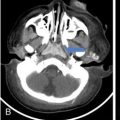
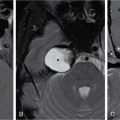
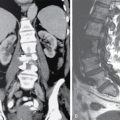
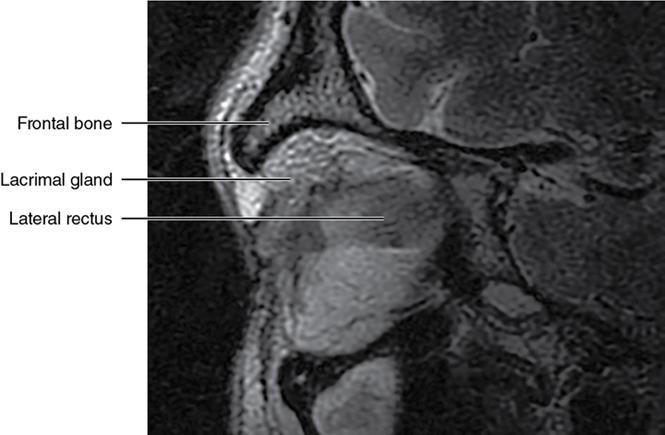
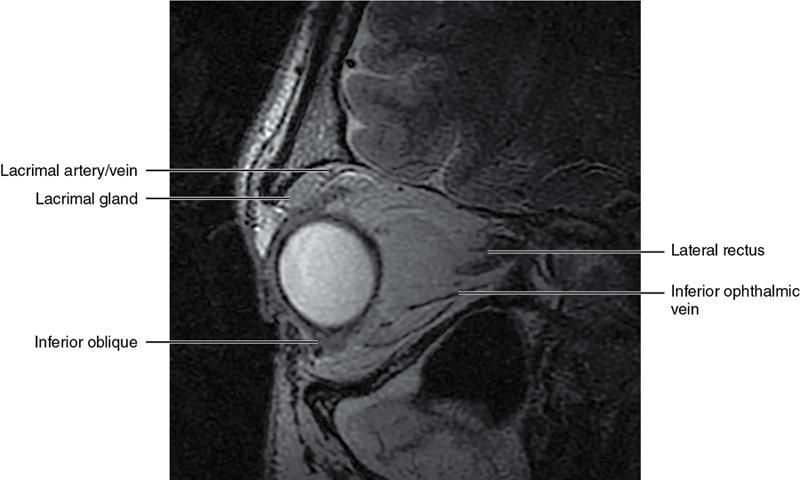
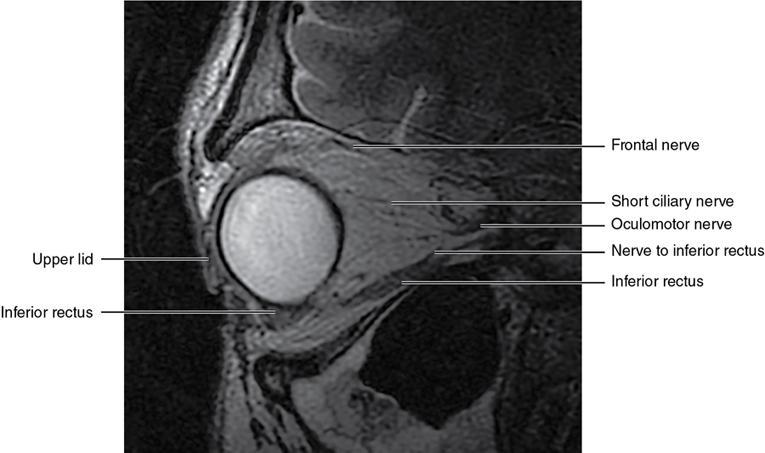
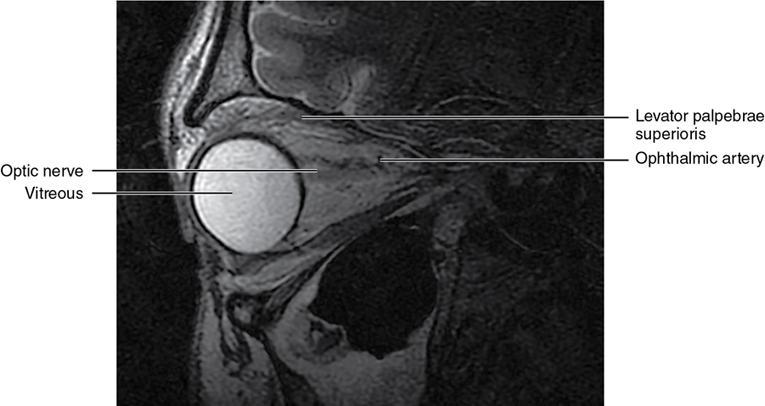
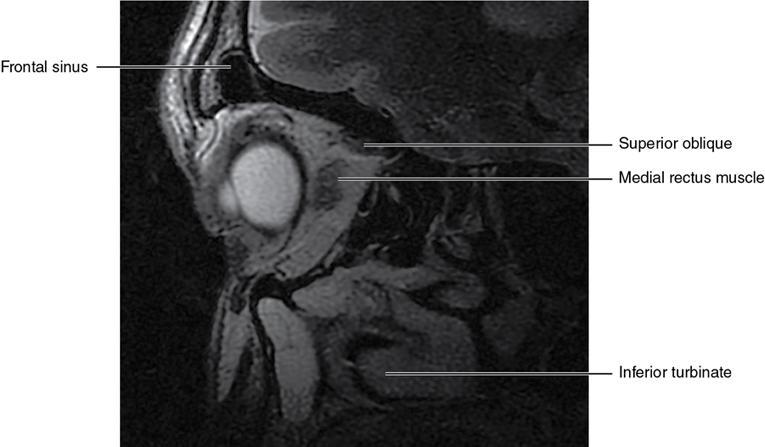
Orbit is a paired pyramidal cavity in the facial skeleton, between the maxillary sinus inferiorly, the frontal sinus superiorly and ethmoid and sphenoid sinuses medially. The apex opens into the cranial cavity via the fissures and optic canal. It contains the eyeball, optic nerve, extraocular muscles, lacrimal apparatus and neurovascular structures. It is aptly called the “window to the brain”, as patients with neurological disorders often present to the ophthalmologist! Imaging of the orbit involves the study of the soft tissues, viz., the eyeball, optic nerve, lacrimal glands and extraocular muscles which are best imaged by magnetic resonance imaging (MRI) and bony orbit best studied using computed tomography (CT) with 3D/volume rendering imaging. The study of the imaging anatomy is an essential part of orbit imaging, and therefore, radiologists should be familiar with the important imaging landmarks and their normal CT and MRI appearance. The four rectus and two oblique muscles are responsible for the ocular movements. The four rectus muscles, viz., the superior, inferior, medial and lateral originate from the annulus of Zinn at the apex and insert into the sclera just behind the limbus. The inferior oblique muscle originates from the maxillary bone just behind the rim, posterolateral to the lacrimal fossa, and inserts in the posterolateral surface of the globe. The superior oblique muscle originates from the periosteum of the body of the sphenoid, above the annulus, and courses along the superomedial wall; the tendon then passes through a fibrocartilaginous ring, trochlea which acts as pulley and then reflects posterior, inferior and lateral and passes beneath the superior rectus tendon and inserts in the superolateral aspect of the globe. The extraocular muscles show intermediate signal in T1- and hypointense T2-weighted images with respect to the orbital fat. All the muscles except the lateral rectus and superior oblique are supplied by the third(oculomotor) cranial nerve. The superior oblique is supplied by the fourth(trochlea) nerve and lateral rectus by the sixth (abducens) nerve. The levator palpebrae superioris is not involved in ocular motility but involved in lid retraction. It originates from periorbita, above the annulus of Zinn, and inserts in the tarsal plate. The blood supply of the extraocular muscles is from the muscular branches of the ophthalmic artery. The optic nerve can be divided into four parts: The lacrimal gland is an almond-shaped structure superolateral to the globe. It is divided into the orbital and palpebral lobes by the LPS aponeurosis. The orbital lobe lies deep to the orbital septum in the lacrimal fossa. It shows isointense signal in T1-weighted images and hypointense signal in T2-weighted images with respect to orbital fat. It is composed of both epithelial and lymphoid elements similar to the salivary glands. Lacrimal apparatus is responsible for the drainage of the lacrimal fluid (tears) into the nasal cavity. It consist of the superior, inferior, common canaliculi, lacrimal sac and nasolacrimal duct. The upper and the lower canaliculi run along the medial margin of the respective eyelids and measure 6–10 mm on length. They open into the common canaliculus. The common canaliculus is 2–5 mm long and enters the lacrimal sac via a small recess in the lateral wall of the lacrimal sac, known as the sinus of Maier. At the opening of the common canaliculus into the lateral wall of the lacrimal sac, two small mucous membrane folds are seen, which have valvular functions. The upper fold, the valve of Rosenmuller, and the lower fold, the valve of Huschke, are responsible for mucocoele formations when there is impaired drainage into the nasolacrimal duct causing the sac pressure to increase and closure of these valves. The lacrimal sac is situated in the lacrimal fossa between the anterior and posterior lacrimal crest and is the dilated superior end of the nasolacrimal duct. The lacrimal sac opens into the nasolacrimal canal. There is a small constriction at the lower end of the sac caused by a mucous membrane fold called the valve of Krause. The nasolacrimal duct is an extension of the lacrimal sac and runs vertically downwards in the bony nasolacrimal canal located in the lateral wall of the nasal cavity and open in the inferior meatus. The valve of Hasner, located at the end part of the nasolacrimal duct, acts as a functional barrier for the backward tear outflow. The lacrimal sac and nasolacrimal duct are best seen on T2-weighted images when fluid filled. The lacrimal canaliculi are visualized on high-resolution surface coil MRI. The orbit is a conical cavity which has a base, an apex and four walls. The base opens in the face and has four borders. The following bones constitute the formation of the orbital rim: The infraorbital foramen contains the infraorbital nerve and artery. It connects the orbit to pterygopalatine and infratemporal fossa. The superior orbital foramen is seen as a small notch on the medial third of the superior orbital rim through which passes the supraorbital nerve and artery. The eyeball develops from the brain as an outpouching called the optic vesicle. The malformations of the eye can therefore be associated with brain malformations, occur in isolation or be part of multisystem anomalies or syndromes. Congenital malformations of the eye include the following: It may be unilateral or bilateral. It may or may not be associated with colobomatous cysts. Those with colobomatous cysts result from failure of fusion of the fetal optic fissure. Imaging – Incompletely developed anterior segment, or a rudimentary cyst-like globe. Congenital cystic eye results due to failure of the optic vesicle to invaginate during the fourth week of embryogenesis. It can be associated with other nonocular abnormalities such as facial clefts, choanal atresia, malformation of the sphenoid bone, agenesis of corpus callosum and midbrain deformities. Imaging: Differential Diagnosis – Microphthalmia with colobomatous cyst. Endophthalmitis is an inflammation of the eye without involvement of the sclera, and panophthalmitis is inflammation of all the ocular coats and is usually caused by an infection. Scleritis is a chronic, painful and potentially blinding inflammatory disease commonly seen in the fourth to sixth decades with a female preponderance. It can be classified as anterior (necrotizing or nonnecrotizing) or posterior (nodular or diffuse). Systemic associations include autoimmune disorders, viz., rheumatoid arthritis, systemic lupus erythematosus, relapsing polychondritis, spondyloarthropathies, Wegener granulomatosis, polyarteritis nodosa and giant cell arteritis. Clinical presentation – Redness and pain are the most common presenting symptoms. MRI findings – FIESTA or CISS imaging is sensitive to identify intraocular cyst with scolex. Typical cyst with scolex is not always identified on imaging. Vogt–Koyanagi–Harada (VKH) syndrome is a rare multiorgan autoimmune disorder affecting the pigmented tissues of the eye, ear, skin and central nervous system. The patients usually present with uveitis, patchy vitiligo, alopecia, hearing loss, tinnitus, headaches and meningismus. Imaging: MRI is the investigation of choice. The uveal tissue is the most common ocular tissue affected by tuberculosis. Diagnosis is clinical, and imaging is done in case of granuloma or abscess and when diagnosis is in doubt. Granulomas larger than 3 mm can be appreciated on surface coil imaging. They are intermediate in T1 WI and hypointense in T2 WI. They show uniform contrast enhancement. There may be associated thickening of the ocular coats. Brain imaging should be performed for intracranial granulomas. Intraocular abscess can result from bacterial, fungal or tuberculous infection. In bacterial infection, the presentation is acute, and tuberculous and fungal have a rather subacute or indolent course. MRI – dome-shaped lesions with central T1 hypointense and T2 hyperintense signal and an isointense rim. Diffusion-weighted imaging (DWI) shows central restricted diffusion. Contrast imaging shows thick-walled uniform enhancement and nonenhancing necrotic center. Associated ocular coat thickening and periocular inflammation may be present. It is the most common intraocular malignancy encountered in clinical practice. Common sites are choroid, ciliary body and iris. The posterior uveal melanoma is usually well-defined dome-shaped lesion and can occur anywhere from the ciliary body to the posterior pole. Factors that predict an unfavourable prognosis are tumour size, extraocular extension, extension to the ciliary body and intense pigmentation. The choroidal melanoma is dome or mushroom shaped. Liver is the common site for metastases; hence, tumour workup should include liver imaging.
Part A orbits:
Introduction
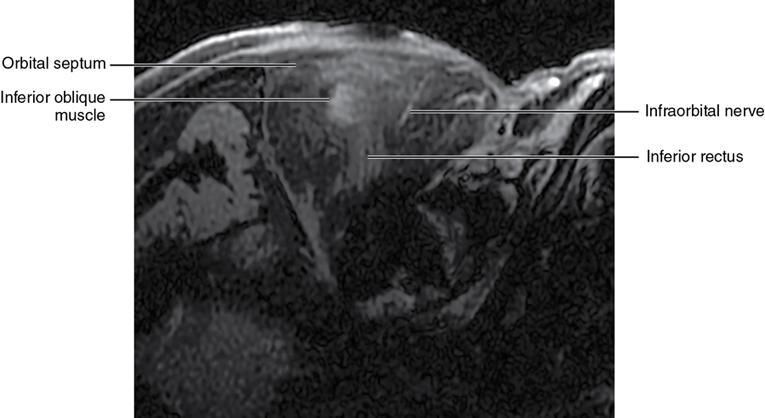
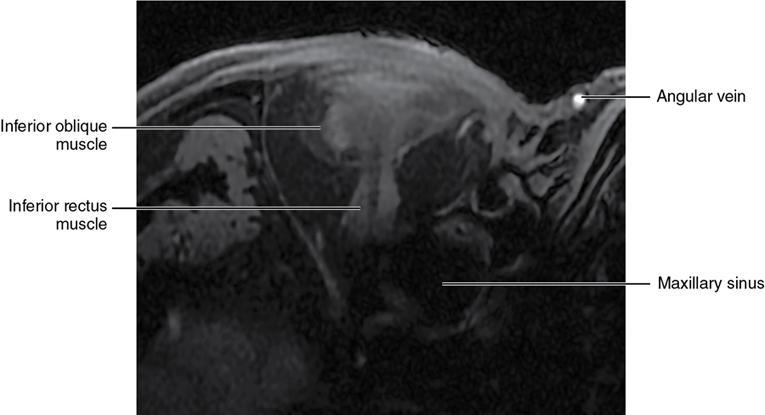
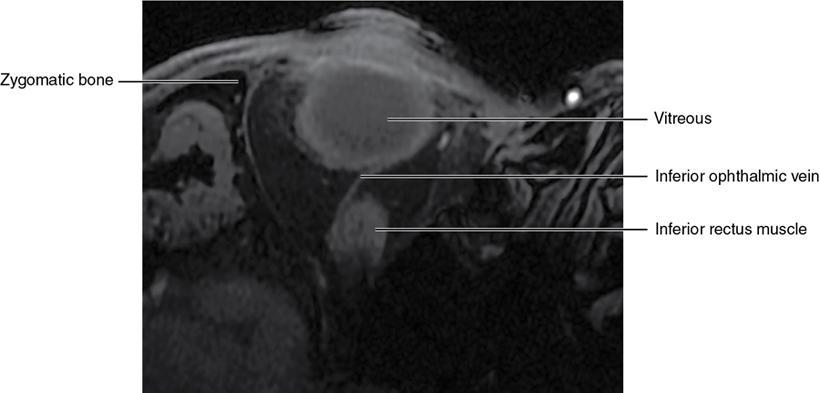
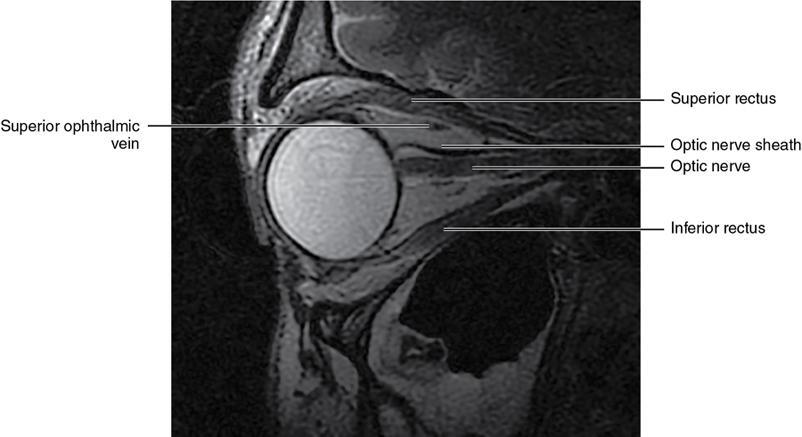
Soft-tissue anatomy
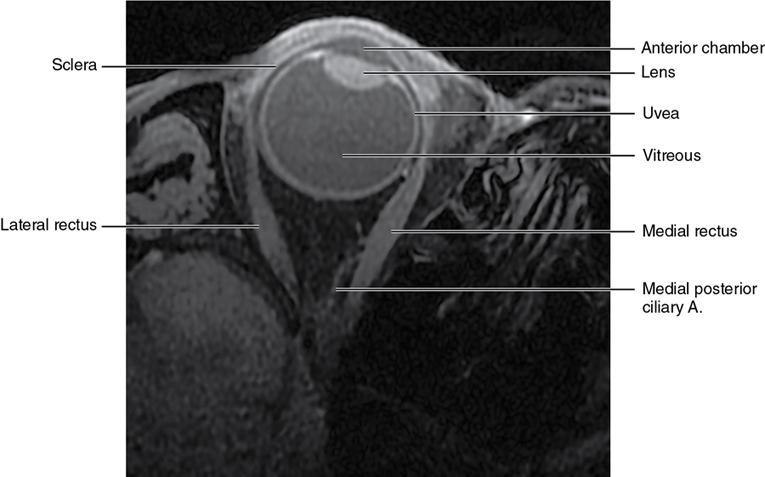
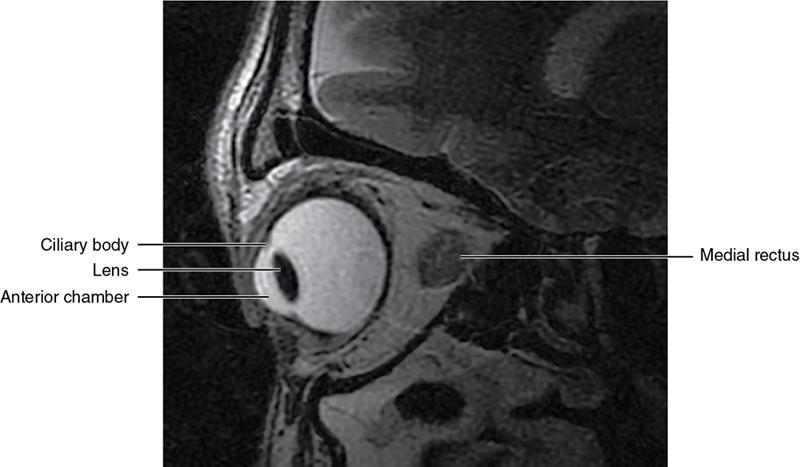
The ocular coats
Extraocular muscles (Table 3.25.1)
Globe (mm)
Axial Length
22–25 mm
Extraocular muscles thickness (mm)
Inferior rectus
3.6–4 0.0 mm
Medial rectus
3.8–4.5 mm
Lateral rectus
3.0–3.5 mm
Superior rectus
3.7–4.0 mm
Optic nerve sheath complex
4 – 5 mm
Orbit dimensions- transverse
36–37 mm
Vertical
37–38 mm
Orbit volume
30 cc
Connective tissue system
Nerves
The optic nerve
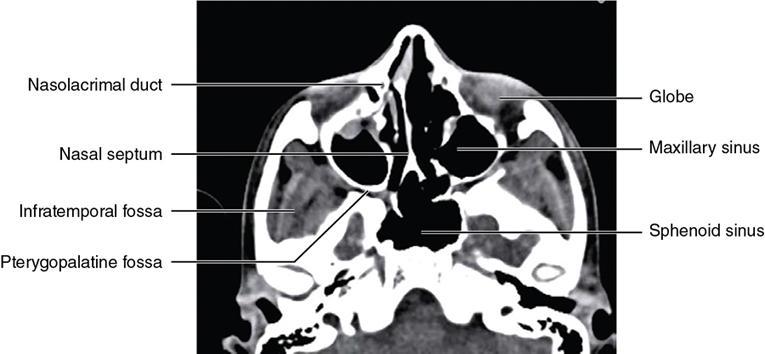
Lacrimal system
Lacrimal apparatus
Bony anatomy
Foramen and fissures
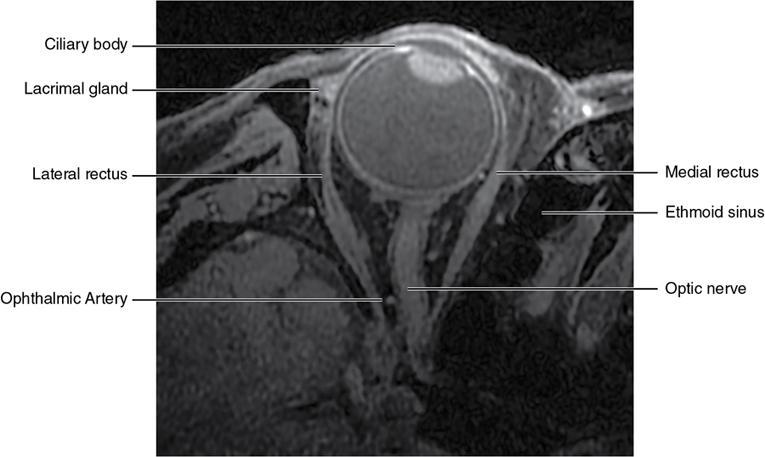
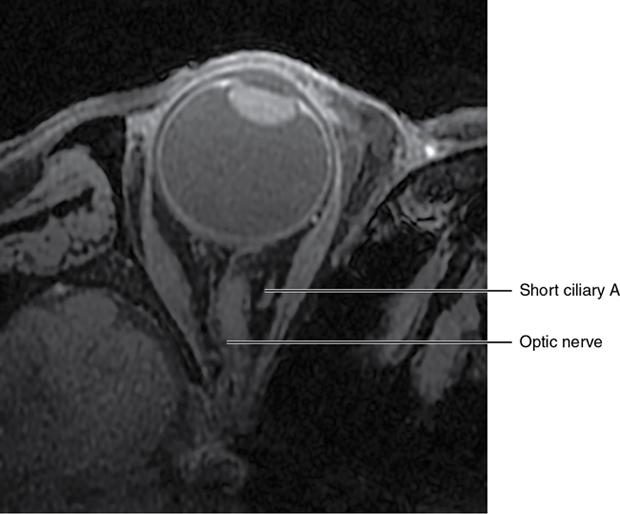
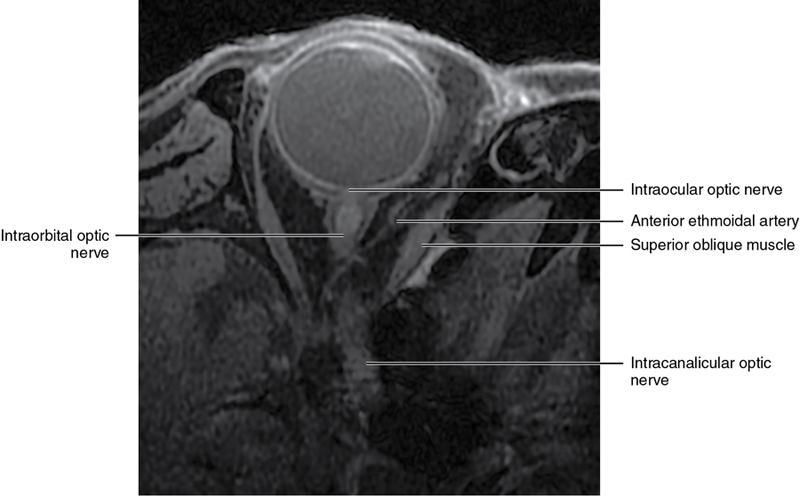
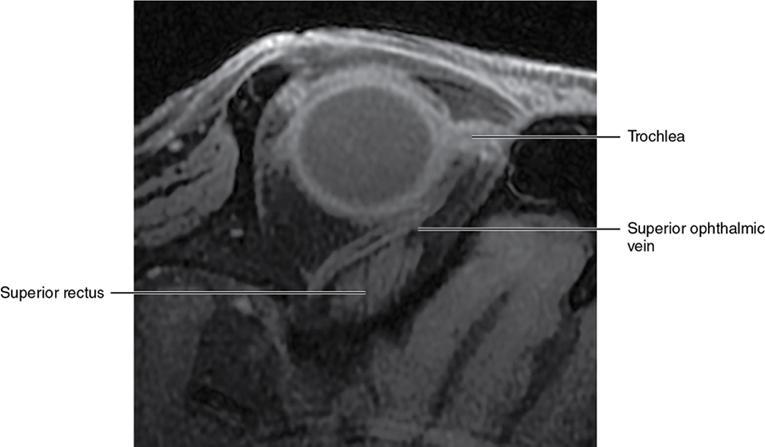
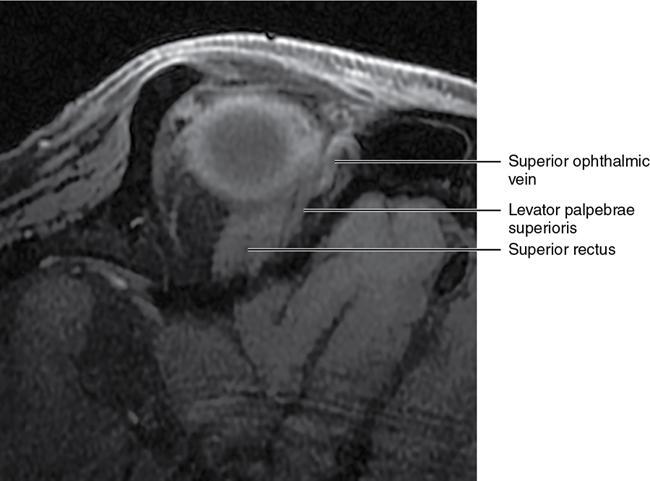
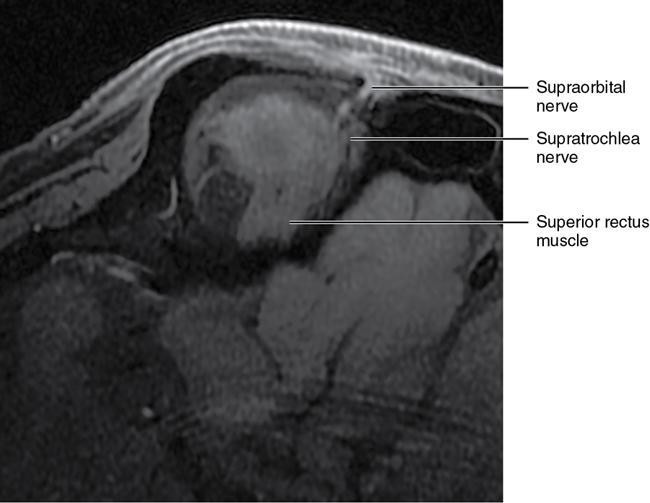
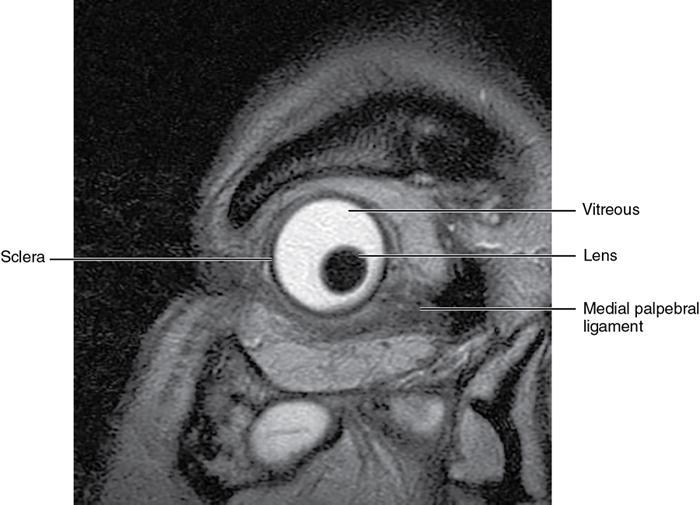
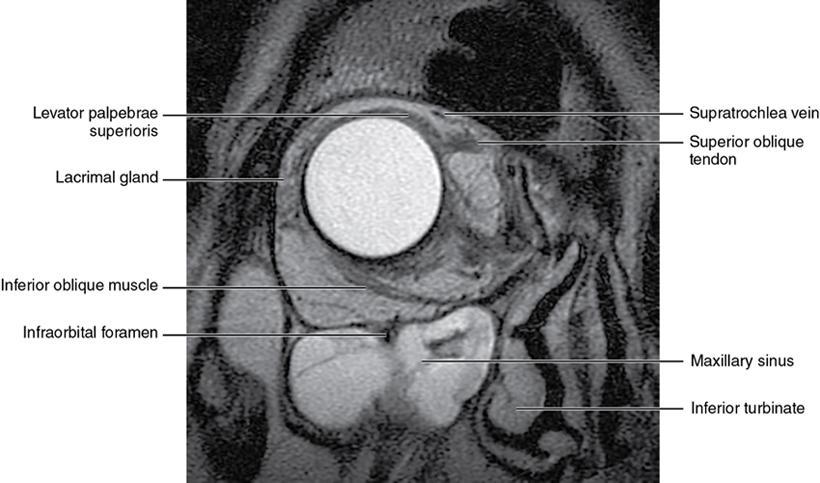
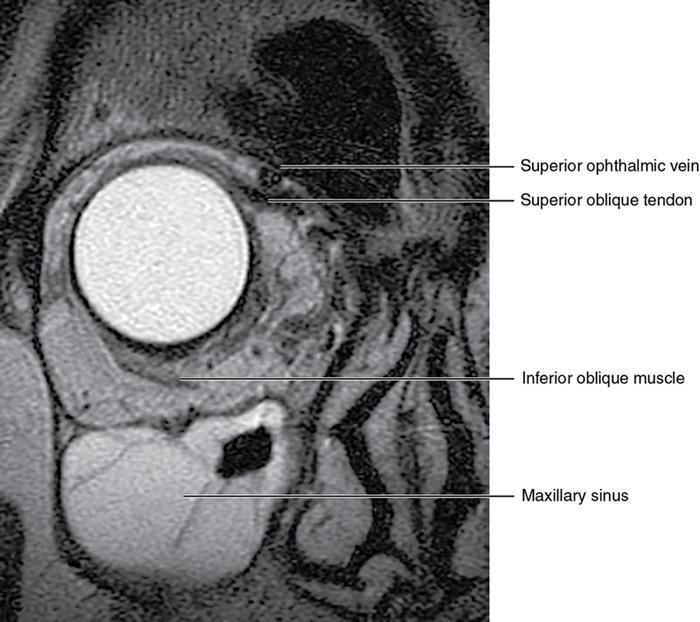
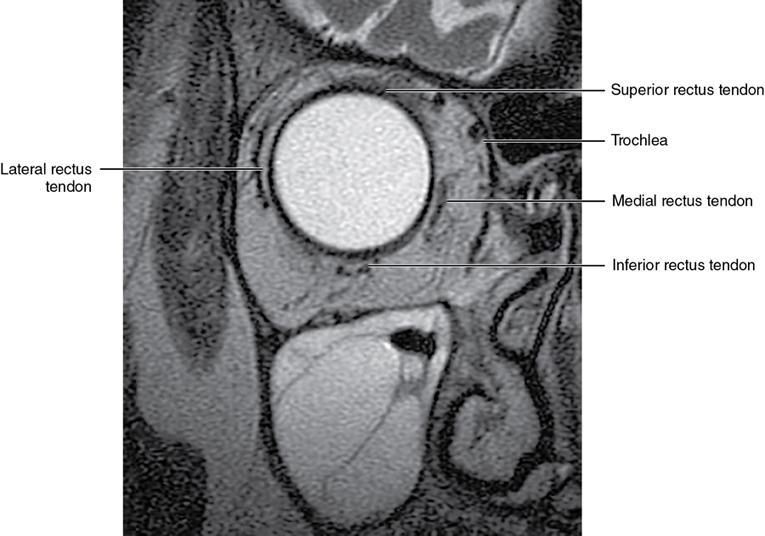
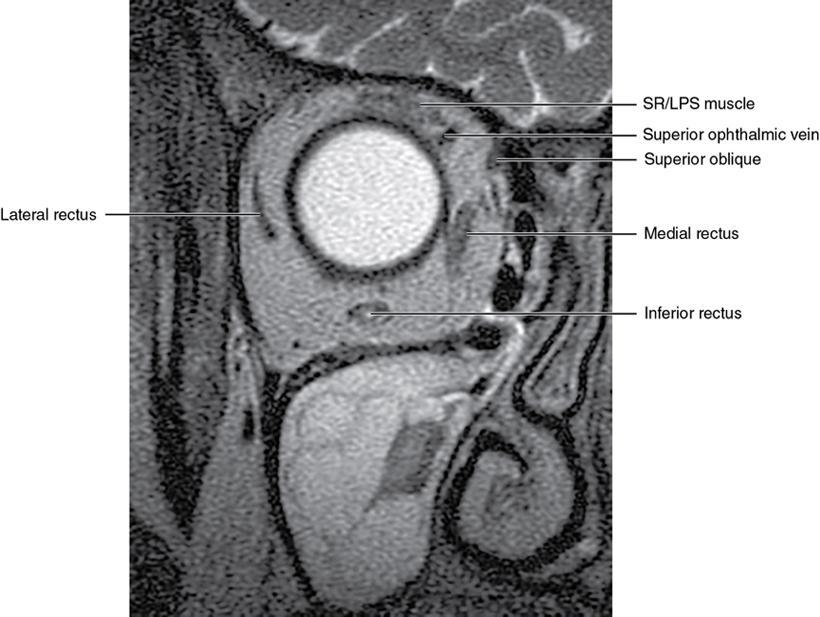
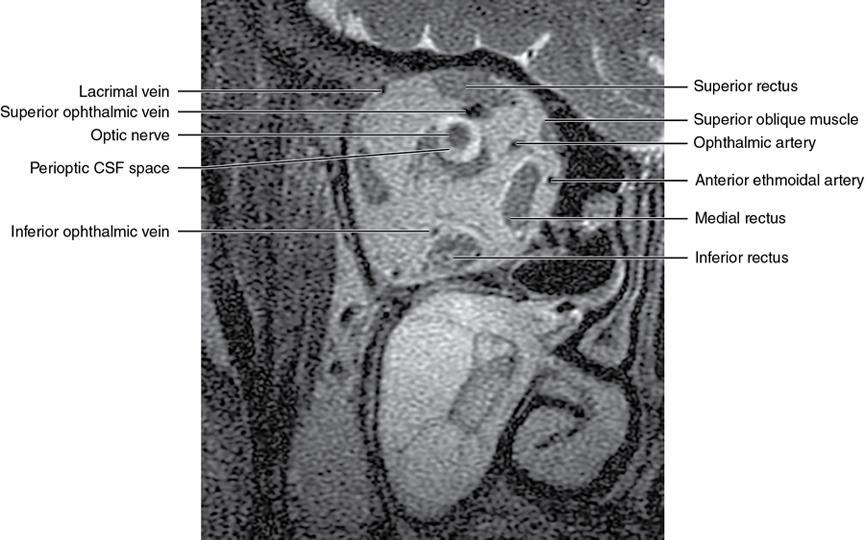
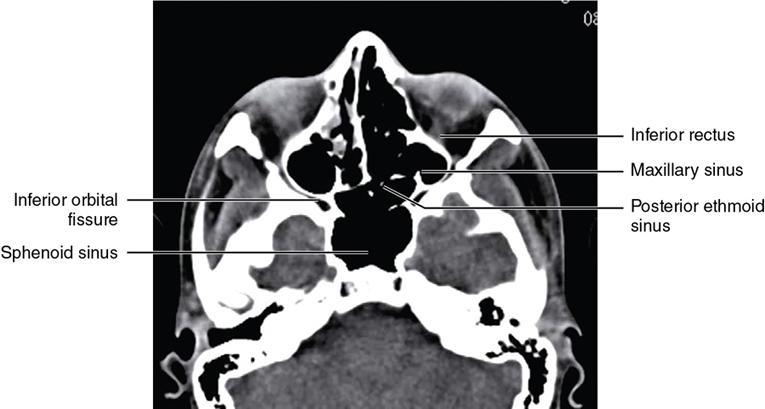
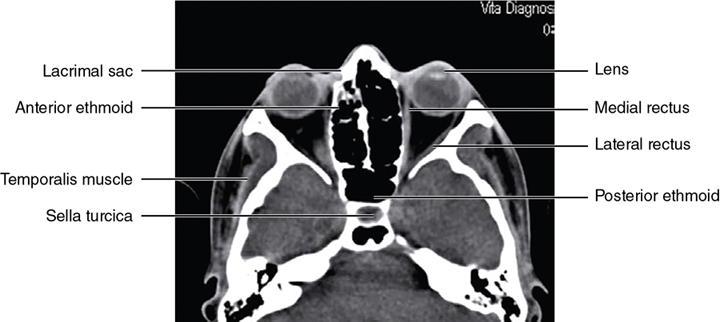
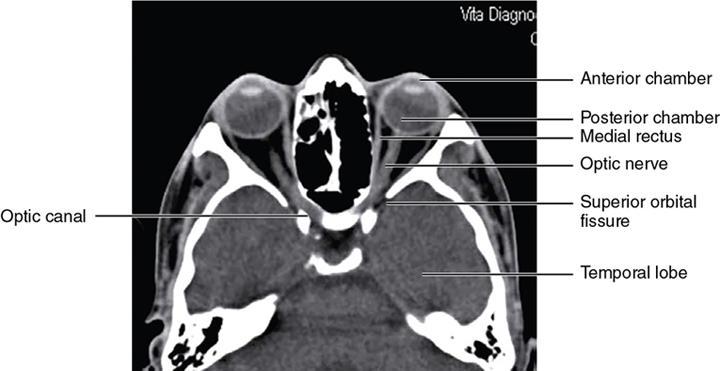
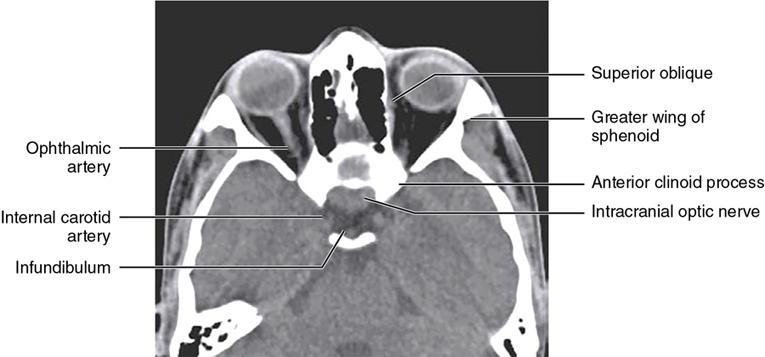
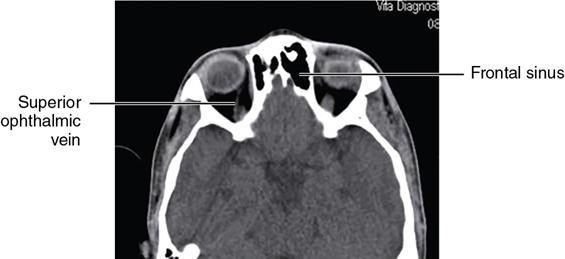
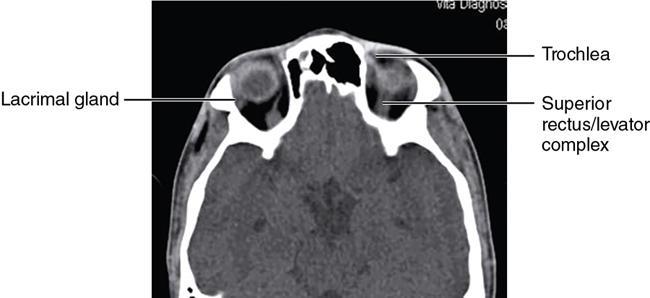
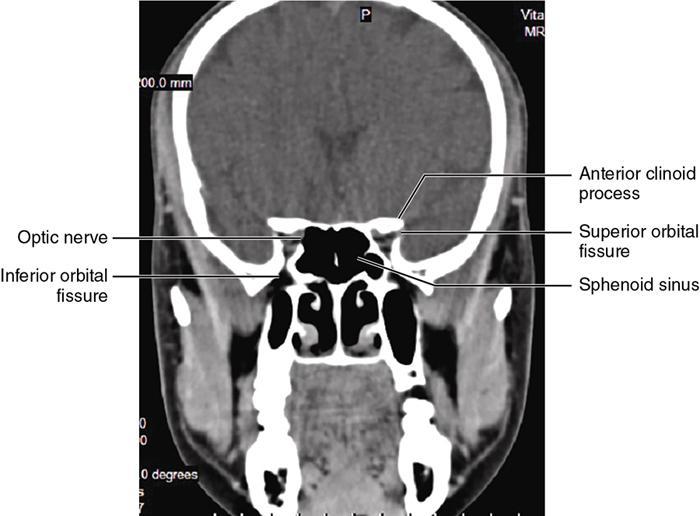
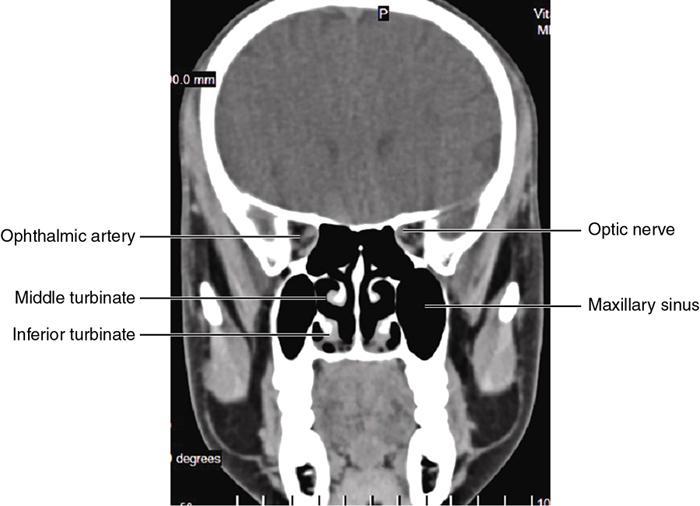
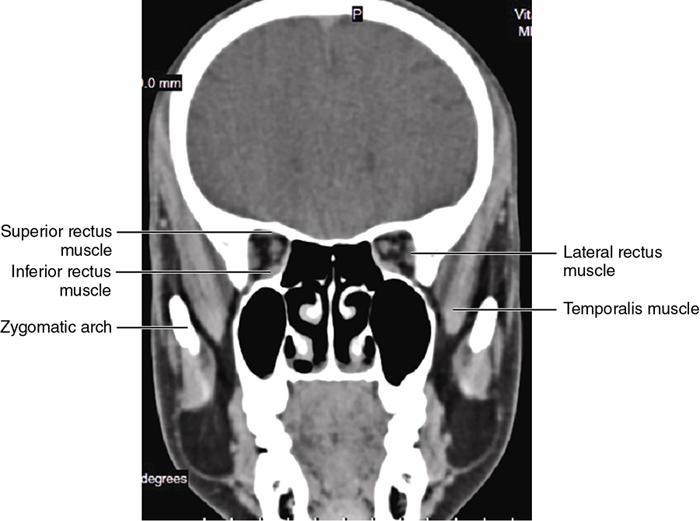
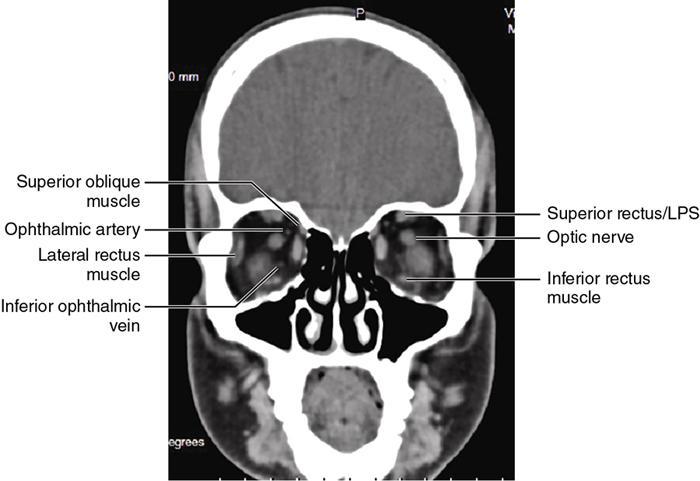
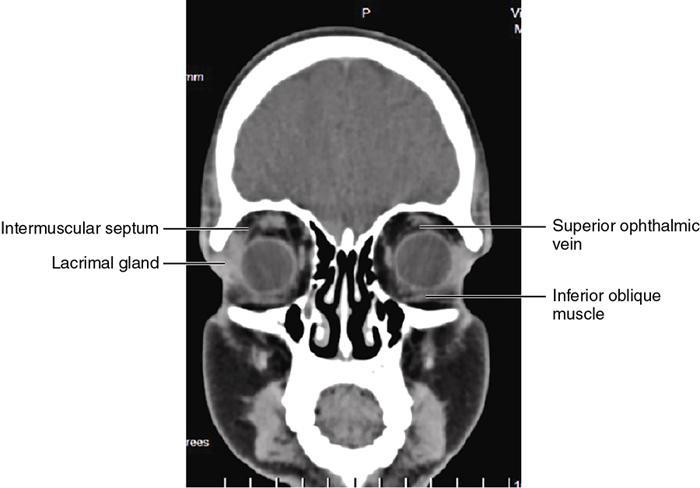
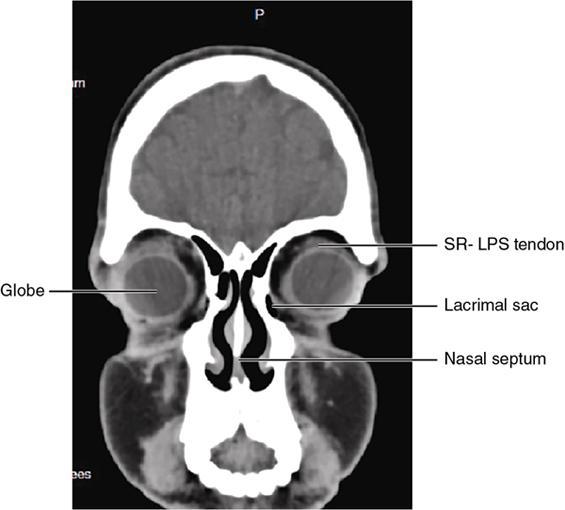
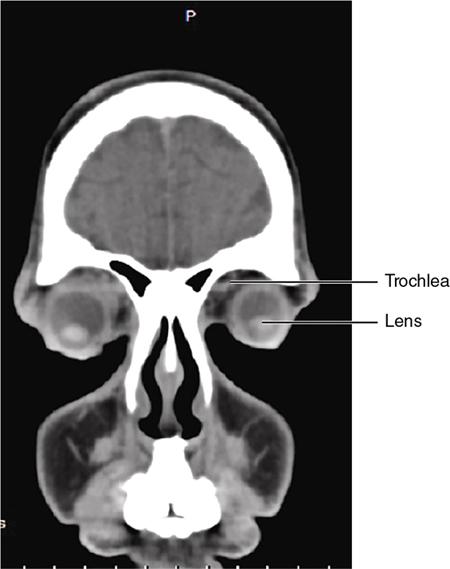
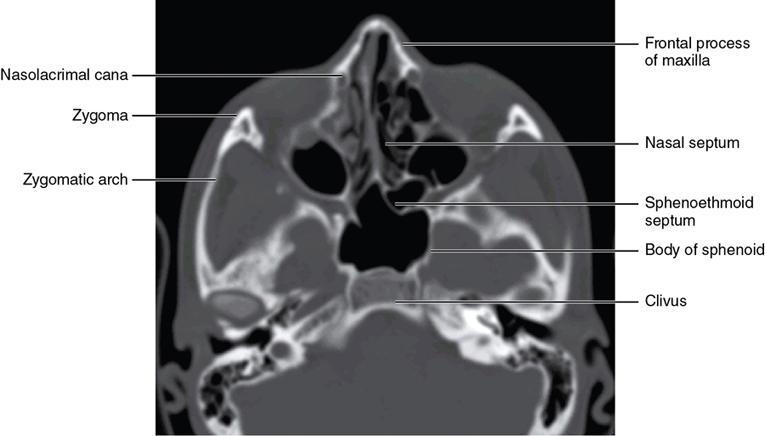
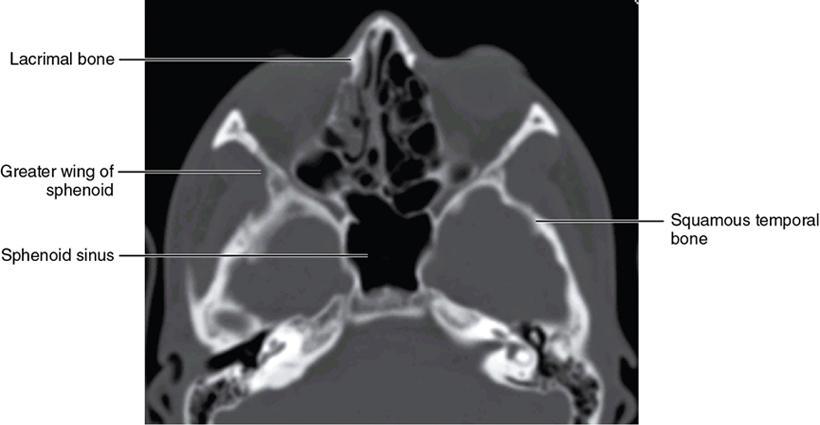
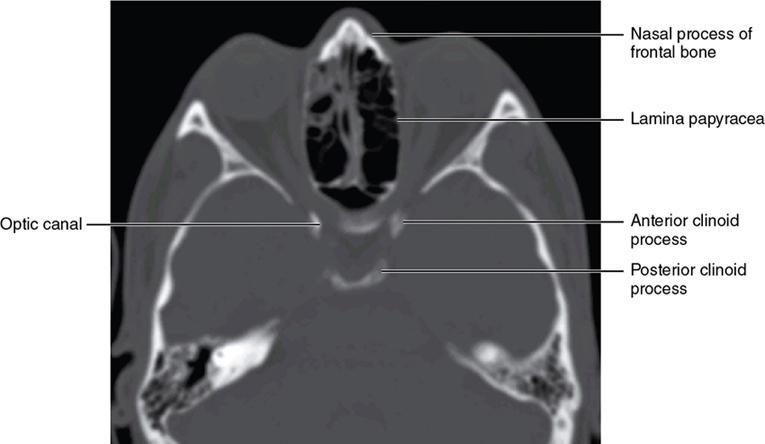
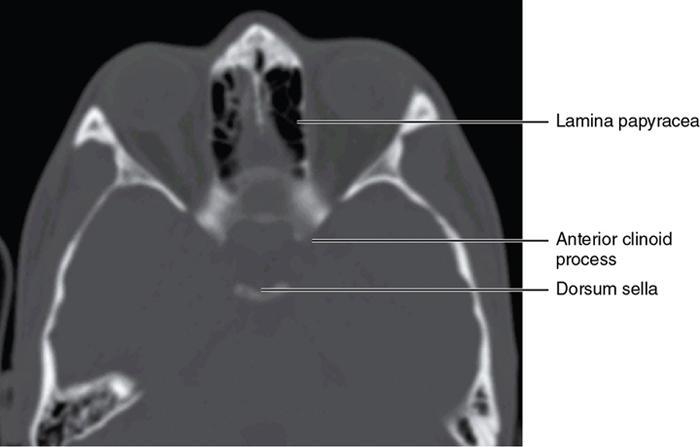
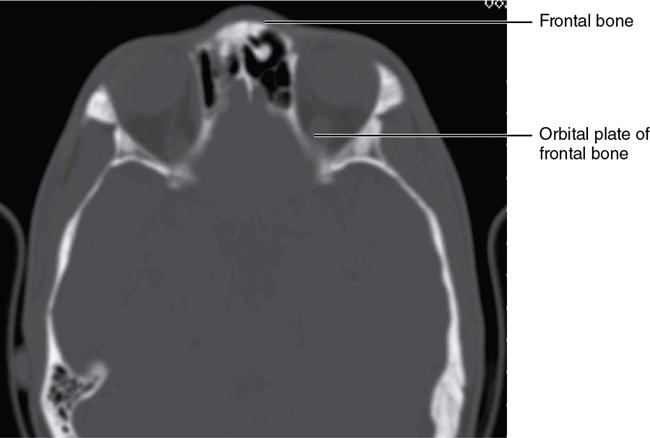
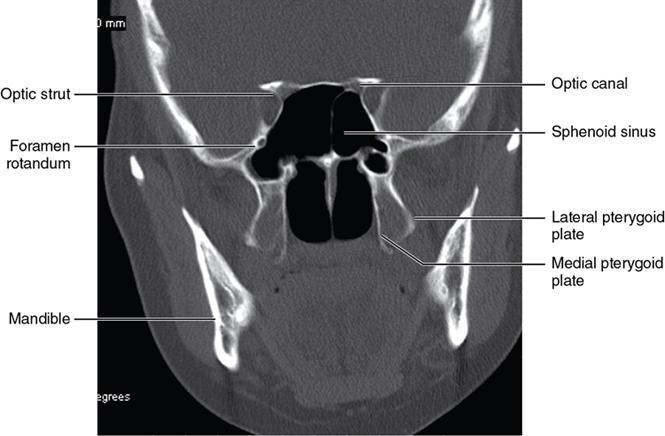
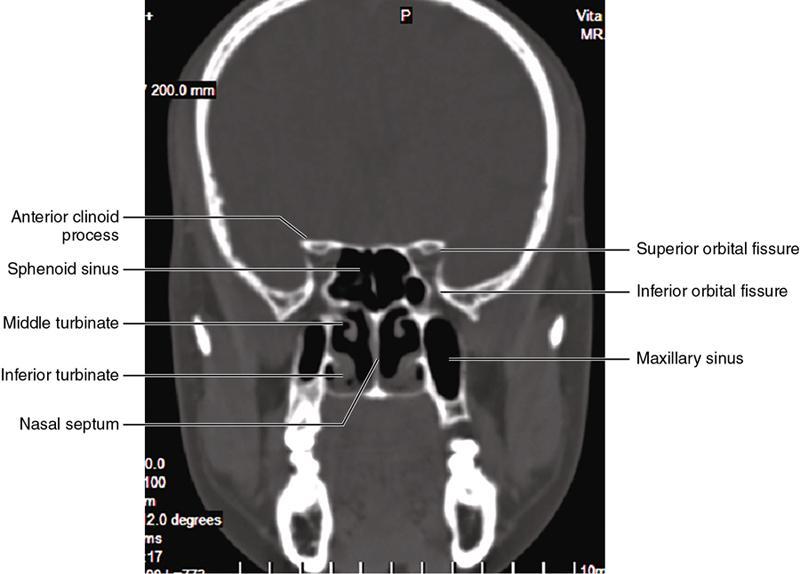
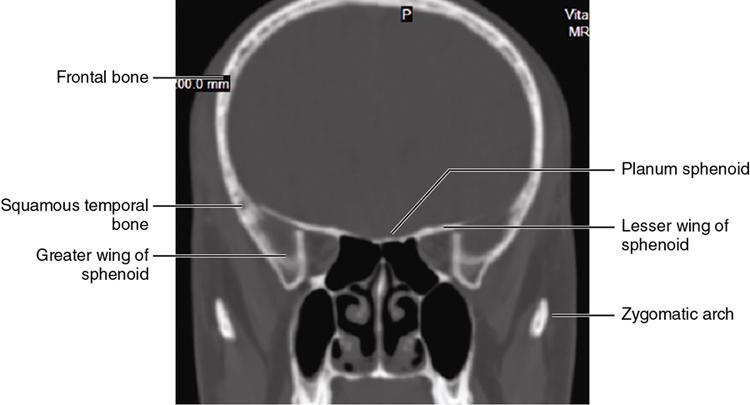
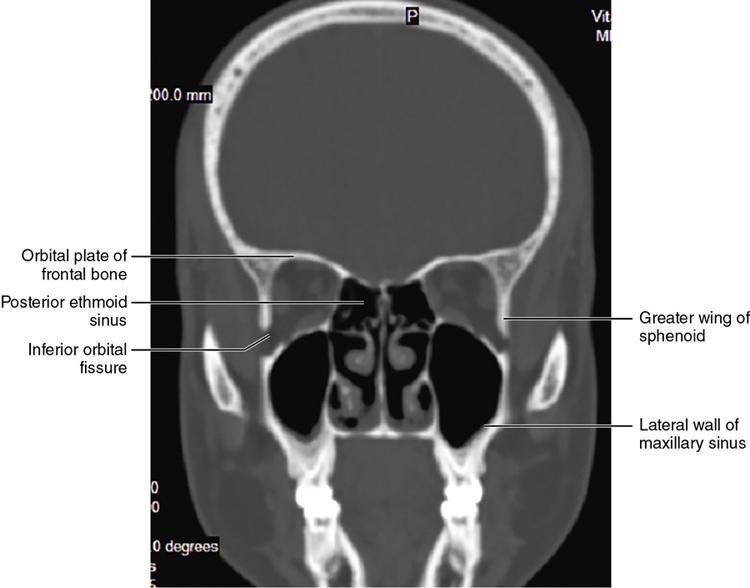
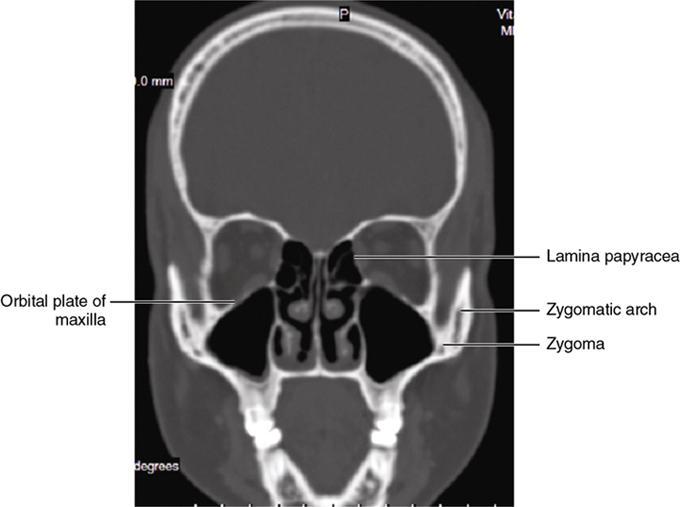
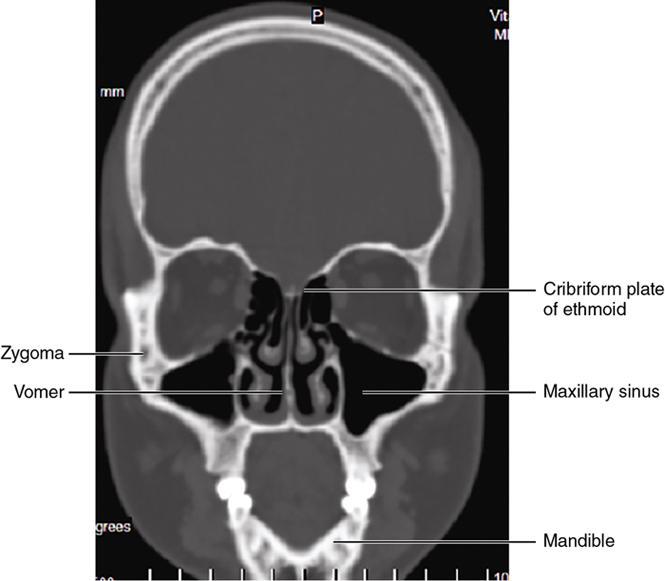
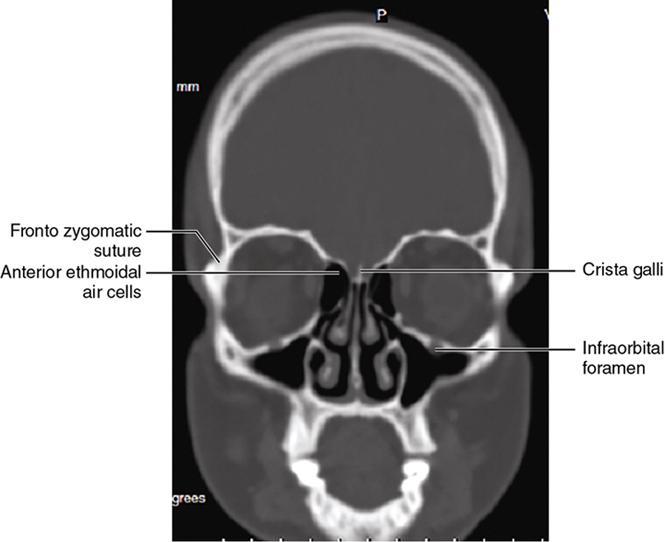
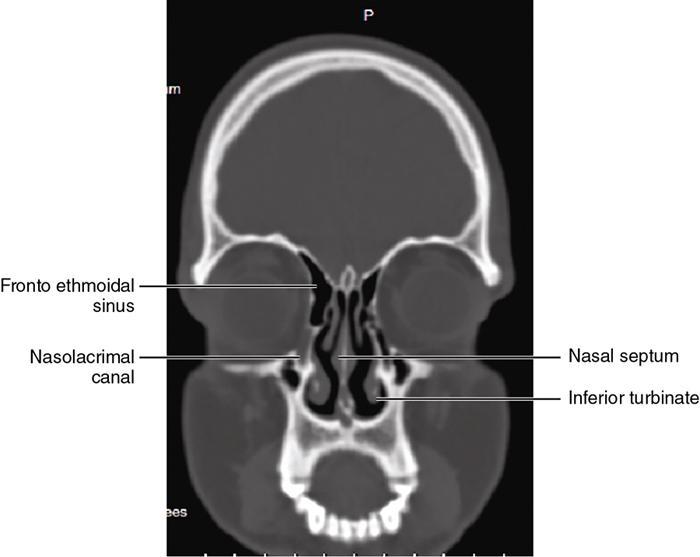
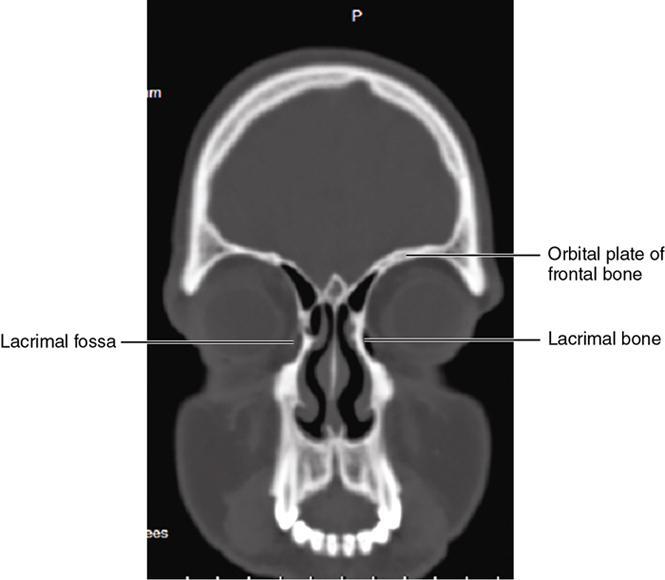
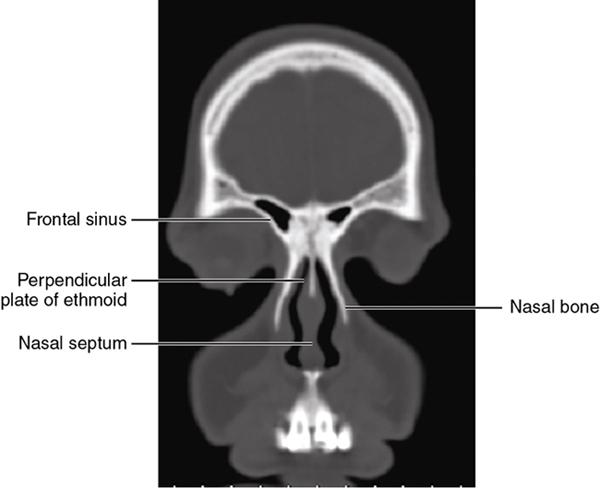
Congenital malformations of the eyeball and orbit
Anophthalmia
Microphthalmia
Computed tomography/magnetic resonance findings
Congenital cystic eye
Inflammatory lesions of the eye
Endophthalmitis and panophthalmitis
Imaging
Scleritis
Vogt–Koyanagi–Harada syndrome
Intraocular tuberculosis
Intraocular abscess
Imaging
Intraocular tumours – adults
Melanoma
Imaging features
Computed tomography
Magnetic resonance imaging
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree