Introduction
The term chronic venous insufficiency most often refers to venous valvular incompetence in the superficial, deep, perforating and/or the nonsaphenous veins of the lower limb. Although it is thought to affect approximately 40% to 50% of the population and has been widely researched, this form of venous dysfunction remains poorly understood. Incompetence of the vein valves permits retrograde flow (reflux), which is opposite to the normal blood flow direction toward the heart. Chronic venous insufficiency (CVI) may result from pathologic dilation of a vein or veins, recanalization of previously thrombosed venous segments, or congenital absence of competent vein valves. Approximately 80% of patients with CVI have reflux alone. Venous reflux and residual venous obstruction are present in 17%, and obstruction alone is an uncommon etiology. Elevated ambulatory venous pressure is a sequela of clinically significant valvular incompetence in the lower extremity veins and, if untreated, commonly results in ulceration. The physical findings associated with venous insufficiency are characteristic but not indicative of the cause of venous valve incompetence. The clinical presentation does not generally allow differentiation between venous obstruction and valvular incompetence, nor does it define the location or extent of valvular dysfunction.
In recent years, duplex sonography has become the procedure of choice for evaluating venous disease. The attractiveness of this modality stems from the fact that it is noninvasive, provides both anatomic and physiologic information, is reproducible, and can be performed portably at relatively low cost. B-mode (gray-scale) imaging excels at defining the presence and extent of venous obstruction, and color Doppler imaging facilitates differentiation of antegrade and retrograde blood flow. Doppler spectral waveform analysis is used to confirm venous blood flow direction and to demonstrate the presence or absence of phasic respiratory changes, the amount of blood flow, and the response to limb compression or Valsalva maneuver.
Lower Limb Venous Anatomy
The anatomy of the venous system was reviewed in Chapter 18 . Because lower limb venous anatomy is variable, the sonographic evaluation may be straightforward or quite complex. Careful attention must be given throughout the examination to spatial relationships of vessels, bones, and muscles; presence of saphenous vein fascial boundaries; vein diameters; venous duplication; agenesis; aplasia or hypoplasia, dysplasia; and atrophy. The examination for venous insufficiency is in large part focused on the superficial veins, their major tributaries, and the perforating veins. To ensure an accurate and complete ultrasound evaluation, it is important to review briefly the anatomy of these veins in normal and diseased limbs.
The great saphenous vein and the saphenofemoral junction
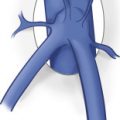
The great saphenous vein (GSV) originates on the dorsum of the foot, anterior to the medial malleolus, and courses cephalad along the tibial border to the level of the knee. It crosses the knee and continues upward, most often to the level of the groin crease, where it becomes confluent with the common femoral vein forming the saphenofemoral junction (SFJ). The GSV is usually easily identified with ultrasound imaging as it lies within the saphenous compartment between the echogenic superficial and muscular fascia. When viewed in a transverse image plane, the vein and its surrounding fascia form what has been termed the Egyptian eye ( Fig. 21.1 ). Superficial venous branches that originate from the GSV in this fascial compartment are called tributaries.

Early studies indicated that the GSV is duplicated in the thigh in approximately 8% of patients and in the calf in approximately 10% of cases. More recent studies based on duplex sonography suggest that true duplication of the GSV is uncommon, occurring in only 1.6% to 2% of the population. A common anatomic variant is quite often the cause of erroneous assumption of duplication of the GSV. In a significant number of patients, the GSV will lie within its fascial compartment for approximately 10 to 15 cm in the proximal thigh. A significant tributary, the anterior accessory of the GSV, exits the fascia and courses superficial to the fascia along the same path as the GSV. Most often, the anterior accessory of the GSV will enter the saphenous fascial compartment below the knee.
A study by Spivack and colleagues reported a mean GSV diameter range between 2.3 mm and 4.4 mm but the commonly assumed tapering pattern of the GSV as it descends the leg was not apparent. An additional finding of interest in this study was that the vein was smaller at the distal calf than at the ankle. This study and others highlight the issue of differentiating the true GSV from its subcutaneous tributaries. Three anatomic sonographic patterns have been identified and are illustrated in Fig. 21.2 . Type “i” consists of a GSV with a normal diameter and no large tributaries coursing parallel in the GSV fascia. With a type “h” pattern, the GSV stays within its fascial compartment along its full length, but there is a venous tributary that may have a larger diameter than the GSV. In limbs with a type “s” pattern, the GSV is absent or hypoplastic in the lower calf. In its place, a superficial tributary ascends the lower calf, piercing the superficial fascia to become the GSV.

Approximately 1 to 2 mm distal to the SFJ, the terminal valve can be identified ( Fig. 21.3 ). A preterminal valve is frequently seen approximately 2 cm distal to the terminal valve. Venous blood is drained from major lateral and medial abdominal tributaries (the superficial circumflex iliac, superficial inferior epigastric, and superficial and deep pudendal veins) that join the GSV between the two valves ( Fig. 21.4 ). In a significant number of cases, these tributaries may be the source of reflux even when the terminal GSV valve is competent.


The anterior accessory of the great saphenous vein
The anterior accessory of the great saphenous vein (AAGSV) can be identified as it courses obliquely in a separate fascial compartment parallel to the GSV in the anterior thigh. Its path is anterior to the superficial femoral artery, deep femoral artery, and the femoral vein and lateral to the GSV. As such, on a transverse ultrasound image from the upper thigh, the AAGSV and the GSV form two saphenous eyes. The AAGSV courses anterolateral to the GSV in alignment with the femoral vessels ( Fig. 21.5A and B ). The alignment sign serves as a reliable tool for recognition of the AAGSV. The AAGSV most often joins the GSV within 1 to 2 cm of the SFJ. On occasion, it may become confluent with the GSV in the distal thigh or it may physically or functionally replace an aplastic or hypoplastic thigh segment of the GSV. It is estimated that the AAGSV is responsible for varicosities in the anterior, mid, or lower thigh in approximately 14% of patients.


The posterior accessory of the great saphenous vein
The posterior accessory of the great saphenous vein (PAGSV) may be seen in the posterior thigh coursing within a separate fascial compartment parallel and slightly posterior to the GSV ( Fig. 21.6 ). Although it is not found as often as the AAGSV and its confluence with the GSV is quite variable, it should be assessed for reflux in patients presenting with mid to distal thigh varicosities. The posterior arch vein , formerly referred to as Leonardo’s vein, is the portion of the PAGSV that originates behind the medial malleolus and courses on the medial aspect of the leg to join the GSV distal to the knee. It is important to note that the posterior tibial vein perforators often join this vein rather than the GSV.

The small saphenous vein
The small saphenous vein (SSV) is a continuation of the lateral marginal vein of the foot and ascends along the midline at the back of the calf, coursing between the heads of the gastrocnemius muscle, and frequently terminating at the popliteal vein within the popliteal fossa. Joh and Park reported that the normal diameter of the vein is approximately 3.1 ± 1.3 mm in patients lying supine. With patients in a standing position, Kurt and his colleagues obtained a mean diameter of 3.89 mm for the right limb and 4.03 mm for the left.
The SSV lies within its own fascia, as does the GSV, appearing as a saphenous eye. As with the GSV, SSV anatomic variants are common. When imaging in a transverse plane, subcutaneous tributaries can be seen entering the saphenous compartment through the superficial fascia to empty into the SSV. These tributaries are frequently the source of reflux. Perhaps the most important of these is the lateral arch vein that communicates, via lateral calf perforating veins, with the peroneal veins.
The saphenopopliteal junction
The SSV normally ends above the popliteal crease but usually within 5 cm of this body surface landmark. The SSV joins the popliteal vein laterally in the majority of cases but the site of confluence is variable. There are three common anatomic patterns ( Fig. 21.7A to C ). The SSV may join the popliteal vein at the popliteal fossa along with other deep veins through a thigh extension (TE). Alternatively, it may ascend as the TE, connecting with the popliteal vein through a small venous channel. It has also been shown that the SSV may not connect at all to the popliteal vein but may ascend as the TE (Giacomini vein) and join the GSV or deep veins. It is also important to note that in 10% to 30% of lower limbs, the SSV may join the gastrocnemius veins (GGV) before joining the popliteal vein. Although the GGV often join the popliteal vein separately, they may merge with the SSV and the popliteal vein as a common gastrocnemius-saphenous-popliteal junction.

The thigh extension
A TE is found in approximately 95% of limbs and may be considered the proximal extension of the SSV into the thigh. It courses deep to the fascia along the back of the thigh and terminates in superficial or perforating thigh veins or in the gluteal veins, but may also terminate in the GSV or deep veins. The Giacomini vein (GV) usually refers to that part of the TE that communicates the SSV with the GSV. Distally, it is located within the fascia surrounding the saphenous and, like the saphenous veins, may course as a single vessel or as multiple muscular or subcutaneous channels. As such, the TE may be the source of reflux from more proximal veins. The TE may terminate into the GSV, the posterior thigh muscle veins, the femoral vein or less commonly, into branches of the internal iliac vein, such as the inferior gluteal or internal pudendal vein .
The perforating veins
Perforating veins connect the superficial and deep venous systems. With the exception of the foot perforators, their role is to direct blood flow from the superficial to the deep venous system. In general, they have limited external support and functionally dilate in response to alterations in pressure and volume. There are more than 40 constant perforating veins and although they are usually single, like many other veins, they may be duplicated and are commonly accompanied by an artery. Perforating veins may empty into the deep veins (direct perforators) or into the venous sinuses in the calf (indirect perforators). Direct perforators tend to be anatomically constant; indirect perforators are usually randomly distributed in the calf.
There are four main groups of calf perforators: the posterior, lateral, anterior, and medial. The posterior leg perforators consist of the medial and lateral gastrocnemius perforators, the soleal perforators (which connect the SSV with the soleal veins), and the para-Achillean perforators (which connect the SSV with the peroneal veins). The lateral leg perforators connect lateral calf veins with the peroneal veins, and the anterior leg perforators connect the anterior GSV tributaries to the anterior tibial veins. The medial leg perforators consist of the paratibial and posterior tibial perforators. The GSV and its tributaries are connected to the posterior tibial and calf muscle veins via the paratibial perforators. The posterior tibial perforators (formerly known as Cockett perforators) connect the posterior arch vein with the posterior tibial veins.
The knee perforators include the medial or lateral perforating veins, the suprapatellar or infrapatellar perforators, and the popliteal fossa perforators.
Thigh perforators include the anterior, medial, lateral, and posterior perforating veins.
Gluteal muscle perforators are divided into superior, mid, and lower perforating veins.
- •
The GSV lies within its own fascial compartment whose boundaries look like an “Egyptian eye” on ultrasound imaging.
- •
The two major great saphenous branches, the anterior accessory and the posterior accessory of the GSV, also have a separate fascial compartment.
- •
The SSV has its own fascial envelope
- •
The SSV can terminate at the popliteal vein but often continues as the TE or GV that then courses up the posterior thigh.
- •
The GV refers to the TE of the SSV that communicates between the SSV and the GSV.
- •
Perforating veins are present in the thigh, around the knee, and in the calf. The calf perforators are grouped as medial lateral, anterior, and posterior.
Pathophysiology of Venous Insufficiency
Normal venous physiology was discussed in Chapter 18 . It is important to appreciate that venous valves are present in the deep, superficial, and perforating veins with the greatest number located in the calf veins. Investigators have shown that there is usually one valve present above the SFJ within the external iliac or common femoral vein, another at the junction, at least six valves along the trunk of the GSV, and seven to ten within the SSV.
Resting venous pressure averages approximately 90 mm Hg in an erect person at rest and is a function of the patient’s height. Ambulation activates calf muscle contraction that then propels, or augments, venous blood flow toward the heart. During contraction, the valves distal to the muscles and those in the perforating veins close to prevent reflux. This reduces venous pressure in the foot to 20 to 30 mm Hg. During relaxation, the venous system is slowly refilled from arterial inflow but venous pressure within the calf remains low and blood flows from the superficial to the deep veins via perforating veins. When extremity vein valves are incompetent, blood is able to move from the deep to the superficial veins during muscle contraction. With relaxation, incompetent valves allow blood to flow in a retrograde direction. This results in an uninterrupted column of blood under the influence of gravitational and hydrostatic pressure, a scenario that leads to a persistent elevation of venous pressure at rest and during exercise. Depending on the degree of reflux, persistent venous pressure elevation can promote the leakage of protein-rich fluid and blood cells through the capillary walls into the intercellular spaces. The immediate result is soft tissue edema, but the long-term sequelae include skin thickening, hyperpigmentation, and ultimately ulceration, particularly in patients who have both valvular incompetence and venous obstruction. Approximately 20% of people with CVI develop venous ulcers and recurrence after treatment is very common.
Although identification and quantification of venous reflux is the primary focus of the sonographic evaluation of venous insufficiency, the presence and location of venous varicosities must also be documented. Lower-limb varicose veins are most often found along the trunk of the GSV. The majority of varicosities are large, with a diameter exceeding 4 mm, palpable and tortuous. Reflux is commonly present when the vein wall weakens and the valve cusps dilate to the point where valve leaflets no longer meet when closed. The prevalence of varicose veins is estimated to exceed 25 million adult cases in the United States. The United States National Venous Screening Program, conducted by the American Venous Forum, identified varicose veins in more than 30% of participants.
Normal vein valve closure depends on the reversal of the normal transvalvular pressure gradient that causes sufficient reversal of blood flow to force the valve leaflets to coapt. Van Bemmelen and his associates noted that valve closure was achieved when reverse blood flow velocities exceeded 30 cm/s. This normally occurred in less than 0.5 s in the saphenous veins when the patient was standing erect. Reflux was demonstrated only when the valves were incompetent and a significant transvalvular pressure gradient was present. During a venous duplex examination, the velocity of retrograde blood flow is related to the external pressure applied to the vein. It must be noted that sufficient pressure is not uniformly achieved with either the Valsalva maneuver or manual compression, particularly in the more distal veins. This can result in failure to detect venous incompetence. To achieve the greatest accuracy, compression of the limb distal to the valve site should be performed using standardized pneumatic cuff compression techniques as discussed later in this chapter.
Segmental valvular incompetence is common in both the deep and superficial venous systems. Patients with venous ulceration often have three or four incompetent segments involving the deep or superficial venous systems. A very high percentage of patients with ulceration will demonstrate incompetence of the superficial veins, whereas incompetence of the deep veins at ankle level is less common.
In an excellent study reported by van Bemmelen and Bergan, the GSV was shown to be incompetent at knee level in 61% of limbs, at the level of the calf in 49%, and in the proximal thigh in 32% of limbs. This finding emphasizes the prevalence of distal superficial vein incompetence, while the more proximal segments of the GSV remain functional. When the GSV was found to be incompetent at knee level, the GSV was continuously incompetent from the SFJ to the knee in less than 50% of patients. Venous reflux was found in the femoral and popliteal veins in 34% of patients who had GSV incompetence at knee level and functional proximal superficial venous segments. In these cases, an incompetent perforating vein was identified at the upper end of the incompetent GSV segment.
Segmental incompetence is often also found in the SSV. The previously cited study reported that valvular incompetence was present in the proximal SSV segment in 36% of limbs, while the calf segments were incompetent in 31% of limbs. If the distal segments of the SSV remained competent, flow from the incompetent proximal segments was diverted to superficial venous branches.
The anatomic sources of lower limb venous insufficiency are not confined to the GSV, SSV, perforator veins, or the deep veins of the extremity, but may include the nonsaphenous and pelvic veins. The sonographic examination should be extended into the inferior vena cava (IVC), renal, ovarian, para-uterine, and iliac veins in women who present with complaints of chronic pelvic pain and, quite often, a dilated and incompetent GSV. The IVC, renal, gonadal, and iliac veins should be assessed in men with proximal saphenous incompetence. Patients presenting with SFJ incompetence may demonstrate reflux in the various proximal saphenous tributaries or the venous plexus surrounding the inguinal lymph nodes.
- •
Persistent elevation of lower leg venous pressure is the cause of the ulcerations seen in chronic venous insufficiency.
- •
Although calf pump action can decrease venous pressure in the case of intact venous valves, incompetent venous valves allow venous pressures to return to their baseline (hydrostatic) levels more rapidly.
- •
Venous blood flow toward the heart is promoted by the open vein valves, whereas a reversal of trans-valve pressure and reversed blood flow are needed for valve closure.
- •
Venous reflux need not be continuous from the SFJ to the foot along the GSV:
- •
in a significant percentage of cases, reflux is seen at the level of the knee but not in the proximal thigh
- •
a plausible source of reflux is then an incompetent thigh perforator
- •
- •
SSV reflux present near the popliteal is often not seen at the mid-calf. Refluxing blood is diverted to proximal tributaries.
Clinical presentation
The clinical hallmarks of CVI include limb edema, dilated cutaneous veins (telangiectasias and reticular veins), varicose veins ( Fig. 21.8 ), limb pain, skin thickening, hyperpigmentation in the region of the ankle, and ulceration. Patients with incompetence of the superficial, deep and perforating veins may present with the full spectrum of symptoms, whereas those with only segmental saphenous vein incompetence may experience lesser degrees of disability.

The initial sign of valvular incompetence is usually ankle edema that is relieved by rest and limb elevation. In more severe cases with multiple levels of venous incompetence, the edema may involve the leg to the mid-calf level and may be associated with pitting when moderate pressure is applied to the skin.
With early saphenous vein incompetence, the superficial veins of the calf may become dilated in response to the persistently elevated venous pressures. This most often affects veins at the medial aspect of the lower calf and around the ankle. Over time and in more severe cases, the veins become quite prominent and tortuous.
Patients with incompetent valves frequently describe a feeling of heaviness and aching in their legs after a period of prolonged standing or sitting with their legs dependent. In the absence of concomitant venous obstruction, the symptoms may subside with ambulation or limb elevation, actions that relieve venous congestion. In contrast, if the deep veins are obstructed, patients describe severe cramping and burning pain associated with exercise. These signs, consistent with venous claudication, persist as long as the extremity veins remain congested and are likely to be the result of the exercise-related rapid increase in pressure in both the superficial and deep veins.
- •
Early venous reflux can cause varicose veins at the distal medial calf.
- •
Clinical symptoms depend on the degree of reflux and venous obstruction:
- •
improved symptoms with ambulation/exercise probably indicates reflux and vein incompetence
- •
worsening symptoms with exercise probably indicates a component of venous obstruction
- •
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree