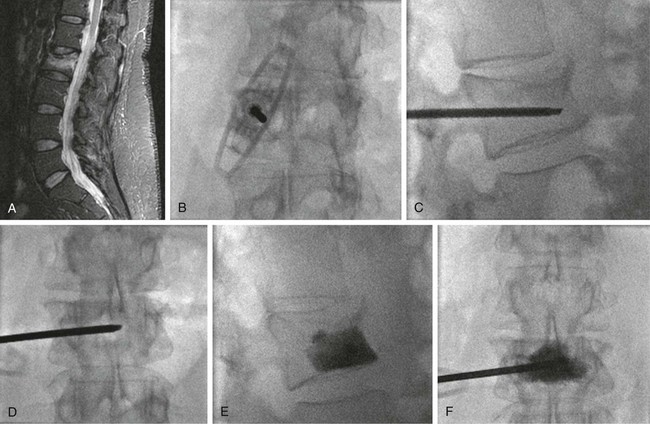
David A. Pastel, Albert J. Yoo, Clifford J. Eskey and Joshua A. Hirsch Osteoporosis is a prevalent disease that affects 200 million people worldwide.1 Fractures from osteoporosis have an incidence of roughly 150 per 100,000 in women (half this rate in men) and result in substantial pain, morbidity, and healthcare utilization.2 In the United States alone, approximately 700,000 osteoporotic vertebral compression fractures occur each year.3 Vertebroplasty and kyphoplasty are minimally invasive image-guided procedures that involve injection of bone cement into a fractured vertebral body, with the primary goal of pain relief and secondary goals of vertebral body stabilization and restoration of height. The primary difference is that with kyphoplasty, a cavity is created with a mechanical device before cement delivery. Before the introduction of percutaneous vertebral augmentation, treatment of compression fractures consisted variably of bed rest, pain control (with nonsteroidal antiinflammatory drugs [NSAIDs], calcitonin, narcotics), and back bracing.4 Percutaneous treatment of vertebral compression fractures was first performed in France in 1984 by Galibert and Daimond.5 Vertebroplasty was first introduced in the United States in the early 1990s and rapidly gained widespread use based on observational data. In 2002, approximately 54,000 patients with vertebral compression fractures were treated with vertebroplasty or kyphoplasty. This number rose to 130,000 in 2007. Since that time, several landmark randomized prospective studies have been published. Two high-profile studies published in the New England Journal of Medicine in August 2009 that compared vertebroplasty to sham vertebroplasty for treating osteoporotic compression fractures found no statistically significant difference between the two groups.6,7 These studies have been criticized for potential inclusion of patients with chronic fractures, absence of a control group without intervention, difficulty in recruitment, and inconsistent use of physical exam and bone marrow edema on magnetic resonance imaging (MRI) as inclusion criteria. These trials contrast with a more recent nonblinded randomized controlled trial where patients who underwent vertebroplasty for treatment of acute vertebral compression fractures experienced superior pain relief compared to a control group that was conservatively managed up to 1 year.8 Another randomized controlled study comparing kyphoplasty to conservative management yielded similar results at 1 year.9 The shortcomings of these trials underscore the difficulty in conducting adequately powered clinical trials. Despite the landmark work done to date, controversy still surrounds even the basic efficacy of vertebral augmentation procedures. The primary indication for vertebroplasty and kyphoplasty is a painful unhealed compression fracture that has failed conventional medical therapy. This failure may represent inadequate pain relief from narcotic analgesia, undesirable side effects from narcotic analgesia, or hospitalization for pain. In the United States, the vast majority of fractures evaluated for treatment are related to primary osteoporosis. Other treatable fractures include those secondary to steroid-induced osteoporosis (e.g., transplant recipients, patients with chronic obstructive pulmonary disease or autoimmune disease),10 metastatic disease, hematopoietic neoplasm (leukemia, multiple myeloma), or rarely trauma.11 Vertebrae may also be treated if there is no fracture but a painful neoplasm or vascular tumor such as hemangioma. At this time, there is little justification for prophylactic treatment of unfractured vertebral levels because of the difficulty predicting which levels, if any, will sustain a fracture in the future.12 Pathologic fractures constitute an important subgroup of treatable fractures because of additional disease-specific issues that have to be addressed both before and during the procedure. One such issue includes the timing of vertebral augmentation in relation to tumoricidal treatment. Some practitioners advocate performing vertebral augmentation after radiation therapy to prevent the theoretic risk of tumor dissemination during pressurized cement injection.10 However, others claim the procedure may be performed at any time—before, during, or after chemotherapy or radiation therapy.13 The few absolute contraindications to vertebroplasty and kyphoplasty include current systemic or spinal infection, uncorrectable bleeding diathesis, insufficient cardiopulmonary health to tolerate sedation or general anesthesia, and myelopathy or radiculopathy secondary to fracture-related compromise of the spinal canal or neural foramina. In the case of infection, the patient must complete an adequate course of antibiotics and must be afebrile with resolution of leukocytosis before the procedure is performed. With regard to fracture-related neurologic dysfunction, some practitioners consider radiculopathy a relative contraindication.10 The American College of Radiology practice guideline also lists allergy to cement or opacification agents as an absolute contraindication. • Disruption of the posterior cortex increases the risk for posterior cement leakage and therefore the risk for spinal cord or nerve root compression. Though rare in osteoporotic compression fractures, this feature is frequently seen in burst fractures and neoplasms. The integrity of the posterior cortex is best evaluated with computed tomography (CT). • Epidural extension of tumor in the setting of pathologic fractures allows egress of cement into the spinal canal, because cement commonly fills the intraspinal soft-tissue mass.14 Shimony et al. have demonstrated that vertebroplasty can be safely performed in this setting, even when epidural tumor is seen to contact the spinal cord or nerve roots.15 • Substantial canal narrowing (without neurologic dysfunction) increases the risk that even a small amount of cement leakage will produce neurologic compromise. However, even in the setting of spinal cord deformity and cord signal abnormality on preprocedural MRI, the procedure can be performed with a high rate of pain relief and without adverse neurologic sequelae.16 • Marked loss of vertebral body height (>70% loss of height) makes the procedure more difficult because there may be little space for placement of a cannula. • Poor visualization of osseous structures on fluoroscopy increases the risk of improper needle placement and cement leakage but can be overcome with the use of CT. • Vertebral compression fractures may occur with little or no trauma. • Classic symptoms from an acute vertebral compression fracture include deep pain with sudden onset, midline location, and exacerbation by motion (especially flexion) and standing. Lateral radiation in a dermatomal pattern may be present. • It is important to document failure of conventional medical therapy, which includes pain that is not adequately controlled by bed rest and analgesics, and intolerance to analgesics (e.g., adverse reaction, constipation). The trial of conservative therapy should not exceed 4 to 6 weeks, because pain from compression fractures usually resolves within this time.12 There has been a growing trend toward earlier treatment with vertebral augmentation (within days), especially for patients who require hospitalization and parenteral narcotics.10,12 However, vertebroplasty may be effective in an unhealed fracture even after more than 1 year has elapsed since the onset of pain. • It is important to determine whether the patient is taking anticoagulant medication. Appropriate steps must be taken to ensure adequate coagulation at the time of the procedure. • The classic physical finding is point tenderness at the spinous process of the fractured vertebra, which may be the best predictor of clinical response.10 Localization to a specific level, if possible, is important in targeting treatment in patients who have multiple compression fractures, some of which may be healed and do not require treatment. In difficult cases, examination can be performed with fluoroscopic assistance to localize the pain to a specific anatomic level. • The absence of typical focal tenderness does not preclude the presence of unhealed fractures, and in one study did not result in difference in treatment outcome.17 Atypical manifestations may be seen in Kümmell disease (see later), including radicular pain and pain remote from the fracture.12 • Assessment of lower extremity neurologic function is especially important in patients with symptoms suggestive of myelopathy, radiculopathy, or spinal stenosis. • Preprocedural laboratory screening for infection, coagulopathy, and metabolic abnormality is important. Every patient must have a complete blood cell count, routine coagulation study, and basic metabolic panel performed. • Additional tests such as urinalysis, electrocardiography, chest radiography, or any combination of these tests are left to the discretion of the practitioner and local practice patterns. • Imaging of the spine is undertaken in all cases to confirm the clinical diagnosis, aid in identification of the acute painful fracture, identify potential difficulties, and plan the procedure. • Radiographs can serve as the initial imaging evaluation; when recent previous radiographs are available for comparison, new compression fractures can be identified. In addition, identification of an intraosseous vacuum phenomenon (Kümmell disease) at a particular level points to that level as the probable source of pain and predicts a good response with regard to pain relief and restoration of height.18 • MRI is the test of choice for further evaluation and should be performed in all patients if not contraindicated. The single most useful sequence is a short tau inversion recovery (STIR) or T2-weighted sequence with fat saturation, on which unhealed fractures show hyperintense signal consistent with edema within the bone marrow (Fig. 159-1, A). In the case of retropulsion of fracture fragments, MRI is important in assessing the degree of spinal canal compromise and compression of the spinal cord or nerve roots. With regard to pathologic fractures, MRI is helpful in suggesting the diagnosis, which can be confirmed with intraprocedural bone biopsy, and allows evaluation of epidural extension of tumor. • In patients who cannot undergo MRI (e.g., those with a pacemaker), bone scintigraphy is the test of choice. It allows differentiation of healed and unhealed fractures; unhealed fractures will take up the injected technetium-99m methylene diphosphonate tracer in much higher concentration. Bone scintigraphy highly predicts positive clinical response to vertebral augmentation.19 The major disadvantage is poor spatial resolution, resulting in imprecise localization of the fracture, although single-photon emission computed tomography (SPECT) can be helpful in this regard. Bone scintigraphy with or without SPECT is limited in that it does not evaluate the spinal canal and its contents. • CT is useful for preprocedural evaluation of the integrity of the posterior vertebral body cortex. This question is important in the setting of burst fracture or metastasis, where a fracture through the posterior cortex increases the risk of posterior leakage of cement or posterior displacement of bone or tumor during the procedure.20 The marrow space of the cancellous bone communicates with a network of venous channels. Some of these channels coalesce to form the basivertebral plexus posteriorly in the midline, and others traverse small perforations in the cortex about the remainder of the vertebral body margin. These veins communicate with venous plexuses within the epidural space, neural foramina, and paraspinal tissue,21 which in turn primarily drain into the lumbar and azygos veins. These venous channels are important because they represent possible pathways for extraosseous passage of polymethyl methacrylate (PMMA). Analgesia is necessary for vertebroplasty and kyphoplasty. In the majority of cases, it is achieved with a combination of local analgesics (e.g., lidocaine with bicarbonate, bupivacaine) and moderate sedation (intravenous midazolam and fentanyl). In some cases, general endotracheal anesthesia is required to provide adequate comfort and safety, but having the patient awake is desirable. A conscious patient can give feedback (e.g., increasing pain, neurologic dysfunction) that can alert the operator to potential complications, and neurologic examination during the procedure is possible. Kyphoplasty is performed with general anesthesia at most institutions but can be successfully undertaken with moderate sedation.22,23 In all cases, continuous monitoring is performed with a minimum of electrocardiography, blood pressure measurement, and pulse oximetry. Drug delivery and monitoring are performed by anesthesiologists, nurse anesthetists, or certified nursing personnel. In patients with substantial preexisting respiratory or cardiac disease, an anesthesiologist can be asked to evaluate the patient and determine whether monitored anesthesia care is warranted. The patient should not eat or drink for at least 4 to 6 hours before the procedure. Prone or oblique prone is the ideal patient position for thoracic and lumbar procedures. In addition to the clear advantage of easy access, with proper cushion support, this position maximizes extension of the fractured segments, thereby promoting reduction of kyphosis.24 The patient’s arms should be placed sufficiently toward the head to keep them out of the path of the fluoroscope. Analgesia should be considered before placement on the table, because this part of the procedure may be the most painful. Particular care must be taken when transferring aged or osteoporotic patients to avoid the potential development of new rib or vertebral fractures. Infection risk is minimized with use of standard operating room guidelines for sterile preparation of the skin, draping, operator scrubbing, and use of sterile gowns, masks, and gloves. Antibiotic prophylaxis for these procedures comes in one of two forms. An intravenous antibiotic such as cefazolin (1 g) or clindamycin (600 mg in a patient allergic to penicillin) may be administered during the procedure. Alternatively, the PMMA may be mixed with an antibiotic (e.g., tobramycin 1.2 g) as the cement is being prepared, but many experienced practitioners have abandoned this once popular practice. There are few data to support or oppose antibiotic administration, but spine infections have been reported after these procedures12,25 (see Complications section), and the presence of PMMA makes them difficult to treat successfully.
Vertebroplasty and Kyphoplasty
Indications
Contraindications
Preprocedural Workup
History
Physical Examination
Laboratory Evaluation
Imaging
Technique
Anatomy
Technical Aspects
Sedation
Patient Positioning
Antibiotic Prophylaxis and Skin Preparation
Vertebroplasty and Kyphoplasty