovascular system to the volume load. Observation of the recipient reveals ventricular dilation and marked hypertrophy. Progressive cardiomegaly, atrioventricular valve regurgitation, and development of a characteristic cardiomyopathy ensue. Increase in fetal brain natriuretic peptide (BNP) as a consequence of these cardiomyopathic changes has been found in recipients. Angiotensin II in combination with increase in preload are the likely agents responsible for what can be observed as a relatively rapid changeover from a normal size and normal functioning heart to a dilated, hypertrophied and poorly functioning heart, all in a matter of weeks.

TTTS can be seen as early as 14 to 16 weeks’ gestation. The cardiovascular changes can be observed to take place with rapid progression from 18 to 28 weeks. Besides the changes of ventricular dilation, hypertrophy, and dysfunction, diastolic changes in myocardial compliance can be seen early in the process and are reflected by changes in ventricular filling parameters such as the E : A wave ratio across the tricuspid or mitral valves. Right-sided changes always precede, and are more severe, than left-sided changes. Initially, cardiac output in the recipient is increased relative to the donor. As the process progresses and systolic and diastolic dysfunction sets in, cardiac output diminishes. Measures of ventricular performance such as the myocardial performance index are abnormal in TTTS and reflect both systolic and diastolic dysfunction.
If TTTS is left unchecked, severe ventricular dysfunction can progress to development of hydrops and fetal demise. In addition, a subset of recipient twins, approximately 10%, develop pulmonic stenosis and right ventricular outflow tract obstruction (RVOTO). This has been referred to as the development of “acquired” congenital heart disease, a fascinating phenomenon that reflects the potential plasticity of the human heart to convert from normal to a form of structural heart disease, in response to flow dynamics, loading conditions, and mediator release during a vulnerable time of gestation.
Recipient cardiomyoapthy can lead to impaired cerebral perfusion and neurological insult. Severe hypovolemia and oliguria in the donor can also result in poor growth. Umbilical arterial flow in the donor is altered as placental vascular resistance is elevated. This is reflected in the findings of either absent or at times reversed umbilical diastolic arterial flow, an ominous sign suggesting impaired placental flow. Death of one twin in untreated TTTS is common. Because there are vascular connections within the placenta uniting the two circulations, death of a twin results in creation of a vascular sink that can acutely drain volume from the surviving co-twin. Such sudden hypoperfusion can also lead to neurological insult or secondary demise in the survivor; hence, the fates of both twins in TTTS are interrelated.
Prenatal Management
Fetal treatment for TTTS is currently possible and very effective. But who is to be treated, and how is the spectrum of the disease best described? The basic criteria for diagnosis of TTTS are
- 1
Monochorionic pregnancy.
- 2
Size discrepancy of at least 10% between the twins.
- 3
Oligohydramnios in the smaller twin and polyhydramnios in the larger twin.
A scoring system to gauge disease severity in TTTS exists. Quintero and coworkers developed a five-stage system based on clinical and ultrasound findings ( Table 38-1 ). This system has been extremely useful in guiding therapy and response to therapy.
| QUINTERO STAGE | FINDINGS |
|---|---|
| I | Twin size discrepancy and polyhydramnios + oligohydramnios sequence |
| II | Absent bladder filling in donor |
| III | “Critical Doppler abnormalities” (UA, UV, and ductus venosus) |
| IV | Fetal hydrops |
| V | Fetal demise |
Because TTTS is inherently a cardiovascular disorder, we have argued that the Quintero system is inadequate in fully describing and characterizing the complement of cardiovascular sequelae that exists. Based on observations made in hundreds of twin pairs that have been referred to our center at Children’s Hospital of Philadelphia (CHOP), we have developed a disease specific cardiovascular scoring system for TTTS ( Table 38-2 ). The CHOP TTTS Cardiovascular Score assesses five different domains of cardiovascular function, with grades given for various abnormal findings. Four of the domains relate to cardiovascular findings in the recipient twin, and the final domain addresses the donor umbilical artery flow. No structural cardiac changes are typically seen in the donor; however, there is commonly evidence of elevated placental resistance manifested as a decreased or reverse flow in diastole when sampling in the umbilical artery. In particular, one of the domains in the recipient addresses the issue of RVOTO. Normally, the pulmonary artery should always be larger than the aorta. We have observed that, in some recipient twins, the pulmonary artery is either the same size or smaller than the aorta, this in advance of the findings of frank pulmonary or right ventricular outflow tract stenosis. This suggests a spectrum of right-sided obstruction, starting with very subtle differences. Furthermore, pulmonary valve insufficiency suggesting early functional change is present in some recipients as well, in advance of frank pulmonary stenosis of outflow tract obstruction. We have, therefore, incorporated these variables in our evaluation system.
| Ventricular Elements | |||
| Cardiac enlargement | None (0) | Mild (1) | >Mild (2) |
| Systolic dysfunction | None (0) | Mild (1) | >Mild (2) |
| Ventricular hypertrophy | None (0) | Present (1) | |
| Valve Function | |||
| Tricuspid regurgitation | None (0) | Mild (1) | >Mild (2) |
| Mitral regurgitation | None (0) | Mild (1) | >Mild (2) |
| Venous Doppler | |||
| Tricuspid inflow | Two peaks (0) | One peak (1) | |
| Mitral inflow | Two peaks (0) | One peak (1) | |
| Ductus venosus | All forward (0) | Decreased (1) | Reversal (2) |
| UV pulsation | None (0) | Present (1) | |
| Great Vessel Analysis | |||
| Outflow tracts | PA > Ao (0) | PA = Ao (1) | PA < Ao (2) RVOTO (3) |
| Pulmonary insufficiency | None (0) | Present (1) | |
| Umbilical Artery Flow in Donor | |||
| UA Doppler | Normal (0) | Decreased diastolic flow (1) | Absent of reversed diastolic flow (2) |
The five domains and the point values for the various elements of the CHOP TTTS Cardiovascular Score are:
- 1
Ventricular characteristics such as ventricular hypertrophy (absent = 0, present = 1), ventricular dilation (cardiothoracic ratio >0.33 = 1, >0.5 = 2), and systolic dysfunction (absent = 0, mild dysfunction = 1, more than mild dysfunction = 2).
- 2
Atrioventricular Valvar function , with assessment of tricuspid regurgitation (absent = 0, mild = 1, greater than mild = 2) and mitral regurgitation (absent = 0, mild = 1, greater than mild = 2).
- 3
Venous Doppler findings that reflect diastolic performance such as tricuspid inflow pattern (normal two-peak inflow = 0, abnormal single-peak inflow = 1) and mitral inflow pattern normal two-peak inflow = 0, abnormal single-peak inflow = 1); ductus venosus flow (normal all forward = 0, decreased forward flow with atrial contraction = 1, reversal of flow with atrial contraction = 2); and umbilical vein pulsations (normal absent = 0, present = 1).
- 4
Great vessel analysis looking at the ratio of size of the pulmonary artery to the aorta (normal pulmonary artery larger than the aorta = 0, pulmonary artery appearing close to the aorta in size, suggesting abnormal smallness = 1, pulmonary artery measures smaller than the aorta = 2, pulmonary stenosis or RVOTO = 3); and looking at grade of pulmonary insufficiency (none = 0, present = 1).
- 5
Umbilical arterial flow in the donor (normal flow with ample diastolic flow = 0, decreased diastolic flow reflecting elevated placental vascular resistance = 1, absent or reversed diastolic flow reflecting severely elevated placental vascular resistance = 2).
The higher the score, the greater the cardiovascular burden of disease, with a maximum score of 20 points possible. Patients can be categorized into quartiles with cardiovascular severity grades offered: score 1-5 = severity grade I; score 6-10 = severity grade II; score 11-15 = severity grade III; score 16-20 = severity grade IV.
A comparison of the Quintero scoring system with the CHOP score reveals some overlap but also substantial discrepancy in that many subjects with relatively low Quintero scores often have higher cardiovascular severity grades, reflecting the inadequate nature of the Quintero system to accurately capture and describe the cardiovascular phenomenon taking place in TTTS ( Figure 38-2 ).