Primary tumor (T)
TX
Primary tumor cannot be assessed
T0
No evidence of primary tumor
Tis
Carcinoma in situ (intraepithelial or intramucosal carcinoma)
T1
Tumor invades into the submucosa
T2
Tumor invades into the muscularis propria
T3
Tumor invades into the subserosa or non-peritonealized pericolorectal fat
T4a
Tumor invades into the visceral peritoneum
T4b
Tumor invades or is adherent to adjacent organs or structures
Regional lymph nodes (N)
NX
Regional lymph nodes cannot be assessed
N0
No regional lymph node metastasis
N1
Metastases in 1–3 regional lymph nodes
N1a
Metastases in 1 regional lymph node
N1b
Metastases in 2–3 regional lymph nodes
N1c
Tumor deposit(s) in the subserosa, mesentery, or non-peritonealized pericolorectal fat without regional lymph node metastasis
N2
Metastases in 4 or more regional lymph nodes
N2a
Metastases in 4–6 regional lymph nodes
N2b
Metastases in 7 or more regional lymph nodes
Distant metastasis (M)
MX
The presence of distant metastasis cannot be assessed
M0
No distant metastasis
M1
Distant metastasis
M1a
Metastasis confined to 1 organ or site (i.e., liver, lung)
M1b
Metastasis in more than one organ or site
19.1.2 T Staging
Accurate T staging requires proper assessment of the depth of tumor invasion. The overall accuracy of T staging is 41–82 % when using CT and 71–91 % when using MRI (Polyps and colon cancer 2008; Kim et al. 2010b).
19.1.2.1 T1 and T2 Staging
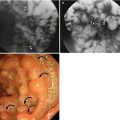
Stage T1 tumors are confined to the mucosa or submucosa and do not show muscularis propria (MP) invasion. T2 tumors have invaded the MP layer without extending into the extracolonic fat. However, distinguishing between T1 and T2 tumors is very challenging, even when using MRI. On CT images, T1 tumors show an intraluminally projected mass without any visible distortion of the colonic wall layer (Dighe et al. 2008). Stage T2 tumors show intraluminal projection, asymmetrical wall thickening with clear adjacent pericolorectal fat, and no disruption of the MP layer (Dighe et al. 2008). When using MRI, partial preservation of the high signal intensity of the submucosal layer beneath the intermediate signal intensity of the tumor on T2-weighted images can be helpful for differentiating between T1 and T2 tumors (Costa-Silva and Brown 2013). In practice, most patients with T1- or T2-stage rectal cancer undergo TME regardless of the specific level of differentiation. However, there are some cases for which accurate differentiation between T1 and T2 is necessary. When surgeons are considering transanal local excision, the high risks associated with surgical resection or a patient’s poor general performance status may make accurate T staging a necessity. Unfortunately, accurate differentiation between T1 and T2 is impossible using CT and difficult even using MRI; therefore, when this type of differentiation is critical, endoscopic ultrasound (EUS) is generally recommended. On EUS, the normal rectal wall consists of alternating hyperechoic and hypoechoic concentric five layers. The first hyperechoic layer represents the interface between the mucosa and lumen. The second hypoechoic layer indicates the muscularis mucosa and the third hyperechoic layer the submucosa. The fourth hypoechoic and fifth hyperechoic layers indicate MP and mesorectal fat, respectively. This more subdivided classification of the rectal wall layer on EUS is helpful to differentiate early-stage rectal cancer. However, the disadvantage of the EUS is operator-dependent, and it takes time to achieve an experienced hand.
19.1.2.2 T3 and T4 Staging
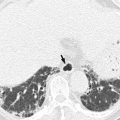
Stage T3 tumors demonstrate invasion into the pericolorectal fat. When using CT and MRI, it is difficult to differentiate between the desmoplastic reactions of a T2 tumor and an early T3 tumor. In the vast majority of cases, fine spicules represent a desmoplastic reaction, whereas extracolonic tumor extension is visible as a thick, nodular band or as broad-based contour bulging (Nougaret et al. 2013). Stage T4a tumors have penetrated the surface of the visceral peritoneum, which covers different sections of the colon and rectum in anatomically different ways. In the ascending and descending colon, only the anterior and lateral aspects are covered by the visceral peritoneum; by contrast, the transverse colon and sigmoid colon are fully covered. In the upper rectum, the anterior and both lateral aspects are covered by the visceral peritoneum, and in the mid-rectum, the anterior aspect is covered by the anterior peritoneal reflection (Nougaret et al. 2013). Stage T4a tumors can be diagnosed by evaluating tumor involvement in this region, but it is difficult to distinguish between T3 and T4a tumors when relying solely on imaging. Fortunately, the currently recommended treatment is the same for both stage T3 and stage T4a tumors. In colon cancer, some types of colectomy will be performed regardless of whether the malignancy is stage T3 or T4a, and in rectal cancer, both T3 and T4a tumors require neoadjuvant CRT. Tumors reach stage T4b when they have invaded adjacent organs such as the liver, adjacent bowel, spleen, pancreas, stomach, prostate gland, seminal vesicle, vagina, uterus, or pelvic side wall.
19.1.2.3 T Staging for Rectal Cancer Involving Anal Sphincter Complex
Occasionally, distal rectal cancer can involve the anal sphincter complex. In these cases, tumor extension into the internal sphincter is considered stage 1 or 2, intersphincteric space stage 3, and the involvement of the external sphincter or adjacent organs around the anal sphincter complex is considered stage 4 (Nougaret et al. 2013).
19.1.3 N Staging
N staging indicates regional lymph node metastasis. The overall accuracy when using CT to identify lymph node metastasis is between 60 and 80 %. The overall accuracy of MRI for N staging has a wide range between 43 and 85 % (Kim et al. 2010b).
19.1.3.1 Pathway of Lymphatic Drainage
In colon cancer, lymph node metastasis follows the pathway of lymphatic drainage along the regional vascular distribution in the mesocolon. The initial lymphatic drainage of colon cancer is primarily into the pericolic lymph node, along the wall of the colon and under the peritoneum. The lymph then drains into the paracolic lymph node along the marginal vessel and follows the regional distributed vessel (i.e., ileocolic or right colic artery), which comprises the intermediate nodal group. Finally, the lymph drains into the principal nodal group at the origin of the superior mesenteric artery (McDaniel et al. 1993). In rectal cancer, the lymph in the upper half of the rectum drains cranially into the inferior mesenteric artery along the superior rectal artery. However, the lymph in the lower half of the rectum usually follows two drainage pathways: cranially via the superior rectal lymphatics to the inferior mesenteric lymph node and laterally via the middle rectal lymphatics to the internal iliac lymph node (Gowdra Halappa et al. 2012). In some instances of far distal rectal cancer, the lymph drains into the inguinal lymph node. Additionally, most nodal spread occurs at the level of the primary tumor or above. Caudal spread below the level of the tumor is very rare except when there is obstruction of the ascending lymphatic pathway.
19.1.3.2 Characterization and Staging of Lymph Node
Cases with one to three metastatic nodes are considered N1, whereas those with four or more are considered N2. Each N stage is further classified into a subgroup, i.e., N1a or N1b, based on the number of metastatic lymph nodes. In rectal cancer, the mesorectal and internal iliac lymph nodes are regarded as regional lymph nodes, even though the internal iliac lymph node is located outside of the TME field. Metastasis to other extra-mesorectal lymph nodes, such as those in the external iliac, inguinal, or common iliac areas, is considered non-regional lymphadenopathy, or M (metastatic) status.
Standard criteria for using CT to identify metastatic lymph nodes have not been established well. The most commonly used criterion is the lymph node size. In case of lymph nodes which are larger than 1 cm in diameter or a cluster of three or more lymph nodes which are smaller than 1 cm, those lymph nodes are considered metastatic (Dighe et al. 2008). However, given that overall 50 % or more of involved lymph nodes are smaller than 5 mm, the size criterion alone is not sufficient. In rectal MRI, the suggested criteria for determining lymph node metastasis consist of the border, shape, internal heterogeneity, and size (Kim et al. 2004). The predictive criteria for identifying metastatic lymph nodes include an indistinct or spiculated border, internal heterogeneous signal intensity, and round shape. Uniform, smooth-bordered lymph nodes with homogeneous signal intensity are regarded as benign. Lymph node size is also a predictive criterion, but there is no cutoff that indicates lymph node involvement (Beets-Tan et al. 2013).
19.1.4 M Staging
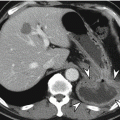
M staging is used to assign the severity of distant metastasis. CRC can be spread by lymphatic, hematogenous, and transperitoneal routes as well as by direct invasion, and M status is classified as M0 or M1 depending on whether distant metastasis is present. There are two subgroups for M1 status: M1a and M1b. M1a indicates that metastasis is confined to a single organ, whereas M1b indicates peritoneal metastasis or metastasis to more than a single organ. The most common distant metastatic organ is the liver, followed by the lungs. Because the venous drainage pathway of the colon and rectum is typically via the portal vein, the most common site of hematogenous metastasis is the liver, followed by the lungs, although metastasis can also reach the brain. However, distal rectal cancer may metastasize directly to the lungs without hepatic metastasis because the middle and inferior rectal veins drain into the inferior vena cava (Gordon and Nivatvongs 2007). Recently, surgical resection has become the preferred treatment, when possible, for the management of hepatic CRC metastasis. Therefore, in addition to accurate detection, radiologists must consider potential surgical perspectives and focus on the precise location, number, and size of hepatic metastasis when using imaging modalities such as gadoxetic acid-enhanced liver MRI.
19.1.5 Circumferential Resection Margin
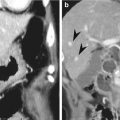
The MRF includes the visceral layer of the endopelvic fascia and represents the TME dissection plane, i.e., the circumferential resection margin (CRM). This fascia is commonly visualized as a fine, low-signal layer on axial or coronal T2-weighted images. Tumor involvement of MRF is an independent negative prognostic factor. A number of studies have demonstrated that MRF involvement is closely related to high local tumor recurrence and poor survival rates. A positive CRM on rectal MRI is indicated when the shortest radial distance between the outermost border of tumor infiltration and the MRF is measured as 1 mm or less. When using 1 mm as the optimal cutoff value for a positive CRM, the classification of CRM using MRI was shown to achieve a 92 % specificity and a 94 % negative predictive value (Beets-Tan and Beets 2011). Additionally, a potentially threatened CRM is defined as a 1–2 mm distance from the margin. The types of tumor infiltration evaluated for CRM measurements include main mesorectal tumor extension, tumor deposits, metastatic mesorectal lymph nodes, and extramural vascular invasion (EMVI) (Nougaret et al. 2013). Meta-analysis of the use of MRI to predict CRM involvement demonstrated a sensitivity ranging from 60 to 88 % and a specificity ranging from 73 to 100 % (Gowdra Halappa et al. 2012).
19.1.6 Extramural Vascular Invasion
EMVI was also shown to be a negative prognostic factor, independent of TNM staging. EMVI is defined as tumor invasion within large vessels in the mesorectum. Positive EMVI is related to local recurrence, metastatic disease, and poor survival, and this invasion can be observed on rectal MR images as the presence of tumor signal intensity within a vascular structure, expanded vessels, and tumoral expansion through and beyond the vessel wall, disrupting the vessel border (Nougaret et al. 2013). The positive predictive value of MRI for EMVI has been calculated with a 62 % sensitivity and an 88 % specificity (Dewhurst and Mortele 2013).
19.1.7 Restaging After Neoadjuvant Chemoradiotherapy
Neoadjuvant CRT followed by TME is the standard of care for patients with locally advanced rectal cancer (T3/T4 or lymph node metastases). Many studies have reported that neoadjuvant CRT decreases the local recurrence rates and increases both the sphincter preservation and disease-free survival rates (Sauer et al. 2004). Of these patients, approximately 10–20 % achieve a pathologic complete response (CR). Although preoperative MRI without neoadjuvant CRT shows relatively high accuracy in determining the T stage or CRM involvement, the accuracy when imaging irradiated cancer remains unsatisfactory. The reported overall accuracy of MRI is approximately 50 % when predicting the T stage of irradiated cancer and 66 % when predicting CRM involvement, and this reduced accuracy has been attributed to the difficulty in differentiating between radiation fibrosis and residual viable tumor (Kim et al. 2010b; Nougaret et al. 2013). The Magnetic Resonance Imaging in Rectal Cancer European Equivalence Study (MERCURY) group reported on the method for assessing the tumor regression grade (TRG) of rectal cancer using conventional high-resolution MRI (Costa-Silva and Brown 2013; Patel et al. 2011). This analysis was based on the proportion of different signal intensities using T2WI, when compared with pre-CRT MRI, given the assumption that low-signal-intensity areas represent fibrosis and intermediate- or high-signal-intensity areas indicate residual viable tumor. Meanwhile, the classification described by Dworak et al. has been widely used as the histopathologic TRG in rectal cancer (Dworak et al. 1997). The histopathologic TRG was graded as follows: grade 0 (no regression) indicates no regression; grade 1 (minimal regression) indicates dominant tumor mass with obvious fibrosis and/or vasculopathy; grade 2 (moderate regression) indicates that the tumor shows dominantly fibrotic changes with few tumor cells or groups (easy to find); grade 3 (near total regression) indicates that the tumor shows very few (difficult to find microscopically) tumor cells in fibrotic tissue with or without mucous substance; and grade 4 (total regression) indicates no tumor cells, only fibrotic mass.
In addition, many studies have demonstrated promising results using diffusion-weighted imaging (DWI) after neoadjuvant CRT to predict a CR, and other studies have suggested that DWI can be used to predict MRF involvement or local tumor recurrence (Kim et al. 2009; Park et al. 2011; Lambregts et al. 2011). In addition, qualitative analyses using DWI and quantitative measurement of apparent diffusion coefficients (ADCs) have been applied (Kim et al. 2011). These studies are based on the notion that residual tumor tissue can be differentiated from radiation fibrosis on high b-value DWI because the residual tumor demonstrates relatively high cellularity and a smaller interstitial space compared to fibrotic tissue. However, these techniques remain controversial for being incorporated into routine sequence and require further validation. Recommendations published after the 2012 European Society of Gastrointestinal and Abdominal Radiology (ESGAR) consensus meeting highlighted the difficulty in reliably differentiating CR from residual tumor using either T2WI or DWI (Beets-Tan et al. 2013). Volumetric analysis before and after neoadjuvant CRT has also been introduced as a means of predicting pathologic tumor response rates (Curvo-Semedo et al. 2011). Studies have reported that the tumor volume reduction after neoadjuvant CRT correlates with the pathologic tumor response rate, and it has also been suggested that an approximately 70–75 % or greater reduction in tumor volume after neoadjuvant CRT indicates a good response or CR (Nougaret et al. 2013). In addition, other studies have demonstrated that post-CRT volumetric analysis on the basis of DWI is superior to T2WI or to qualitative or quantitative analysis of DWI when predicting CR (Ha et al. 2013).
Following neoadjuvant CRT, metastatic lymph nodes may disappear or may be downstaged. Restaging of irradiated lymph nodes can be difficult because characterization of the border, internal heterogeneity, shape, and size may be unfeasible in smaller lymph nodes, and post-radiation changes may also make accurate restaging difficult (van der Paardt et al. 2013). However, interestingly, studies have demonstrated that restaging of irradiated lymph nodes is more accurate than initial nodal staging using pre-CRT MRI. These results might be attributed to the lower prevalence and relatively larger size of non-sterilized lymph nodes after neoadjuvant CRT. The accuracy and negative predictive value of nodal restaging was reported as 67–90 % and 83–93 %, respectively (Beets-Tan 2013).
19.1.8 Common Complications Associated with Colorectal Cancer
CRC can be accompanied with diverse complications such as obstruction, intussusception, ischemia, perforation, and fistula formation with adjacent organ, and primary tumor can be masked by complications. Obstruction is the most common complication associated with colon cancer. Left-sided colonic malignancy is more vulnerable to lumen obstruction than the right because the diameter of left-sided colon is smaller (Kim et al. 2010a). The incidence rate of bowel obstruction associated with colon cancer is 8–29 %. Perforation can occur either at the site of primary tumor due to necrosis or proximal portion of the tumor due to increased luminal pressure. Reported frequency of perforation and abscess formation associated with colon cancer is 2.5–10 % and 0.3–4 %, respectively (Kim et al. 2010a). Although intussusception in adults is very rare, approximately 65 % of cases are associated with neoplasm. On CT image, intussusception can be seen as bowel within the bowel or bowel accompanying with mesenteric fat and vessels within the bowel. Colon cancer as a leading point can be usually found at the tip of the distal intussusceptum, and approximately 30 % of cases cannot be distinguished from edematous bowel.
19.2 Other Rare Types of Colorectal Carcinoma
According to the 2010 WHO classification guidelines, there are a number of histologic variants of colorectal carcinoma. Of these, mucinous adenocarcinoma and signet-ring cell CRC account for 5–15 % and 0.6 % of all primary CRCs, respectively. Histopathologically, mucinous adenocarcinoma is diagnosed when extracellular mucin represents more than 50 % of the total tumor volume, whereas signet-ring cell carcinoma is the appropriate classification when more than 50 % of the tumor cells are signet-ring cells (Tumours of the colon and rectum 2010). Other types of CRC tumors such as small cell carcinoma, squamous cell carcinoma, adenosquamous carcinoma, medullary carcinoma, and undifferentiated carcinoma are extremely rare. Especially, in mucinous adenocarcinoma, intratumoral fine calcifications and low attenuated mucin deposit can be an important clue to diagnose the histologic type on CT examination (Polyps and colon cancer 2008). It is notable that on T2-weighted and diffusion-weighted images, mucinous adenocarcinoma shows a higher signal intensity than the intermediate signal intensity of usual colorectal adenocarcinoma (Kim et al. 2010b). In addition, hepatic metastases from colorectal mucinous adenocarcinoma can be seen as higher-signal-intensity masses on T2-weighted image and mistaken as hepatic cysts.
19.3 Gastrointestinal Stromal Tumor
GISTs are mesenchymal neoplasms originating from the interstitial cells of Cajal which serve as intestinal pacemaker cells of the myenteric plexus and are responsible for controlling intestinal motility. Colorectal GIST is very rare, especially in the colon. In particular, colorectal GIST accounts for 5 % of all gastrointestinal GISTs and for 0.1 % of all neoplasms occurring in the gastrointestinal tract. Approximately 70 % of these tumors demonstrate strong and uniformly positive CD117 (c-kit) expression. GISTs typically occur in populations older than 50 years of age. The clinical presentations of colorectal GIST are nonspecific, vary according to the tumor size, and include abdominal pain, palpable mass, and GI bleeding.
CT plays a role in the evaluation of distant metastasis as well as local invasion in cases of primary GIST. On CT images, small tumors appear as sharply marginated, homogeneously enhancing soft tissue masses in the colorectum. These tumors can also be observed as a subepithelial mass with intact overlying mucosa. Internal homogeneous attenuation can change with tumor growth due to hemorrhage, necrosis, or cystic alterations. Moreover, communication with the bowel lumen can manifest as an air-containing cavitary mass or the formation of a fistula (Lau et al. 2004). When a GIST becomes enlarged, it tends to preserve the smooth border and push adjacent bowel loops or vessels, rather than invading or causing a desmoplastic reaction, and these findings can be helpful for discriminating GISTs from adenocarcinomas or carcinoid tumors (O’Sullivan et al. 2006). Rectal MRI for anorectal GIST can be performed to accurately delineate the malignancy’s relationship with adjacent structures. For example, the solid portion of a tumor is typically visible as low to intermediate signal intensity on T1WI, intermediate to high signal intensity on T2WI, and moderate to intense enhancement. However, the signal intensity of the hemorrhagic portion of the tumor can vary when using MRI. Although the majority of GISTs are benign (60–70 %), they can also be malignant or potentially malignant (O’Sullivan et al. 2006). Therefore, risk assessment is based on the tumor size and mitotic index (the number of mitoses on 50 high-power fields). A malignant GIST can be spread via a hematogenous route or via peritoneal seeding, and the common sites to which a GIST may spread include the liver, omentum, and peritoneum. In contrast, lung and lymph node metastases are very rare. The rarity of lymph node metastases, and even the presence of bulky and large tumors, may be important clues for properly differentiating GISTs from adenocarcinomas or lymphomas (Lau et al. 2004).
19.4 Lymphoma
Colorectal lymphoma can occur either as a primary lesion or as colorectal involvement of systemic disease. In fact, the gastrointestinal tract is the most common site of extranodal non-Hodgkin’s lymphoma. Lymphoma with colorectal involvement constitutes 10–20 % of all gastrointestinal lymphomas, although primary colorectal lymphoma is very rare, accounting for 0.2–0.6 % of all malignant colorectal tumors (Chang and Menias 2013; Dionigi et al. 2007). The most frequent site of colorectal lymphoma is the cecum (57 %), due to the relative abundance of lymphoid tissue, followed by the ascending colon (18 %), the transverse and rectosigmoid colon (10 %), and the descending colon (5 %) (Dionigi et al. 2007). The majority of colorectal lymphomas include non-Hodgkin’s lymphoma, B-cell lineage lymphoma, and, most commonly, a histologic subtype of diffuse large B-cell lymphoma. These tumors frequently occur in patients who are between 50 and 70 years of age. The presenting clinical symptoms include abdominal pain, anorexia and weight loss, a palpable mass, a change in bowel habits, and bloody stool. Moreover, the radiographic findings of colorectal lymphoma can vary according to the histologic subtype. The imaging features may include a localized lesion which can be seen as a polypoid mass, frequently near the IC valve; annular circumferential infiltration with a smooth surface or extensive ulceration; a cavitary mass; a focal mucosal nodularity; or mucosal fold thickening (Lee et al. 2002; Quayle and Lowney 2006). Radiographic imaging can occasionally be used to classify a lesion as diffuse, ulcerative, or nodular, and the most common manifestation of primary colorectal lymphoma is a localized, bulky, polypoid mass. Although annular circumferential infiltration can cause severe luminal narrowing, colonic obstruction is rare due to lack of a desmoplastic reaction and weakening of the muscularis propria in the colon wall, resulting from submucosal lymphoid infiltration (Lee et al. 2002). Imaging findings indicating colorectal lymphoma can overlap with those of other colorectal diseases. For example, peripheral T-cell colorectal lymphoma mimics inflammatory bowel disease (Lee et al. 2001); the diffuse nodular forms of colorectal lymphoma, also known as lymphomatous polyposis, which can be observed in cases of mantle cell lymphoma or low-grade B-cell lymphoma arising from the mucosa-associated lymphoid tissue, can mimic familial adenomatous polyposis; and pseudopolyps can also be associated with inflammatory bowel disease (Lee et al. 2002). However, particular imaging findings, such as homogeneous enhancement, aneurysmal dilatation, extensive lymphadenopathy, and a relatively well-demarcated lesion with a preserved fat plane and no invasion into adjacent structures, can be helpful for differentiating localized forms of colorectal lymphoma (Ghai et al. 2007).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree