3 Mediastinum and Thymus
Introduction
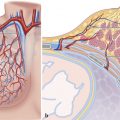
The mediastinum (Latin, media = middle and stare = to stand; i.e., that which stands in the middle) is a compartment within the thorax which contains all the thoracic organs, apart from the lungs and pleura. It is bounded superiorly by the thoracic inlet and inferiorly by the diaphragm. The sternum and ribs form the anterior-most margin of the mediastinum and the vertebral column forms the posterior margin. Laterally, it is bounded by the mediastinal pleura on both sides. The mediastinum is a complex area containing structures vital to life, both large and small, such as the heart, major vessels, trachea, esophagus, nerves, and lymph nodes. Fat also makes up a large component of the mediastinum. In this chapter, the anatomy of above structures will be reviewed. Azygous system, systemic veins, aorta, and pulmonary arteries are discussed in related chapters.
Compartmentalized Anatomy
It is useful to divide the mediastinum into compartments for the purposes of forming a differential diagnosis when dealing with masses in the mediastinum. However, the mediastinum lacks true anatomical boundaries to serve as a basis for compartmentalized anatomy. As a result, several differing models have been developed in attempts to compartmentalize the mediastinum. 1 , 2 Each model is different from the next, and some are based on plain chest radiography. As may be expected, there is discordance between the different models.
Traditionally and historically, radiologists have mostly used systems which divided the mediastinum into anterior, middle, posterior, and sometimes superior mediastinal compartments, based on chest radiograph. More recently formalized systems based on cross-sectional computed tomography (CT) imaging and expert consensus have been proposed. Initially a four-compartment model (anterior, middle, posterior, and superior mediastinum) by the Japanese Association for Research of the Thymus (JART) and then a three-compartment model by the International Thymic Malignancy Interest Group (ITMIG) have been introduced. 3 It should be emphasized; these models and all the various historical models are based on expert opinion and preferences, as there are no real anatomical planes to separate the “anterior,” “middle,” and “posterior” mediastinum. These compartments exist to optimize the clinician’s ability to identify and distinguish pathology based on location. 4 , 5 We will further discuss the ITMIG classification schema which will likely become the most widely accepted system.
ITMIG Classification
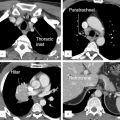
The ITMIG adopted the following system for compartmentalization of the mediastinum (Fig. 3‑1 , Fig. 3‑2 , Fig. 3‑3). ITMIG divides the mediastinum into three compartments: (a) prevascular (anterior mediastinum), (b) visceral (middle mediastinum), and (c) paravertebral (posterior mediastinum) compartments (Table 3‑1).



The prevascular compartment (anterior mediastinum) is bounded by the thoracic inlet superiorly, the diaphragm inferiorly, the sternum anteriorly, the anterior aspect of the pericardium posteriorly, and the mediastinal pleura laterally. The major components of this compartment are the thymus, lymph nodes, left brachiocephalic vein, and fat. The prevascular compartment extends from the thoracic inlet to the diaphragm, always anterior to the heart and pericardium. Displacement of these structures posteriorly is a useful tool to localizing any mass of questionable origin to the prevascular compartment. Common pathologies include thymic lesions, teratoma, lymph node diseases, and hematoma. The prevascular compartment is a common location for collection of hematoma after trauma or surgery.
The visceral compartment (middle mediastinum) is bounded by the thoracic inlet superiorly, the diaphragm inferiorly, and the anterior pericardium anteriorly. The posterior margin is defined by a line connecting points on each vertebral body 1 cm posterior to the anterior cortex of the vertebral bodies, the so-called “visceral–paravertebral compartment boundary line.” The major components of this compartment are the pericardium, heart, superior vena cava, ascending thoracic aorta, aortic arch, descending thoracic aorta, proximal (intrapericardial) pulmonary arteries, thoracic duct, trachea/carina, esophagus, nerves, and lymph nodes. 6 , 7
The paravertebral compartment (posterior mediastinum) is bounded by the thoracic inlet superiorly, the diaphragm inferiorly, the visceral compartment anteriorly (“visceral–paravertebral compartment boundary line” which is defined in the preceding paragraph) and a vertical line along the posterior margin of the chest wall and the lateral margin of vertebral transverse processes posteriorly. The major structures of this compartment are the thoracic spine, paravertebral soft tissues/fat, and autonomic nervous plexus. Enlarged lymph nodes can be found anywhere in the mediastinum. Common focal pathologies include nerve/nerve sheath masses (i.e., neurofibroma, schwannoma) and tumors of the sympathetic chain such as paraganglionic cell neoplasms. Other uncommon pathologies extramedullary hematopoiesis (i.e., thalassemia), neurenteric cysts, and rarely meningoceles.
Components
Ligaments
The contents of the mediastinum are suspended/anchored to the chest wall or connected one another via a series of ligaments. While many of them are quite small, several of these ligamentous connections are large enough to be appreciated by CT or magnetic resonance imaging (MRI).
Inferior Pulmonary Ligament
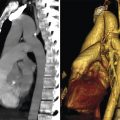
The inferior pulmonary ligament (IPL) is a double layer of pleura which becomes continuous with intervening connective tissue to form a contiguous ligament (Fig. 3‑4) (see Chapter 4, Pleura). Specifically, at the level of the pulmonary hilum both the anterior and posterior pleura come together and encase the hilum. Just below the pulmonary vein, artery, and bronchus the two pleura (along with intervening veins and lymphatics) fuse together to form the IPL. 8 , 9 The IPL, which takes a triangular shape at the hilar level, travels caudally along the mediastinal pleural surface to either terminate at the diaphragmatic pleura or end as a free edge. At axial CT, it is most commonly seen as a well-defined linear opacity extending horizontally from the paravertebral (posterior) mediastinal pleural surface. 10 It is best seen at the level of the diaphragm, where a slight widening of its base (in order to contact the diaphragm) makes it more apparent (Fig. 3‑5 , Fig. 3‑6).



While the IPL is an extraparenchymal structure, yet it can be surrounded by lung parenchyma especially in emphysematous lungs, giving it the appearance of intraparenchymal subsegmental atelectasis or scarring. The IPL has clinical significance for lung cancer staging, as lymphadenopathy in the IPL is considered a station 9 lymph node. IPL functions to anchor the lower lobe to both the mediastinum and the diaphragm. In patients with disruption of the IPL, there is a reported increased incidence of lung torsion, thought to be due to increased mobility of the lower lobe. IPL may be injured in trauma, and posttraumatic pneumatocele especially in children can be observed at this anatomical area.
Ligamentum Arteriosum
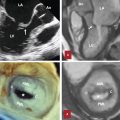
Ligamentum arteriosum (LA) is a small ligament which is a remnant of the fetal ductus arteriosus. The LA is seen as a calcified structure in nearly 50% of adults near the aortic arch passing into the aortic–pulmonary window. 11 A calcified LA is considered an insignificant normal finding; however, it is many times mistaken for aortic atherosclerotic disease 12 (Fig. 3‑7). The calcification may be a single focus in the setting of a noncalcified tubular structure, a linear or curvilinear calcification, or large clumped calcifications. The LA is a notable piece of anatomy as it is theorized that a fixed tethering of the aorta by this ligament accounts for a large proportion of aortic injuries in the area of the LA. It should be remembered that the LA serves as an important landmark/border when distinguishing aortopulmonary (AP) window (station 5) lymph nodes from left paratracheal (station 4L) lymph nodes, the former being to the left of the LA.

Sternopericardial Ligaments
There are both superior and inferior sternopericardial ligaments (SPLs) which attach the fibrous pericardium to the posterior surface of the sternum. The superior SPL attaches the pericardium to the manubrium, while the inferior SPL attaches to the xiphoid process (Fig. 3‑8).

Pericardiophrenic or Phrenopericardial Ligament
The pericardiophrenic ligament (PPL) is a triangular structure which is formed by the combination of pleural reflection, right phrenic nerve, and pericardiophrenic bundle vessels. At CT, it has a very similar appearance to the IPL, however, the PPL has a characteristic position either lateral or anterior to the inferior vena cava (IVC) (Fig. 3‑9). Sometimes, the phrenic nerve and/or pericardiophrenic bundle can be seen as distinct structures entering the PC.

Thyrothymic Ligament
The thyrothymic ligament connects the thyroid to the upper parts of the thymus on either side of the neck and mediastinum (Fig. 3‑10). The inferior extents of these ligaments pass into the mediastinum as they connect to the thymus. They serve as an important anatomical landmark during neck dissection when searching for the parathyroid glands. In cases of mediastinal parathyroid adenoma, many times the adenoma is located along this ligament.

Thymus
The thymus is relatively centrally located in the prevascular compartment of the mediastinum (Fig. 3‑10 , Fig. 3‑11). It resides between the sternum anteriorly and the pericardium, aortic arch, and left brachiocephalic vein posteriorly. 13 , 14 Laterally the thymus is bordered by the mediastinal pleural surfaces.
While this is the classic normal location of the thymus, it can also be located ectopically in the neck. This is in part the result of thymic development, in which the normal thymus descends from the third and possibly fourth pharyngeal pouches down the neck and into the mediastinum. Any arrest of this pathway will result in ectopic thymic tissue. In 20 to 25% of people, separate islands of residual thymic tissue can be found in the neck, mediastinum, and lung as a result of the thymic development process.
The thymus grows during fetal development, through birth into puberty where it achieves its largest relative size (Fig. 3‑12). After puberty, a process of involution begins and the thymus diminishes in size as age increases 15 , 16 (Fig. 3‑12). This normal involution is known to accelerate with exogenous steroid use. The thymus has a prime role in the peripheral immune system, and the age-related normal involution is thought to be related to weaker overall immune systems in the elderly. Why this occurs remains a mystery. 17
The thymus is comprised of two distinct lobes which are connected centrally by loose connective tissue or an intermediate lobe (Fig. 3‑12 , Fig. 3‑13). A capsule made of collagenous fibers encases both of these lobes. Capsular septa separate the lobes into smaller cortical lobules, with the deeper medullary space remaining as one continuous structure. 18 , 19 These lobules and lobes can be appreciated on CT scans readily as a normal finding which distinguishes the thymus from normal and abnormal adjacent structures.
The thymus may also become hyperplastic in patients. When the body is under stress, the thymus may actually shrink with a rebound enlargement after removal of stress. This phenomenon is most common in pediatric and adolescent patients after periods of stress (burns, surgery, infection, steroids) but is also known to happen commonly in adults after chemotherapy or radiotherapy and has also been reported with steroid use, after surgery, and with infection (Fig. 3‑13). Common pathologies of the thymus include thymic cyst and thymoma (Fig. 3‑13). Thymoma usually presents as a well-defined mass in the prevascular mediastinum. It should be differentiated from other common pathologies in this region such as teratoma or lymphoma. Myasthenia gravis is a distinct clinical presentation of the thymoma and thymic hyperplasia.
The thymus maintains several connections to adjacent structures via ligaments and direct connections to maintain a relatively suspended position in the prevascular space of the mediastinum. 19 Superiorly, a thyrothymic ligament connects directly to the inferior thyroid (Fig. 3‑10). Posteriorly, a layer of fascia maintains a connection to the brachiocephalic veins and aorta/great vessels. Inferiorly, connective tissue maintains a connection with the pericardium, a connection which persists from early development.
When discussing thymic arterial or venous anatomy, it is always important to remember that considerable variation exists. 20 , 21 Thymic arterial blood supply arrives via the superior, lateral, and posterior thymic arteries. The ultimate origin of these arteries is variable; arising from the inferior thyroid artery, internal thoracic artery, pericardiophrenic artery, or anterior intercostal arteries. The superior thymic artery usually arises from the inferior thyroidal artery or middle thyroid artery. The lateral thymic arteries are usually branches of the right and sometimes left internal thoracic artery. When present, the posterior thymic arteries are direct branches of the brachiocephalic artery and aorta. Venous drainage is accomplished by a venous plexus predominating on the posterior side of the thymus (Fig. 3‑14). This plexus is drained by two small veins on the posterior side of each thymic lobe. Finally, these smaller veins join to form a larger single thymic vein (sometimes referred to as the great vein of Keynes). 20 This thymic vein ultimately drains into the left brachiocephalic vein. Variant anatomy exists where separate thymic veins drain into the left inferior thyroidal vein, internal thoracic vein, or superior vena cava.




Mediastinal Fat
Mediastinal (pericardial) fat is generally unencapsulated fat which encases the various mediastinal structures without compression or displacement, a characteristic trait of benign mediastinal fat. It should not be confused with the epicardial (subepicardial) fat which is located between the visceral layer of the pericardium and the myocardium (Fig. 3‑15). 22 , 23 Mediastinal fat predominates in the prevascular compartment (i.e., anterior mediastinum). The epicardial adipose tissue is supplied by branches of the coronary arteries, whereas the mediastinal fat is supplied from different sources including the superior pericardial, middle pericardial, and pericardiophrenic arteries, branches of the internal mammary artery. 23 When the amount of fat is abnormally increased, it is usually referred to as mediastinal lipomatosis which is commonly due to obesity, diabetes, and exogenous steroid use (Fig. 3‑16). Excess fat in the mediastinum can influence patency of the vessels. 24


Lymph Nodes
Thoracic mediastinal lymph nodes serve to filter lymph through a series of lymphatic vessels. Lymph nodes play a vital role in filtering infections and debris before entering the bloodstream, but they also act as a conduit to collect malignant cells. In this regard, they serve as an important prognosticator when evaluating lung and other types of cancers.
The pattern of mediastinal lymphadenopathy has become an integral part of lung cancer staging. Chief among the staging systems is the clinical TNM (cTNM) staging system which is typically used before any treatment regimen for lung cancer is begun. This system attempts to reliably stage a patient’s cancer based on the extent of disease with local, regional, or distant lymph node involvement as a key determinant of disease stage. Several standardized nomenclature systems have been developed; however, the one adopted by the International Association for the Study of Lung Cancer (IASLC) appears to be the most widely used 25 (Fig. 3‑17 , Fig. 3‑18 , Fig. 3‑19). Lymph node stations of the mediastinum are classified in Table 3‑2.
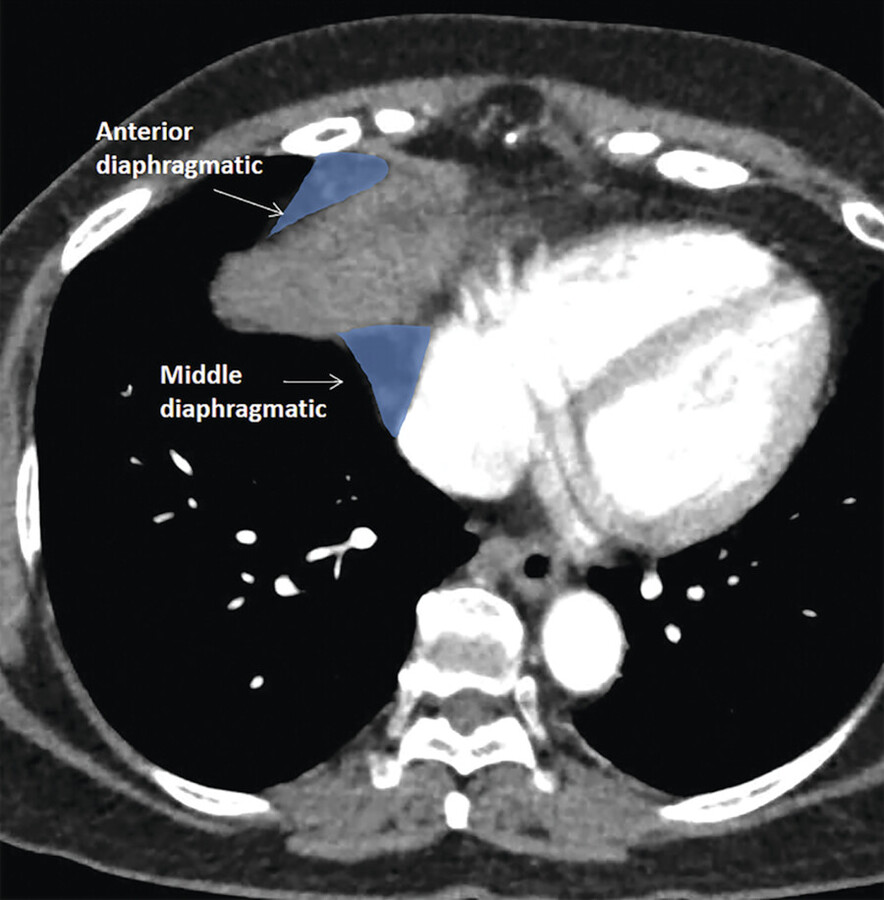
However, care must be taken when localizing lymph nodes, as not all the mediastinal lymph nodes are classified under the IASLC lymph node map. Lymph node groups that have not been explicitly classified include diaphragmatic lymph nodes, which appear around the diaphragm; some authors may consider these to be prevascular lymph nodes as they are anterior to the pericardium (see Chapter 10, Diaphragm) (Fig. 3‑20). Intercostal lymph nodes and internal mammary artery lymph nodes are very close and appear contiguous with the mediastinum but would be considered chest wall structures. 26
Mediastinal lymph nodes primarily drain lymph from the lungs and pulmonary hila in certain patterns. While we will review some of the most common pathways for lymph node drainage, it should be noted that considerable variability exists between many of studies done to date on this topic. 27 , 28
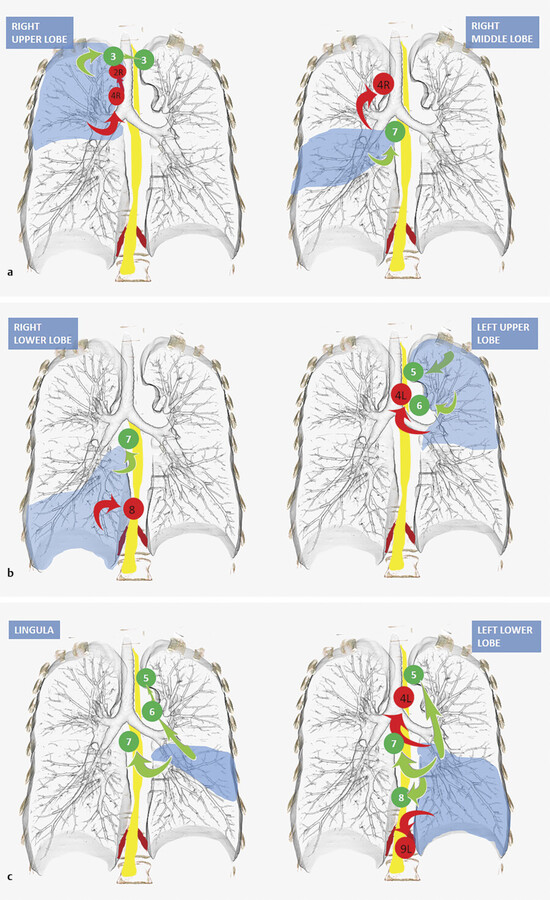
With that said, the right upper lobe tends to drain to the right paratracheal station (level 4R) and prevascular (level 3A) lymph nodes, both of which are above the level of the heart (Fig. 3‑20). For example, isolated subcarinal lymphadenopathy is much less common in right upper lobe cancers. From the 4R level, lymph tends to drain into the high right paratracheal (2R) and then scalene/supraclavicular lymph nodes. The right middle lobe, however, most frequently drains to the subcarinal station (level 7) followed by the lower right paratracheal station (level 4R). The right lower lobe is most commonly seen draining to the subcarinal (level 7) and paraesophageal (level 8) stations. 29
The left upper lobe malignancies seem to drain to the subaortic and para-aortic lymph nodes (level 5 and 6) and left low paratracheal (4L). Note should be made that the lingula actually drains to the subcarinal lymph nodes first, then to the subaortic and para-aortic stations.
Finally, the left lower lobe tends to drain to the subcarinal station 7 followed by subaortic, 5 left paratracheal (4L), paraesophageal, 8 and inferior pulmonary ligament 9 lymph nodes. The left lower lobe is notable as it tends to drain to contralateral mediastinal lymph nodes more frequently than any other lobe (Fig. 3‑20).



The mediastinal lymph nodes receive vessels from the lungs, thymus, thyroid, esophagus, diaphragm, liver, pericardium, and heart.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree