Chapter Outline
Percutaneous Abscess Drainage: Clinical Considerations
Percutaneous Abscess Drainage: Technique
Additional Techniques for Abscess Drainage
Catheter Management after Drainage
Factors Affecting Outcome of Abscess Drainage
Tips and Tricks to Improve Outcome
Intra-abdominal abscesses occur from a wide variety of causes and are characterized by accumulation of an infected fluid collection within the structures of the abdomen and pelvis. Depending on the etiology, the abscesses can develop either within the solid or hollow visceral organs or within the intraperitoneal and retroperitoneal spaces. Without timely and adequate drainage, intra-abdominal abscesses cause increased morbidity and mortality in spite of appropriate antibiotic coverage. Image-guided percutaneous drainage provides a safe substitute to surgical débridement in the management of these abscesses. Advances in percutaneous techniques have resulted in abscess cure rates of more than 90%, and image-guided drainage is currently considered the preferred treatment option in the management of intra-abdominal abscesses. Higher cure rates in treatment of intra-abdominal abscesses have also resulted from improved diagnostic imaging techniques, such as computed tomography (CT) and magnetic resonance imaging (MRI), which allow early diagnosis and thereby timely intervention. Percutaneous catheter drainage of abscesses has several advantages over surgical lavage for treatment of intra-abdominal abscess. These include absence of a laparotomy scar, shorter hospital stay, avoidance of general anesthesia, lower risk of complications, and lower morbidity and mortality.
Rising use of CT and MRI in patients thought to have intra-abdominal sepsis has permitted early and accurate detection of abscesses. This is particularly true in patients with predisposing factors for development of abdominal abscesses, such as individuals with Crohn’s disease, diverticulitis, and appendicitis, and in postoperative patients after abdominal surgery. Imaging not only provides precise diagnosis but also guides interventional radiologists in safely draining the infected fluid collections. Despite the high success rates reported for percutaneous abscess drainage, it can at times be technically challenging because of the presence of fistulas, inaccessible locations, and comorbidities of the patient. In this chapter, our endeavor is to provide the reader with a synopsis of a radiologist’s role in the diagnosis and management of intra-abdominal abscesses, particularly focusing on percutaneous abscess drainage.
Pathogenesis
The development of abscesses in the abdomen and pelvis is pathophysiologically similar to that elsewhere in the body, although the cause is often multifactorial. Intra-abdominal abscesses can arise from a wide variety of conditions but are commonly encountered in postoperative patients or in patients with infective or inflammatory diseases, such as inflammatory bowel disease, acute diverticulitis, or acute appendicitis. They can result from superinfection of a previously sterile fluid collection (e.g., postoperative biloma, hematoma) or arise de novo from an infectious nidus in a solid or hollow visceral organ (e.g., pyogenic liver abscess). In gross appearance, abscesses can have varied size and shape. Abscesses developing within solid visceral organs, such as liver and spleen, are often spherical or ovoid in configuration because of uniform pressure from surrounding tissue. Intraperitoneal and retroperitoneal abscesses have varied morphologic features and shape, depending on their location, spread along various fascial spaces, and pressure of surrounding structures. Mature abscesses often have a well-defined capsule composed of connective tissue (fibrin, collagen), blood vessels, and leukocytes. The contents of the abscess cavity gradually undergo liquefaction due to the enzymatic action of leukocytes. Bacterial isolates from abscess confirm the predominance of polymicrobial microorganisms in intra-abdominal infection.
Imaging Techniques
Technologic advancements in imaging techniques, particularly CT and MRI, have greatly enhanced their ability to accurately diagnose intra-abdominal abscesses. In addition to detection, imaging permits accurate depiction of extension of abscess across various fascial spaces in the abdomen. Imaging also aids the interventional radiologist in planning of interventional procedures for safe and effective drainage of the abscesses ( Table 72-1 ).
| Advantages | Disadvantages | |
|---|---|---|
| Ultrasound |
|
|
| Computed tomography |
|
|
| Magnetic resonance | Superior soft tissue resolution allows accurate diagnosis |
|
| Fluoroscopy |
|
|
Plain Radiography or Fluoroscopy
Plain film radiography has a limited role in the diagnosis and management of intra-abdominal abscess. The radiographic signs that are helpful in the diagnosis of intra-abdominal abscesses include an abnormal gas pattern, extraluminal air-fluid level, free intraperitoneal air, soft tissue mass, and loss of normal fat planes and interfaces. Nonetheless, plain radiography is an insensitive technique that is not reliable in day-to-day practice. Real-time fluoroscopy, on the other hand, is useful in guiding percutaneous drainage of abscesses in specific locations; for example, it can be used in conjunction with ultrasound to facilitate drainage of subphrenic abscesses while avoiding pleural transgression. Fluoroscopic guidance is also useful for aiding catheter manipulations, such as catheter repositioning and exchanges after initial placement of the percutaneous catheter.
Ultrasound
Ultrasound is often used as a screening modality for initial diagnosis of intra-abdominal fluid collections. Ultrasound has several advantages over other imaging modalities; it is relatively inexpensive, easily available, and portable and does not use ionizing radiation. Ultrasound is particularly beneficial in rapid bedside diagnosis of abscesses in seriously ill patients in hospital intensive care units who are unable to travel to the radiology department for a CT scan. The ability to perform bedside ultrasound scans is especially valuable for guiding placement of percutaneous drainage catheters into abdominal abscesses in intensive care unit patients.
Ultrasound is also the preferred imaging modality for diagnosis and for guiding percutaneous drainage of superficially located abscesses and multiloculated collections. Because of its real-time nature, it also confers a low risk for traversing vascular structures, bowel, or the pleural cavity during ultrasound-guided abscess drainage. In patients with deep-seated pelvic abscesses, ultrasound also provides the opportunity for drainage through the transrectal or transvaginal route. In experienced hands, ultrasound allows rapid and accurate drainage of intra-abdominal abscesses compared with CT.
The sonographic appearance of abdominal and pelvic abscesses depends on their location and internal contents. In general, the abscesses are seen as anechoic to hypoechoic cystic collections with internal echoes and debris. More complex abscesses can have variegated hypoechoic and solid cystic appearance with irregular margins, internal septation, and debris ( Fig. 72-1 ). Presence of gas within a fluid collection often demonstrates “dirty” shadowing and is highly suggestive of infection. The demonstration of multiloculations within a fluid collection is a valuable feature of ultrasound, which has important treatment implications because such abscesses often require insertion of multiple catheters or instillation of thrombolytic agents to facilitate optimal drainage. On occasion, the diagnosis of intraperitoneal abscesses can be challenging on ultrasound because of the need to differentiate these collections from surrounding fluid-filled bowel loops. The real-time nature of ultrasound aids in this differentiation by demonstrating peristalsis within bowel loops. The characteristic sonographic signature of the bowel wall is an additional differentiating feature. Doppler interrogation of intra-abdominal fluid collections is essential to rule out the possibility of pseudoaneurysms. This is particularly relevant in fluid collections adjacent to major vessels or in conditions with a high incidence of pseudoaneurysm formation, such as acute pancreatitis.
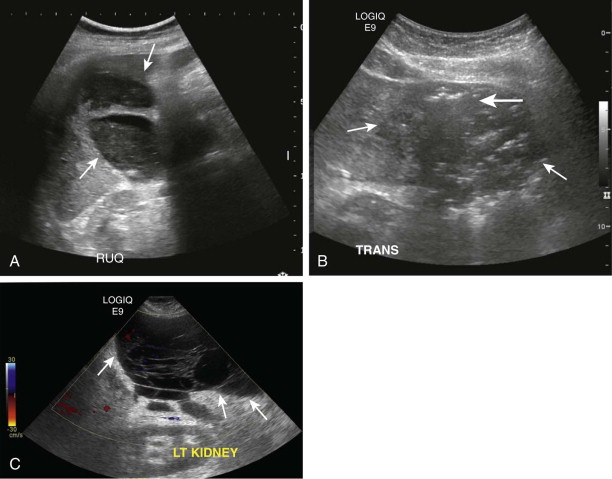
Ultrasound, however, has several limitations. Despite its value in abscess detection, ultrasound has a limited ability to define the spread of abscesses and inflammation across fascial planes within the abdomen. Ultrasound is highly operator dependent, and detection of fluid collections is rendered challenging in patients with ileus or those with extensive surgical wounds. For the same reason, ultrasound fares poorly in the detection of deep-seated abscesses in the retroperitoneum because of overlying bowel gas. Wound dressings and postsurgical drains also limit adequate visualization of intra-abdominal abscess.
Computed Tomography
CT is the imaging modality of choice for initial diagnosis and management of patients with suspected intra-abdominal abscess. * CT allows precise anatomic localization of the abscess, defines its extent, and delineates its relationship to other intra-abdominal structures, such as bowel loops and vascular structures. * Multiplanar reformations (coronal and sagittal) are extremely valuable in defining abscess extent and in assessment of collections in the subphrenic location around the dome of the liver and the spleen. In patients with intra-abdominal sepsis and in postoperative patients, CT affects management by allowing detection of multifocal abscesses at distant locations within the abdomen and pelvis. CT is valuable not only for diagnosis of intra-abdominal abscess but also in the planning of percutaneous interventions. * CT is less successful in the depiction of internal septations within an abscess, a feature better visualized on ultrasound.
* References .
In patients thought to have an infected intra-abdominal process, CT should ideally be performed after the administration of intravenous and oral contrast media. Intravenous injection of contrast media not only improves abscess detection but also is essential for enhanced characterization of the abscess features, such as the abscess wall. The wall of a mature abscess demonstrates enhancement after injection of contrast media, and it remains a key feature in the diagnosis of intra-abdominal abscess. Ingestion of positive oral contrast media is preferred before the CT scan as it allows differentiation of abscesses from adjacent bowel loops because unopacified bowel loops can mimic an abscess ( Fig. 72-2 ). Some authors advocate the use of delayed scans in different positions for differentiation of questionable areas of abscess from bowel loops as bowel loops generally change in position and configuration with change in the patient’s position. Positive oral contrast media are often helpful in detecting the presence of bowel perforation, particularly in patients presenting with intra-abdominal sepsis after bowel surgeries. Bowel perforation is diagnosed by extraluminal leak of the orally administered contrast material. Colonic perforations are less reliably depicted with orally administered contrast material and might require administration of rectal contrast. Instillation of rectal contrast material is optional but can be helpful in the differentiation of deep pelvic and perirectal abscesses.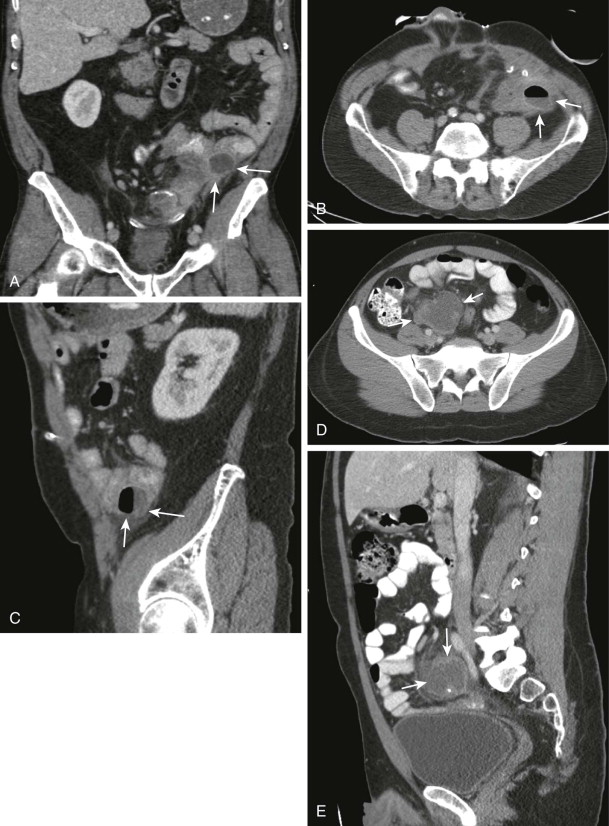
Administration of an oral contrast agent, is however, not recommended for patients presenting for follow-up CT examination after initial catheter drainage of abdominal abscess. Because these patients might require catheter manipulations for optimal positioning of the drainage catheter, oral contrast media ingestion is not recommended if the patient is to shortly receive conscious sedation.
On CT, abscesses often have fluid attenuation with internal attenuation measurements ranging between 0 and 25 HU. The density of an abscess largely depends on the abscess contents, degree of liquefaction, and presence of gas ( Fig. 72-3 ). The abscess wall often appears as a high-density irregular peripheral rim that enhances after contrast medium injection. Abscess wall enhancement is considered to be a sign of abscess wall maturation and therefore predicts favorable response to percutaneous drainage. The abscess contents often do not enhance because of the ongoing process of liquefaction and lack of internal vascularity; internal enhancement in a suspected abscess should therefore raise concern for underlying tumor. Gas within a fluid collection is highly diagnostic of an abscess and is seen in up to 50% of infected intra-abdominal fluid collections ( Fig. 72-3 ). Gas can be seen as either small locules and air pockets or air-fluid levels. Presence of gas within a collection could be due to infection with anaerobic organisms, fistulous communication with bowel, or prior interventions. Demonstration of large amounts of air in intraperitoneal or retroperitoneal collections should raise concern for possible fistulous communication with bowel loops. Bowel communication could be due to either bowel perforation or the primary cause of the abscess itself, such as Crohn’s disease or diverticulitis.

Typically, an abscess within a solid visceral organ is ovoid or spherical in shape, whereas intraperitoneal or retroperitoneal abscesses have varied morphology because they conform to the shape of the compartment in which they are located and often displace surrounding structures. The adjacent fascial planes may be obliterated or thickened by spread of inflammation, and surrounding mesenteric fat can show increased attenuation. The CT appearance of sterile and infected fluid collections can show a substantial degree of overlap. On CT, abscesses of solid visceral organs can simulate soft tissue masses. Not infrequently, needle aspiration is required to differentiate between an abscess and solid mass.
Magnetic Resonance Imaging
In the past few years, there has been an increasing trend in the use of MRI for diagnosis of intra-abdominal abscesses. MRI offers several advantages over CT, the chief ones being the absence of ionizing radiation and the superior soft tissue resolution. MRI is particularly preferred in pediatric patients and young adults because of the concerns about the harmful effects of ionizing radiation. This is particularly true in patients with Crohn’s disease, who often undergo multiple cross-sectional imaging studies in their lifetime, and intra-abdominal abscesses frequently complicate their clinical course. MRI has a problem-solving role in the characterization of complex adnexal lesions in women of reproductive age and aids in the diagnosis of tubo-ovarian abscesses.
The routine use of MRI for detection of abscess is however limited by the lack of MRI expertise, high cost, and long scanning times in abdominal MRI examinations. It is also difficult to perform good-quality examinations on critically ill patients.
On MRI, intra-abdominal abscesses typically demonstrate inhomogeneous areas of hypointensity on T1-weighted images and intermediate intensity to hyperintensity on T2-weighted images ( Fig. 72-4 ). After intravenous administration of gadolinium, the abscess demonstrates intense wall enhancement as on CT.

Scintigraphy
Nuclear scintigraphy has a limited role in the diagnosis of intra-abdominal abscesses and is often not the first-line imaging investigation to be performed. Scintigraphic studies are often obtained in patients with suspected intra-abdominal sepsis or abscess when other diagnostic modalities, such as ultrasound, CT, or MRI, have yielded negative results. The most commonly used isotopes for detection of abscess are gallium (Ga 67) and indium (In 111) to label white blood cells. Newer scintigraphic agents, such as technetium Tc 99m HMPAO-labeled white blood cells, 111 In-labeled polyclonal immunoglobulin G, and 99m Tc-labeled monoclonal antibodies, have also been reported to improve diagnosis of intra-abdominal abscess. 67 Ga scans, although sensitive, have limited specificity for abscess diagnosis because of false-positive results in conditions such as granulomatous disease, lymphoma, and normal gut. One of the main limitations of nuclear scintigraphic studies in the characterization of abdominal abscesses is the poor anatomic detail of these scans, which limits their value in planning of percutaneous interventional procedures.
Imaging-Guided Intervention
Percutaneous catheter drainage is a safe and effective means for treatment of the entire gamut of intra-abdominal abscesses irrespective of their cause and anatomic location. * Percutaneous catheter drainage is widely considered the first-line treatment option for management of intra-abdominal abscesses, superseding surgical drainage. * Percutaneous catheter drainage of abscesses is less invasive compared with surgery and is associated with fewer postprocedural complications, such as atelectasis, pneumonia, pain, and venous thrombosis. A combination of high success rate and low complication rate has made percutaneous drainage a mainstay in the treatment of abdominal abscess.
* References .
Diagnostic Aspiration
In patients with clinical and imaging confirmation of an intra-abdominal abscess, the definitive treatment is percutaneous drainage through a catheter. However, in certain circumstances in which the imaging features are not conclusive for the presence of pus, simple diagnostic aspiration of the fluid collection is indicated. Fine-needle aspiration of small amounts of fluid permits laboratory evaluation of fluid chemistry and microbiology. Diagnostic needle aspiration of fluid collections is also valuable as a temporizing measure before surgical intervention in anatomically difficult locations like the pelvis to render the surgical field clean.
Diagnostic needle aspiration of fluid collections is usually accomplished with a 20- or 22-gauge Chiba needle. The placement of the needle into the collection can be guided with either ultrasound or CT ( Fig. 72-5 ). The type of fluid aspirated from the needle often dictates the next strategy. If pure pus is extracted, the drainage catheter is immediately placed. If no pus is obtained, a Gram stain can be performed to assess the origin and cause of the contents. If Gram stain of the contents reveals leukocytes without bacteria, it is likely to be a sterile abscess, which is typically seen in hospitalized patients who have already been administered antibiotics. If Gram staining reveals leukocytes and bacteria, it is likely to be an abscess and requires catheter drainage. If there are bacteria without leukocytes, the possibilities include abscess in an immunocompromised patient and a likelihood of fistulous communication with bowel. Aspiration is not indicated if the collection has potential communications with bowel, the biliary system, or the urinary tract. Aspiration is ineffective in these situations as immediate reaccumulation of the collection occurs after initial successful percutaneous aspiration and needle removal. In addition to Gram stain, the extracted fluid should also be sent for fluid chemistry analysis to study fluid characteristics because the radiologic signs of abscesses, hematomas, urinomas, bilomas, postoperative seromas, and even loculated ascites can often overlap. Accordingly, analysis of fluid chemistry leads to recognition of the source of the fluid collection (e.g., urinomas demonstrate elevated creatinine concentration, bilomas are characterized by elevated bilirubin levels, and pseudocysts show amylase).
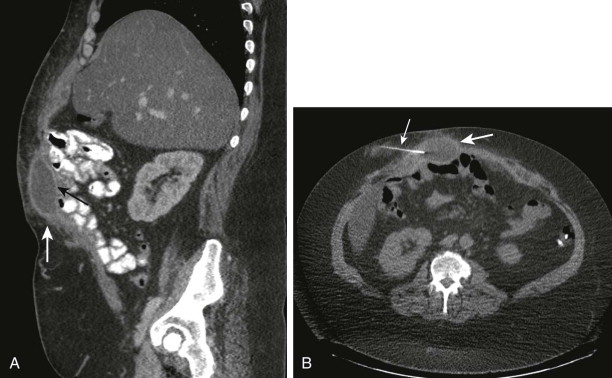
Percutaneous Abscess Drainage: Clinical Considerations
Indications
Appropriate patient selection is important before percutaneous drainage as improper patient selection with incomplete drainage often leads to an increase in morbidity. The typical indication for image-guided percutaneous drainage is the presence of an intra-abdominal fluid collection with features of abscess in the background of abdominal pain, fever, and leukocytosis. The goal of drainage in these circumstances is palliation and treatment of sepsis associated with the infected fluid collection. Another common indication for percutaneous drainage is for alleviation of symptoms caused by the size of the fluid collection (e.g., in pancreatic pseudocysts).
Contraindications
Percutaneous abscess drainage is contraindicated in patients with severe uncorrectable coagulopathy and thrombocytopenia as they predispose to increased risk of bleeding. Other relative contraindications for abscess drainage are severely compromised cardiopulmonary function and hemodynamic instability. In these situations, efforts should be made to improve clinical status to a level at which the drainage procedure can be safely performed. Lack of a safe trajectory to the abscess for optimal drainage because of overlying vital structures is another relative contraindication. However, in many of these situations, changes in the patient’s position or other maneuvers can often create a safe pathway for successful drainage.
There are several situations in which image-guided abscess drainage, although not contraindicated, should be avoided. When free hollow organ perforation occurs as evidenced by a large amount of intraperitoneal free air, an open surgical procedure should be primarily considered. Similarly, acute peritonitis is best managed by immediate surgical intervention, although in dire settings when a patient is considered unfit for surgical treatment, percutaneous catheter drainage could be performed. Symptomatic noninfected fluid collections in the vicinity of surgical implants of any type, including vascular grafts, hernia repair mesh, and joint prosthesis, should not be drained unless they are superinfected. Percutaneous catheter drainage of these noninfected fluid collections could potentially risk infection of the implants. In these situations, infection can be confirmed with diagnostic fine-needle aspiration ( Fig. 72-5 ). In addition, drainage of noninfected pelvic collections through a transvaginal route is not appropriate. Collections related to pancreatic abscess or pancreatitis are usually refractory to percutaneous drainage compared with intra-abdominal abscesses from other sources. A multidisciplinary approach is needed in these patients, that is, collaboration with endoscopic methods to facilitate resolution of the collections by placement of cystogastrostomy stents.
Role of Antibiotics
Effective management of intra-abdominal abscess needs administration of intravenous antibiotics in addition to prompt percutaneous drainage. Prophylactic antibiotic coverage is also essential before image-guided abscess drainage to prevent septic complications due to transient bacteremia resulting from the drainage of intra-abdominal abscess. Adjunctive antimicrobial therapy must be directed at enteric gram-negative rods, gram-positive cocci, and anaerobes.
Preprocedure Work-up
A comprehensive patient work-up including informed consent is indispensable before the procedure. Before abscess drainage, details including the technique and the complications and effectiveness of the procedure should be explained to the patient and the family, and written informed consent should be obtained. During the consent process, it is essential to explain to the patient and the family about the need for regular care of the drainage catheter after the procedure. It is equally important to communicate to the patient and the family that often the drainage catheters need to be retained in place at least for a few weeks before they are removed.
The necessary preliminary work-up includes laboratory evaluation of complete blood count, coagulation profile (international normalized ratio should be <1.5, activated partial thromboplastin time normal 25-35 sec, platelet count >50,000). The recent imaging studies including CT and MRI should be carefully reviewed to determine the abscess number, size, and location and the extent of involvement. In general, the yield of drainage in abdominal abscesses smaller than 2 cm is low because the diameter of the pigtail catheter is more than 2 cm. * Preprocedure imaging should be carefully reviewed to plan the drainage procedure by giving special consideration to the proposed path of the drainage catheter. Careful interrogation is also essential to avoid inadvertent catheter drainage of infected pseudoaneurysms, which can lead to catastrophic bleeding. Color Doppler ultrasound and contrast-enhanced CT or MRI are key to making this diagnosis. It is also important to carefully review the preprocedural diagnostic imaging studies to avoid bowel transgression and vascular injury. To avoid inadvertent injury to superficial vessels in the abdominal wall, such as epigastric arteries, it is crucial to review the diagnostic contrast-enhanced CT or MRI study to map the location of these vessels. In addition, one can perform color Doppler ultrasound before the percutaneous drainage to map the location of these vessels.
* References .
Catheter Selection
Wide ranges of drainage catheters are available with diameters varying from 6F to 18F. † The selection of catheter is performed on the basis of several factors, including the size of the collection and the nature and the viscosity of the fluid obtained during initial needle placement immediately before catheter placement. † In general, smaller 8F to 12F catheters can be used successfully for initial percutaneous drainage. The newer catheters have hydrophilic coating (capability of absorbing and retaining water when wet), which reduces their friction coefficient and substantially improves the ease of insertion. Catheters with an internal locking pigtail configuration are preferred for retention. Larger catheters are generally preferred in abscesses with thick and viscous abscess contents. Percutaneous drainage of complex abscesses is sometimes limited despite appropriate positioning of catheters. Catheter selection in such situations is of crucial importance as increasing catheter size and number of catheter side holes often allows effective drainage of difficult abscesses.
† References .
Patient Positioning
Planning of the patient’s position is vital to successful abscess drainage; not only does this determine a safe percutaneous path to the abscess, but optimal positioning ensures the patient’s comfort and minimizes motion during the procedure. An ideal position is one that allows the shortest and least complicated access to the abscess by avoiding vital structures such as bowel and vessels. The positioning during the procedure is often dependent on the location of the abscess and the planned percutaneous path for the drainage catheter. In most cases, successful drainage of the abscess can be performed with the patient in the supine position. The other common positions are prone, lateral decubitus, and lateral oblique positions.
Patient Preparation
Percutaneous drainage of intra-abdominal abscess is often performed in hospitalized patients but can also be done on an outpatient basis. When it is performed on an outpatient basis, it might be necessary to admit the patient for overnight observation on the basis of the patient’s clinical condition. Before the procedure, the patient should be fasting for at least 8 hours, and anticoagulant medications should be discontinued before the procedure. The patient should, however, continue other medications in the preoperative period. The procedure is mostly performed under intravenous conscious sedation, but general anesthesia should be considered in critically ill patients. Cardiorespiratory monitoring including electrocardiography, blood pressure, and pulse oximetry are crucial for monitoring of the patient during the procedure.
Percutaneous Abscess Drainage: Technique
After the details of the procedure and the associated risks are explained, the patient is placed on the procedure table in a position most optimal for a safe drainage as determined by the preprocedural imaging. During ultrasound-guided abscess drainage, a preliminary real-time scan is performed to identify the percutaneous needle path to the abscess. After the path is identified, color Doppler ultrasound should be used to determine if any intervening vessel lies in the proposed path. It is also crucial to interrogate the abscess with color Doppler to avoid placing a drainage catheter in a pseudoaneurysm, which could lead to catastrophic bleeding. Once a safe path is identified, the skin over the needle entry site is marked with a skin marker. Similarly, during CT-guided procedures, a preliminary CT scan is obtained to localize the abscess and to determine the path of the needle. Just as in ultrasound, review of the preprocedural imaging is critical to identify any vessels in the needle path. The skin entry site for the needle and the catheter are then cleansed with antiseptic solution, and the site is draped with sterile drapes to create a sterile field.
There are two basic techniques for percutaneous drainage of intra-abdominal abscesses, namely, the trocar technique and the Seldinger technique. Both these techniques are equally effective and have their distinct advantages and disadvantages ( Table 72-2 ). The choice of technique to drain a particular abscess is mostly dependent on the preference of the interventionalist and the size, shape, and location of the abscess.
| Advantages | Disadvantages | |
|---|---|---|
| Trocar technique | Allows rapid deployment of catheters Best suited for uncomplicated well-defined collections, particularly in solid organs | Difficulty of repositioning a catheter that has been initially deployed suboptimally Not preferred for complicated ill-defined collections in open spaces (retroperitoneum or mesentery) |
| Seldinger technique | Allows the ability to direct the wire to the precise location desired for catheter deployment Allows accurate placement in large complex abscesses, such as those in subphrenic locations and where access is tightly restricted | Difficulty in working in confined spaces Multiple steps involved in dilation Under CT guidance, any buckling or kinking of the wire and dilators can be a problem |
Trocar Technique
In this technique, a catheter is mounted on a metal cannula–sharp trocar system and introduced into the abscess directly or in tandem with a guiding needle. Direct trocar technique is often performed when the abscess drainage is performed under ultrasound guidance ( Fig. 72-6 ). In this technique, after the direct path to the abscess is identified, local anesthetic is administered and a skin incision is made at the planned skin entry site wide enough to accommodate the catheter. Blunt dissection is then performed at the incision site to facilitate easy passage of the catheter. The preselected drainage catheter is then mounted onto a metal stiffening cannula and inner trocar. Subsequently, the catheter loaded on the trocar and the cannula are then advanced into the abscess under direct visualization by real-time ultrasound. Once the tip of the catheter-trocar system is within the abscess, the catheter is fed off of the cannula and trocar into the abscess cavity. Pulling on the string then forms the pigtail of the catheter, and the catheter is connected to a bag for gravity drainage.
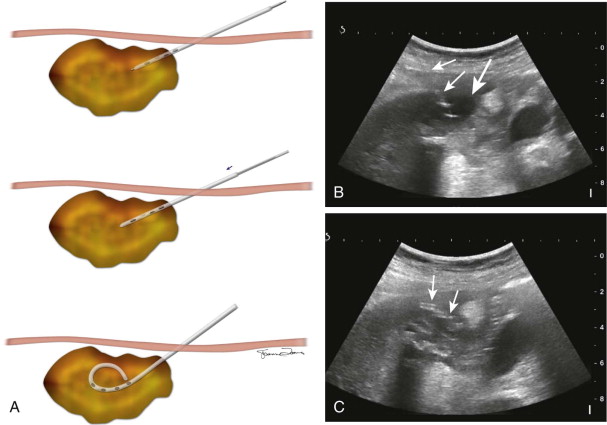
The tandem trocar technique is most often employed when the drainage catheter placement is performed under CT guidance, during which the catheter-trocar system is inserted into the abscess parallel to a guiding needle. The initial step in this technique involves placement of a guiding needle at the planned skin entry site after subcutaneous infiltration with 1% lidocaine. The guiding needle, which is usually a 20-gauge needle (Chiba biopsy needle), is slowly advanced under imaging guidance into the abscess. Accurate placement of the guiding needle is crucial to ensure safety of this technique and also permits precise deployment of the catheter in an appropriate position. Because the portion of the needle outside the body serves as a guide for subsequent catheter placement, the length of the needle should be appropriately selected such that a substantial portion of the needle extends outside the skin as the needle is firmly positioned inside the abscess ( Fig. 72-7 ). It is key to have several centimeters of the guiding needle outside the skin because the outer portion of the localizer needle directs the appropriate trajectory and angle of entry of the catheter into the abscess even if the shape of the abscess is affected by respiratory or other motion. After accurate positioning of the needle within the abscess is confirmed, the catheter is placed adjacent to the needle in tandem to it. A small incision is made in the skin adjacent to the needle, and blunt dissection is done to allow placement of the catheter. For accurate positioning of the catheter, the depth of the abscess cavity from the skin entry site is measured and appropriate marking is made on the catheter. The catheter-trocar system is then advanced alongside the needle, holding it perfectly parallel to the guiding needle to the predetermined depth. When the catheter has been advanced to the premeasured depth, feeding the catheter over the trocar-cannula system deploys the catheter. The catheter tip position within the abscess cavity is then ascertained by obtaining postprocedure CT images. If the position of the catheter is deemed satisfactory, it is secured externally and connected to a bag drainage system.











