4 Management of Complications of Portal Hypertension: Transjugular Intrahepatic Portosystemic Shunt (TIPS)
Anthony M. Esparaz and Nicholas Fidelman
4.1 Introduction
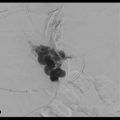
In the more than 20 years of clinical research that has been performed since the transjugular intrahepatic portosystemic shunt (TIPS) intervention was introduced, multiple indications, contraindications, and technical standards for TIPS placement have been described. 1 Originally described by Rösch et al in 1969, 2 TIPS acts as a side-to-side portacaval shunt to decrease the portosystemic pressure gradient by diverting blood from the portal to the systemic circulation. 3 Hepatic sinusoidal pressure is lowered and circulatory flow is increased, decreasing the effects of portal hypertension. 4 TIPS placement has become an established procedure for the treatment of complications related to portal hypertension, including variceal bleeding and ascites. 5 , 6
A number of factors have influenced the current application of TIPS, including multiple clinical trials exploring procedural safety and efficacy, as well as the establishment of the Model of End-Stage Liver Disease (MELD) scoring system for assessment of short-term mortality in patients undergoing TIPS placement. 3 Furthermore, long-term shunt patency has markedly improved since the introduction of polytetrafluoroethylene (PTFE)-covered stents. 7
In this chapter, common indications and contraindications for TIPS are discussed. The standard technique for stepwise TIPS placement is described, as well as common complications and the management of these events. The role of pediatric TIPS and TIPS in unconventional situations such as portal vein occlusion, hepatic vein occlusion, and split-liver transplantation is discussed in a separate chapter.
4.2 History of TIPS
The TIPS procedure was initially described by Rösch et al in 1969. 2 In this study, the authors used a silicone-coated spring coil to create portosystemic shunts in dogs; these shunts remained patent for up to 2 weeks. However, the technique only became reproducible in humans with the later development of endovascular stents. 4 In 1982, Colapinto et al 8 and Gordon et al 9 were the first to create TIPS in 20 patients with variceal bleeding, but long-term results were poor, as most patients had rebleeding and 9 died within 1 month of the procedure. In the mid-1980s, Palmaz et al 10 , 11 developed expandable metallic stents and demonstrated their long-term patency in cirrhotic livers of dogs. This led to the implantation of metallic Palmaz stents in human patients at the University of Freiburg in 1988. 1 In the following years, numerous randomized studies, consensus conferences, and clinical efforts led to a vast improvement in the TIPS technique, as well as delineation of the most appropriate indications and patient selection for TIPS placement. 12 , 13 , 14
Multiple mechanisms are responsible for shunt dysfunction and occlusion, including acute intrashunt thrombosis, pseudointimal hyperplasia from iatrogenic bile leaks into the shunt, and intimal hyperplasia in the outflow hepatic vein. 4 Several experimental and clinical trials assessed the use of covered stent grafts in achieving long-term shunt patency. 15 , 16 , 17 , 18 Ultimately, the best results were realized with the use of stents covered with PTFE. 18 , 19 , 20 , 21 , 22 More recent comparisons of covered stents with bare metal stents have shown decreased intimal hyperplasia and prolonged shunt patency with expanded PTFE (ePTFE)-covered stents. 7 , 23 , 24 , 25
Steady adaptation and incorporation of these results, combined with increasing clinical experience and improved technical facilities, have led to the rapidly increasing acceptance of TIPS placement as an established procedure for the treatment of complications related to portal hypertension.
4.3 Indications
By diverting blood flow from the portal to systemic circulation, TIPS placement results in decreased hepatic sinusoidal pressure and increased circulatory blood flow, successfully reducing the portosystemic pressure in more than 90% of cases. 26 , 27 , 28 TIPS efficacy has been established for the secondary prevention of variceal bleeding and the treatment of refractory ascites, as evidenced by multiple randomized controlled trials (RCTs) and meta-analyses of RCTs. 3 Typical indications for TIPS are summarized in ▶ Table 4.1. 3
4.3.1 TIPS for the Secondary Prevention of Variceal Bleeding
The strongest evidence for the efficacy of TIPS placement exists for the secondary prophylaxis of variceal bleeding. 3 , 29 Since 1994, a total of 13 RCTs assessing TIPS for this indication have been published, depicting results for 948 patients, 472 of whom underwent TIPS placement. 26 , 27 , 28 , 30 , 31 , 32 , 33 , 34 , 35 , 36 , 37 , 38 , 39 Recent meta-analyses by Burroughs et al 40 and Zheng et al 41 highlighted a greater than threefold decrease in the risk of recurrent variceal bleeding after TIPS placement compared with various forms of endoscopic therapy. Rates of rebleeding after TIPS ranged from 9 to 40.6%, whereas continued endoscopic treatment demonstrated rebleeding rates of 20.5 to 60.6%. All-cause mortality was similar in the two groups, but there was a greater than twofold increase in the rate of development of hepatic encephalopathy (HE) after TIPS placement. 26 , 27 , 28 , 30 , 31 , 32 , 33 , 34 , 35 , 36 , 37 , 38 , 39
4.3.2 TIPS for the Treatment of Refractory Ascites
A diagnosis of refractory ascites portends a poor prognosis in patients with liver cirrhosis because of the risk of complications (e.g., spontaneous bacterial peritonitis, dilutional hyponatremia, hepatorenal syndrome); this condition is associated with 1-year survival of less than 50%. 4 Despite this high mortality rate, patients with refractory ascites tend to have paradoxically low MELD scores, thus conferring a lower position on the waiting list for a liver transplant. 42
Although large-volume paracentesis has been shown to be safe and easy to perform for the treatment of refractory ascites, it negatively affects systemic hemodynamics and renal function, limiting its use as a long-term treatment option. 43 TIPS placement, on the other hand, improves renal function and system hemodynamics while treating ascites. In a 2010 review, Rössle and Gerbes 44 found that within 4 weeks of TIPS placement, urinary sodium excretion and serum creatinine levels improved significantly, and within 4 to 6 months, there was an associated increase in serum sodium concentration, glomerular filtration rate, and urinary volume with normalization of plasma renin, aldosterone, and noradrenaline concentrations. These findings suggest that TIPS placement reverses hyperdynamic circulation and amends central underfilling.
A recent analysis that included 16 studies of TIPS placement for refractory ascites demonstrated a complete response in 51% of patients and a complete or partial response not requiring paracenteses in 68%. 45 Furthermore, a meta-analysis by D′Amico et al 46 assessing results from five earlier RCTs demonstrated a 7.1-fold decrease in the risk of recurrent tense ascites after TIPS placement, along with rates of improvement of 38 to 84% with TIPS versus 0 to 43% with large-volume paracentesis. Although four earlier RCTs found no significant difference in survival between TIPS and large-volume paracentesis, 47 , 48 , 49 , 50 two of the more recent studies demonstrated improved survival in patients who underwent TIPS placement compared to patients treated with large-volume paracentesis. 51 , 52 Finally, in a meta-analysis of four studies, 48 , 49 , 50 , 51 Salerno et al 53 observed a recurrence of tense ascites in 42% of patients who underwent TIPS placement versus 89% of those who underwent paracentesis (p < 0.0001), with an associated decrease in the number of paracenteses per patient in those with TIPS (1.6 ± 3.5 vs. 7.1 ± 8.8; p < 0.0001). The overall rate of complications related to portal hypertension (gastrointestinal bleed, hepatorenal syndrome, spontaneous bacterial peritonitis) was also significantly lower in the TIPS group than in the paracentesis group (15 vs. 28%, p = 0.005).
4.3.3 TIPS for the Treatment of Acute Variceal Bleeding
When variceal bleeding occurs, endoscopic and/or pharmacologic therapies are initially used. 29 For cases in which bleeding is not controlled by conventional methods, TIPS has been used for rescue treatment with good results.
Two RCTs 54 , 55 and a retrospective surveillance study 56 compared TIPS with medical therapy for acute variceal bleeding. In 2004, Monescillo et al 54 randomized high-risk patients with a measured portosystemic pressure gradient of more than 20 mm Hg within 24 hours of admission to receive either early barestent TIPS (n = 26) or medical treatment (n = 26). Early TIPS placement reduced treatment failure (p = 0.003) and in-hospital and 1-year mortality (p < 0.05).
In 2010, García-Pagán et al 55 randomly assigned 63 high-risk cirrhotic patients with acute variceal bleeding at index endoscopy within 24 hours after admission to receive either a covered-stent TIPS (n = 32) or medical therapy including propranolol or nadolol plus a nitrate followed by endoscopic band ligation if drugs were not effective (n = 31). During the median 16-month follow-up, the TIPS group demonstrated a significantly lower rate of rebleeding or failure to control bleeding (3 vs. 45%, p = 0.001). Moreover, actuarial survival at 1 year was significantly better in the TIPS group (87.5 vs. 61.3%, p < 0.001). These results were corroborated by a post-RCT surveillance study by the same authors, 56 who concluded that early use of TIPS in hospitalized patients with acute variceal bleeding was associated with a significant improvement in rates of failure to control bleeding or rebleeding (7 vs. 47%, p < 0.001).
4.3.4 TIPS for the Treatment of Hepatic Hydrothorax
Hepatic hydrothorax occurs when there is direct passage of peritoneal ascitic fluid into the pleural cavity via diaphragmatic leaks. This condition is defined as a significant pleural effusion, usually more than 500 mL, in a cirrhotic patient without primary cardiac or pulmonary disease. 3 Several retrospective case series have described the use of TIPS for hepatic hydrothorax in more than 150 patients. 57 , 58 , 59 , 60 At least, partial improvement in clinical symptoms (dyspnea, rate of thoracenteses) was reported in 68 to 82% of patients, and complete resolution of hepatic hydrothorax was reported in 57 to 71% of patients. 57 , 58 , 59 , 60 One-year survival rates, as described in two studies, 57 , 58 were 64 and 48%. While TIPS placement was associated with strong long-term survival and response rates in these studies, it is also noteworthy that TIPS is the only therapeutic option that concurrently treats refractory ascites and hepatic hydrothorax.
4.4 Patient Selection and Contraindications
A dynamic collaboration must take place among hepatologists, gastroenterologists, interventional radiologists, and the patients themselves when deciding on TIPS placement as the most appropriate treatment for complications of portal hypertension. Absolute and relative contraindications to TIPS placement are listed in ▶ Table 4.2. 3 Generally speaking, absolute contraindications include congestive heart failure, severe tricuspid regurgitation, and severe pulmonary arterial hypertension (with mean pulmonary pressure >45 mm Hg). Relative contraindications include HE (which may worsen after TIPS placement), active sepsis, and anatomical conditions that may complicate creation of the shunt, such as chronic organized portal vein thrombosis, hepatic vein thrombosis, polycystic liver disease, and multiple hepatic masses. 61 , 62
Absolute contraindications | Relative contraindications |
Primary prevention of variceal bleeding | Hepatocellular carcinoma (especially if central) |
Congestive heart failure | Obstruction of all hepatic veins |
Severe tricuspid regurgitation | Portal vein thrombosis |
Severe pulmonary hypertension | Moderate pulmonary hypertension |
Multiple hepatic cysts | Severe uncorrectable coagulopathy (INR > 5) |
Uncontrolled systemic infection or sepsis | Uncorrectable thrombocytopenia of < 20,000 cells/cm3 |
Unrelieved biliary obstruction | Hepatic encephalopathy |
Abbreviation: INR, international normalized ratio. | |
Multiple studies have focused on models to predict survival after TIPS placement. 63 , 64 , 65 , 66 Child–Pugh class C disease, 63 variceal hemorrhage requiring emergent TIPS placement, alanine aminotransferase level greater than 100 IU/L, serum bilirubin level greater than 3 mg/dL, and pre-TIPS HE unrelated to bleeding 64 are all associated with an increased risk of post-TIPS mortality. A MELD score higher than 18 predicts a significantly increased 3-month mortality rate after TIPS placement (85 vs. 55%, p = 0.037), compared with MELD scores less than or equal to 18. 42 , 67 , 68
Irrespective of the predictors considered, the risks and benefits associated with TIPS creation must be balanced in the context of each patient′s clinical scenario. There may be situations in which the added risks of a difficult TIPS procedure are justified (e.g., in patients with hepatocellular carcinoma and refractory variceal bleeding). Predictors can only be used to inform the treating team and the patient about expected outcomes and must always be weighed against the urgency of treatment escalation. 1
4.5 Preparation for TIPS Placement
As with any procedure, TIPS placement is associated with notable risks that must be weighed against the clinical benefits for the individual patient. A thorough understanding of the patient′s history and a detailed physical examination are required. Additionally, laboratory results obtained within 24 hours of the procedure should be reviewed, including complete blood count, coagulation panel, and metabolic panel, to evaluate liver and renal function. 3 Coagulopathy (international normalized ratio [INR] for prothrombin time > 1.5), thrombocytopenia (platelet count < 50,000 cells/mL), and anemia (hematocrit < 25%) should be corrected before initiation of the procedure. Patients with renal insufficiency should receive hydration.
Preprocedural cross-sectional liver imaging must be performed shortly before TIPS placement. 69 Thorough preprocedural planning requires assessment of the size and configuration of the liver, including the patency and anatomical course of the hepatic venous structures in relation to the portal venous structures. In an emergent situation, bedside sonographic liver evaluation is required at the very least to demonstrate hepatic and portal vein patency. In those patients with known cardiac or pulmonary disease, an echocardiogram should be obtained to exclude pulmonary arterial hypertension. 3
Finally, TIPS placement carries a risk for periprocedural infection or sepsis, including the possibility of infection in the stent itself. Nevertheless, little published evidence exists to support the use of prophylactic antibiotics before TIPS placement. 69 In patients who do not receive prophylactic antibiotics, infection rates range from 3 to 16%. 70 Prophylactic administration of ceftriaxone has been suggested by some authors. 71 Additionally, antibiotic administration may be considered to prevent infection of the stent lumen, termed “endotipsitis”; this condition has a published prevalence of 1.3%. 1 , 72 , 73 Overall, acute infection secondary to TIPS placement appears to be infrequent, leaving the argument regarding prophylactic antibiotics unresolved. It is likely that improved technical skills combined with increasing global clinical experience (e.g., fewer catheter exchanges) have lowered the incidence of postprocedure infection. Nevertheless, one must consider the generally poor clinical status and tolerance of infection in patients requiring TIPS when the decision regarding prophylactic antibiotic use is made.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree