Fig. 1
Biocontinuum of adverse early and late effects of the eye and orbit (with permissions from Rubin and Casarett 1968)
The embryogenesis determines its histogenesis:
-
Neuroectodermal outpouching of the optic cup forms the retina and optic nerve.
-
Surface ectoderm forms the eyelid epidermis and its epithelial extensions which become the conjunctival cover inside the eyelid and cover the eye globe. As in skin epithelial cells give and cover the eye globe.
-
Mesoderm forms the connective tissue and mesenchyme of eye and includes the sclera of the eye, the vasculature in retina and choroid, its arterial and venous drainage, as well as the extraocular muscles and orbital soft tissues, and boney orbit.
Many tissues compose the eye, and each varies greatly in its sensitivity to cytotoxic cancer therapy. Radiation-induced eye damage can occur following direct ocular irradiation for a malignancy of the eye, or incidentally when the eye is in the treatment field of another malignancy, such as paranasal sinus or central nervous system. Chemotherapy, biologics, and bone marrow transplantation cause ocular side effects via systemic exposures and consequential tissue effects. The chapter section begins with relevant anatomical and physiological descriptions of the ocular or adnexal structure—thereby providing a basis for the discussion on the late effects of cancer treatment on the various eye structures. The effects of radiation, chemotherapy, and biologicals are discussed along with a description of the late effects of steroids, bone marrow transplantation, and the concept of secondary cancers. Finally, the later part of the chapter describes therapeutic management and preventative measures.
A toxicity scoring system for the eye was created by the EORTC/RTOG (Rubin et al. 1995) in 1995 as a component of the combined late effects of normal tissues (LENT)/subjective, objective, management, and analytic descriptors (SOMA) grading system for radiation-induced late effects of normal tissue. As part of that effort, Gordon et al. proposed a clinically useful classification system to be used in prospective trials to evaluate the effects of radiation on the visual system (Gordon et al. 1995). Like the LENT/SOMA of other tissues, the purpose of this ocular focused grading system was to standardize and improve data recording on the radiation effects of the eye and ocular adnexa. The National Cancer Institute also created a series of grading systems that best describe chemotherapeutic effects, of which the Common Terminology Criteria of Adverse Events version 3 (CTCAE v3.0) is most recent. Ocular and adnexa tissue is one of the 35 anatomic sites and includes 21 eye-specific adverse event criteria for grading. By providing a system for standardized data, both the LENT/SOMA and CTCAE v3.0 offer a format through which to develop and compare the toxicity profiles of different cancer regimens, and to enhance future treatments (CTE 2006). Biocontinuum of adverse early and late effects are shown in Fig. 1.
2 Anatomy and Histology
2.1 Anatomy
The eye globe is the anatomic isocenter with the anterior component containing the eyelid, the lacrimal gland, and the posterior component containing the orbital content of muscles and nerves (Fig. 2).
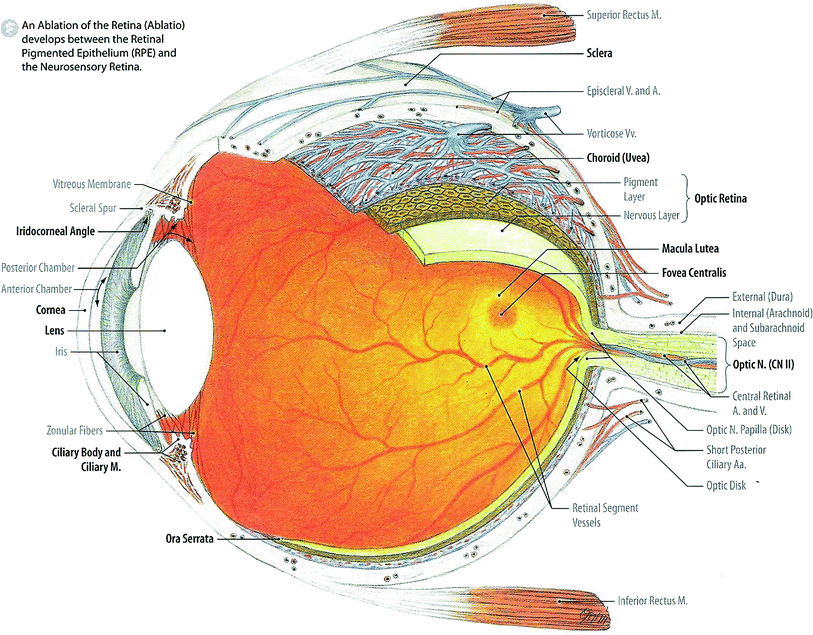

Fig. 2
Illustation (in layers) of the Retina, Choroid, and Sclera in the Outer Area: Median Sagittal Section through the Anterior Eye (with permissions from Tillman 2007)
The orbital contents consist of the globe, adipose tissue, extraocular muscles (medial, lateral, superior, inferior recti and the superior and inferior obliques). These muscles are innervated by cranial nerves III, IV and VI.
2.2 Histology
The histology of the eye can be described as follows (Fig. 3a, b, c):
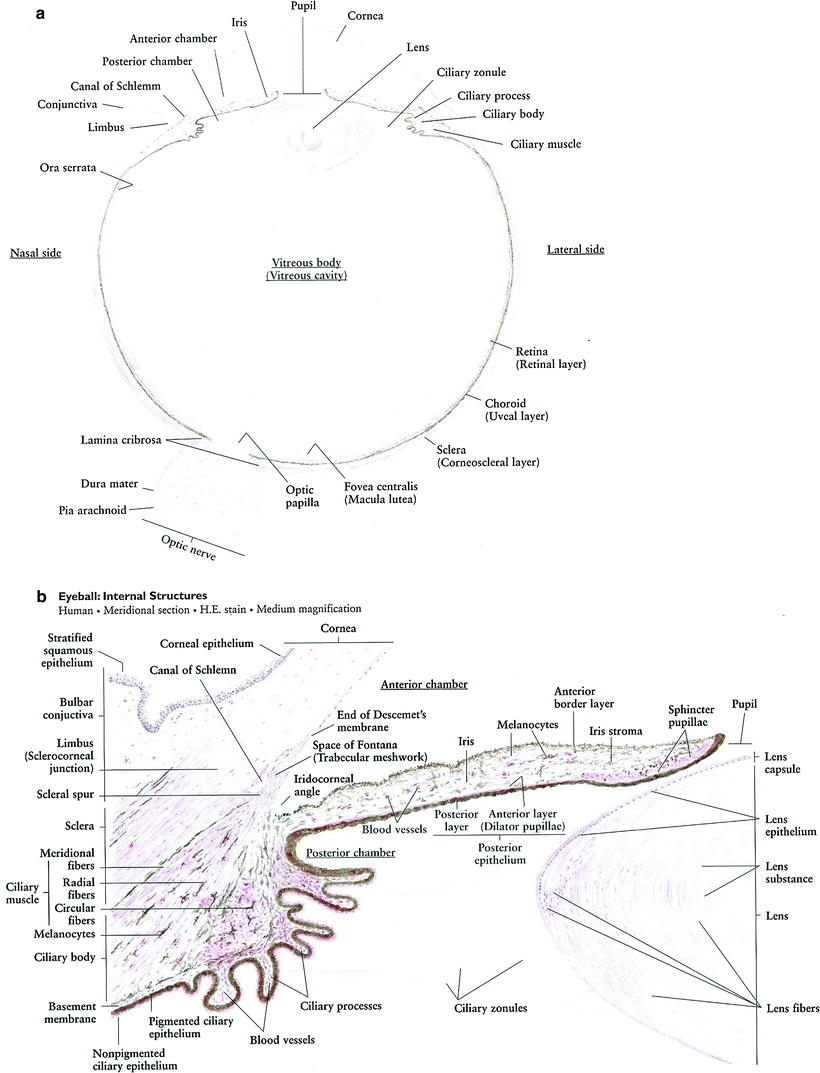
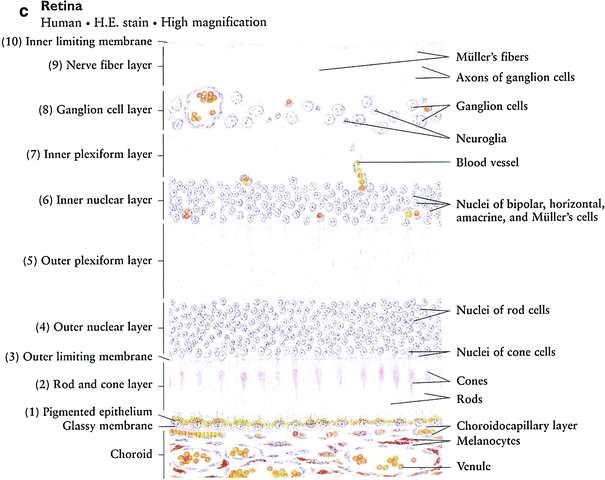


Fig. 3
a Eye globe: low magnification. b Iris and ciliary body. c Retina (with permissions from Zhang 1999)
-
The eyelid can be partitioned into the anterior and posterior lamella.The anterior lamella consists of the skin and orbicularis muscle while the posterior lamella is made up of the eyelid retractors, tarsus and conjunctiva.
-
The tear film is made up of three layers: mucous, aqueous and lipid layers. The inner mucous layer is produced by the goblet cells of the conjunctiva and helps tears adhere to the eye. The middle aqueous layer is predominantly produced by the accessory lacrimal glands, while the outer lipid layer is produced by meibomian glands and prevents evaporation.
-
The nasolacrimal system is composed of upper and lower puncta in the medial eyelids which collect tears, descend into the canaliculi, then into the lacrimal sac and the nasolacrimal duct which opens below the inferior turbinate of the nares.
-
The outer portion of the globe is composed of the sclera and cornea, while the posterior segment is lined with retina and retinal pigment epithelium (RPE). The choroid is a vascular bed that lies between the RPE and sclera.
-
Chambers of the eye consist of (i) the anterior chamber, between the cornea and iris, (ii) the posterior chamber is between the posterior surface of the iris and anterior surface and equator of the lens, and (iii) the vitreous chamber, the space between the lens and retina. The vitreous is a gelatinous substance (Fig. 3b, c).
-
Uvea contains the iris, ciliary body, and choroid with special emphasis on the canal of Schlemm which drains into episcleral venous system via minute channels through sclera. Obstruction to drainage of aqueous humor through the trabecular meshwork results in increased intraocular pressure, which may lead to optic nerve damage and glaucoma.
-
Pupil of the eye consists of 3 sets of circular muscles: outer ciliary muscle, mid-dilator pupillae, and inner sphincter pupillae innervated by 3 different nerves: parasympathetic (outer), sympathetic (mid), and V1 (inner).
3 Physiology and Pathophysiology
3.1 Physiology
The physiology of each component of the eye will be correlated with its anatomy and histology (Fig. 4a, b).
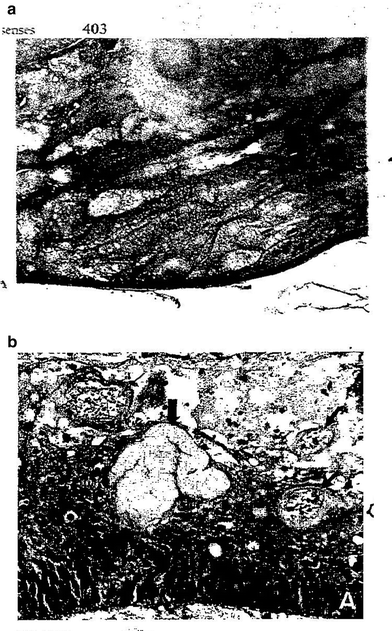

Fig. 4
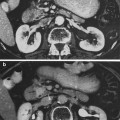
a Lens-cataract: posterior subcapsular cataract in an irradiated lens. Notice the epithelial cells (lower right) that have migrated to the posterior pole. Prominent “Morgagnian globules” derived from preexisting fibers appear in the lower center as sharply demarcated polygonal spaces of variable gray density. b Retina-atrophy: eye resected 6 months after irradiation with 60 Gy in 25 fractions over 44 days. There is marked atrophy in the innermost layers, with irregular thickening and dilation of capillaries (small arrows). An arteriole in the center (large arrow) is totally replaced by convoluted fibrillary material (mostly collagen) (with permissions from Fajardo et al. 2001)
3.1.1 Eyelids, Periorbital Skin, Lacrimal Apparatus
The thinnest skin in the body is located on the outer surface of the eyelids. It is devoid of subcutaneous fat allowing for the accumulation of fluid to manifest rapidly as swelling. The upper and lower eyelids contain fibrous connective tissue, known as the tarsal plates, which function as structural support. The eyelashes are located on the anterior portion of the eyelids and aid in protection of the eye.
The tears drain from the ocular surface via two puncta located on the medial aspect of the upper and lower lid margin. The puncta lead to the canaliculi that empty into the lacrimal sac and, in turn, into the nose via the nasolacrimal duct.
3.1.2 Conjunctiva and Sclera
The conjunctiva is a thin, transparent mucous membrane that lines both the posterior aspect of the eyelids (palpebral conjunctiva) and the anterior surface of the eye (bulbar conjunctiva). The folds between the palpebral and bulbar conjunctiva are known as the superior and inferior fornices, respectively. Tissue is redundant in the fornices to allow for adequate movement of the globe. The main lacrimal gland, which functions during reflex tearing, empties into the superior fornix, while the accessory lacrimal glands, supplying basal tear secretion, are found throughout the conjunctiva, concentrating in the fornices.
The conjunctiva contains a stratified non-keratinized epithelium overlying a stroma, known as the substantia propria. Goblet cells supplying the mucin layer of the tear film are found intermixed with the epithelial cells. The mucin produced by goblet cells contains a mucopolysaccharide which is crucial in allowing for adherence of tears to the corneal epithelium. With goblet cell damage and a lack of this mucin substance, the tears lose their adhering qualities, become unstable and contribute to surface desiccation even in the presence of adequate aqueous production. Besides acting as a physical barrier, the conjunctiva aids in host defenses by hosting immune cells as well as colonizing bacteria.
The sclera is an acellular, avascular, collagenous protective layer of the eye. It is continuous with the cornea at the limbus and covered anteriorly by the conjunctiva. The superficial coating of the sclera, known as the episclera, consists of a loose, transparent, vascular coating.
3.1.3 Lacrimal Gland, Conjunctiva, Cornea
The tear film covers the anterior surface of the conjunctiva and cornea. It serves the vital role of supplying the cornea with moisture and nutrients in the form of mucins, enzymes, immunoglobulins antimicrobial proteins, and growth factors. It also allows for the maintenance of a clear, non-keratinized epithelium in the visual axis. Furthermore, the tear film comprises the smooth outer refractive coating essential to vision by filling in corneal irregularities. The tear film consists of three layers. The aqueous layer is produced by the lacrimal gland and accessory lacrimal glands found in the conjunctiva. Meibomian glands located within the tarsal plates produce an oily layer that sits on top of and acts to stabilize the aqueous layer. The goblet cells of the conjunctiva produce the third, or mucous, layer. The overall function of the tear film is vitally dependent on each of these individual layers, and a deficiency in any layer will adversely affect the entire ocular surface.
The cornea is the transparent, avascular anterior portion of the eye that refracts and transmits light to the inner structures of the eye, while also absorbing some of the harmful UV radiation. Along with the overlying tear film, it provides approximately two-thirds of the refracting power of the eye. The conjunctiva borders the cornea in an area known as the limbus. This region contains corneal stem cells; therefore, compromising this zone leads to the loss of corneal transparency and often its integrity. The cornea is an avascular tissue and thus depends on the limbal vessels along with the tear film and aqueous fluid from the anterior chamber for nutrients and waste removal. The direct derivation of oxygen from air gives the cornea a unique characteristic compared to other body tissues. The cornea consists of five specialized layers, including, from anterior to posterior: epithelium, Bowman’s membrane, stroma, Descemet’s membrane, and endothelium. The epithelium is stratified, non-keratinized and replaces itself every 5–7 days. The stroma contains approximately 90 % of the overall corneal thickness, including a specialized superficial region known as Bowman’s membrane. Descemet’s membrane is a tough, thickened basement membrane secreted by the endothelium. The endothelial cells form a monolayer, which controls corneal hydration via ionic pumps. Small changes in corneal hydration (thickness) drastically change the optical properties of the cornea; therefore, the endothelial pumps are essential to maintain clear vision. Endothelial cells can migrate to fill a damaged area, but they do not regenerate; therefore, all loss of endothelial cells is permanent. Inflammation of the cornea, known as keratitis, may cause edema, increased corneal thickness and blurred vision.
3.1.4 Lens
The crystalline lens is a biconvex refractive structure that provides the second major refractive surface of the eye after the cornea. It is located behind the iris and pupil and is suspended circumferentially by ligaments known as zonule fibers. Anteriorly it is immersed in aqueous humor and posteriorly in vitreous humor. The encapsulated structure is devoid of blood vessels and nerves and depends on nutrients from the aqueous and vitreous humor. The lens is composed of densely packed fibers that arise from lens epithelial cells. These cells, located within the anterior periphery of the lens, undergo mitotic division at the germinative zone (the most mitotically active zone), elongate, lose their nuclei, and extend anteriorly and posteriorly to meet at the “Y” shaped suture lines in the center of the lens. The fiber cells contain specific proteins called crystallins that keep the lens transparent by inhibiting aggregation of proteins (Yanoff et al. 2004). The avascular nature of the lens prevents dispersing heat efficiently and the arrangement of the lens does not allow for removal of cells. Thus, any injured cells leave a permanent, visible defect; and like the rings of a tree, the timing of the insult is evident and irreparable.
3.1.5 Uvea: Iris, Ciliary Body, and Choroids
The uvea consists of three structures with a common embryologic origin: the iris, ciliary body, and choroid. The iris acts as the light aperture of the eye. It is a muscular membrane with a central circular opening (the pupil). Despite the wide variation in iris color on the anterior surface, the posterior surface of the normal iris characteristically contains a thick layer of heavily pigmented cells that act to absorb and thus limit the influx of light. The size of the pupil is controlled by the autonomic nervous system with input from both sympathetic and parasympathetic systems.
The ciliary body is a muscular structure located posterior to the iris and peripheral to the lens. The ciliary body produces the aqueous humor, the fluid that fills the anterior segment of the eye. This fluid drains through a structure known as the trabecular meshwork located anterior to the iris. As a result, the fluid must travel through the pupil in order to exit the eye. Any disruption to this flow will result in a backup of fluid and increased pressure within the eye, known as glaucoma. The ciliary body is also responsible for adjusting the tension on the zonule that allows for lens accommodation. The choroid, located between the retina and sclera, is the posterior segment of the uveal tract. It is a highly vascular structure that supplies the outer retina with oxygen and acts as a heat sink to the highly active photoreceptors; thereby preventing them from getting overheated when converting light energy to electrical stimuli.
3.1.6 Optic Nerve and Retina
The retina is a thin, transparent structure that functions to convert light energy into electrical stimuli for the brain to interpret. The macula, located temporal to the optic disc, is responsible for central vision and contains the highest concentration of photoreceptors. It is thought the retinal vasculature contributes roughly 5 % of the oxygen used by the retina, while the choroid provides the rest. The retinal vessels are sensitive to changes in vascular permeability which may lead to swelling of the retinal layers (i.e., macular edema). The nine layers spanning from the inner to outer aspect of the retina are histologically distinguishable, and may distort retinal pathologies in characteristic ways depending on their location.
The optic nerve contains 1,100,000 axons from the superficial layer of the retina. These axons leave the eye through an area known as the optic disc and comprise the pathway through which visual stimuli reach the brain.
3.1.7 Orbital Bones and Tissue
The orbital cavity is composed of seven bones: the maxilla, palatine, frontal, sphenoid, zygomatic, ethmoid, and lacrimal bones. They form the shape of a quadrilateral pyramid with the apex forming posteriorly and the medial walls parallel. The soft tissues of the orbit consist of the extraocular muscles, orbital fat, fascia, and vascular structures. The function of the orbital bones is to protect the eye, while the soft tissues act to cushion the eye and optic nerve during movement.
3.2 Pathophysiology
Each structure of the eye, because of its complex structure will be described separately:
3.2.1 Eyelids, Lacrimal Gland
Eyelid reaction to irradiation is similar to skin and its appendages, depending on dose/time factors. It may result in acute erythema to desquamation, loss of eyelashes, inward turning of re-grown lashes and meibomian gland disturbance. Late effects include keratoconjunctivitis sicca and squamous metaplasia of the conjunctiva.
3.2.2 Lens
The formation of cataracts is most often cited as an adverse event due to radiation. The lens is an ectodermal structure, as is the skin. The germinal epidermal cell (stratum granular) is located at the equator of the lens, migrates posteriorly, and loses its nucleus to form clear epithelial layers (stratum clear) to the lens. Radiation has been primarily associated with posterior sub-capsular cataracts owing to abnormal epithelial cells migrating posteriorly and centrally. The exact mechanism of damage remains unclear, but proposals have included free radical formation or thermal effects of radiation.
3.2.3 Uvea
Inflammation can result in synechiae or scarred attachments between the iris and anterior lens capsule. Scarring of these structures can lead to narrow angles, obstruction of trabecular meshwork outflow, elevated intraocular pressure and possibly glaucoma.
3.2.4 Retina
The mechanism of radiation retinopathy is still debated. One proposal suggests the primary site of damage is the vasculature. Endothelial cell damage with secondary loss of pericytes leads to leaky vasculature and retinal edema, while microaneurysm formation can cause intraretinal hemorrhage. Capillary loss leads to ischemia, instigating release of vasoproliferative factors and eventual neovascularization of the retina and optic disc. In very advanced cases, proliferative retinopathy can result in a tractional retinal detachment.
3.2.5 Optic Nerve
Radiation to the optic nerve may result in free-radical mediated damage to the glial cells and vascular endothelium of the white matter. With time this may result in a pale optic disc from degenerated nerve fibers.
4 Clinical Syndromes: Radiation-Induced
4.1 Eyelids, Periorbital Skin, Lacrimal Glands
Radiation can cause a number of insults to the eyelids including pigment changes, telangiectasia, madarosis (or eyelash loss), lymphedema, and architectural alterations to the eyelids and nasolacrimal system. Hyperpigmentation may follow the acute stages of erythema within a year of treatment. It is speculated that activation of angiogenic factors may contribute to telangiectasia formation (Riekki et al. 2001), which occurs at doses greater than 55 Gy, and typically at 1–5 years post treatment. In one study, lid margin epilation occurred in 20 % of patients receiving external beam radiation at doses of 60 Gy, followed by regrowth of sparse and differently pigmented cilia (Nakissa et al. 1983). In the Collaborative Ocular Melanoma Study (COMS), madarosis was the only complication that was significantly greater in those patients whom had received pre-enucleation external beam radiation therapy compared to those whom received enucleation only (COMS 1998) (Tables 1, 2, 3).
Table 1
Clinical syndromes (LENT SOMA) of the Eye-orbit
|
Eye
|
||||
|---|---|---|---|---|
|
Grade 1
|
Grade 2
|
Grade 3
|
Grade 4
|
|
|
Subjective
|
||||
|
Vision
|
Indistinct color vision
|
Blurred vision, loss of color vision
|
Severe loss of vision, symptomatic visual field defect with decrease in central vision, some ability to perform daily living activities
|
Blind, inability to perform daily living activities
|
|
Light sensitivity
|
Photophobia, no change in vision
|
Increased photophobia, decreased vision
|
Photophobia, major loss of vision
|
|
|
Pain/Dryness
|
Occasional & minimal
|
Intermittent and tolerable
|
Persistent and intense
|
Refractory and excruciating
|
|
Tearing
|
Occasional
|
Intermittent
|
Persistent
|
|
|
Objective
|
||||
|
Best corrected vision
|
>20/40
|
20/50–20/200
|
<20/200 count fingers at 1 meter
|
Cannot count fingers at 1 m
|
|
Cornea
|
Increased tearing on exam
|
Non-infectious keratitis
|
Infectious keratitis, corneal ulcer
|
Panophthalmitis, corneal scar, ulceration leading to perforation/loss of globe
|
|
Iris
|
Rubeosis only
|
Rubeosis, increased intraocular pressure
|
Neovascular glaucoma with ability to count fingers at 1 m
|
Neovascular glaucoma without ability to count fingers at 1 m, complete blindness
|
|
Sclera
|
Loss of episcleral vessels
|
≤50 % scleral thinning
|
>50 % scleral thinning
|
Scleral or periosteal graft required due to perforation
|
|
Optic nerve
|
Afferent pupillary defect with normal appearing nerve
|
≤1/4 pallor with asymptomatic visual field defect
|
>1/4 pallor or central scotoma
|
Profound optic atrophy, complete blindness
|
|
Lens
|
Asymmetric lenticular opacities, no visual loss
|
Moderate lenticular changes with mild-moderate visual loss
|
Moderate lenticular changes with severe visual loss
|
Severe lenticular changes
|
|
Retina
|
Microaneurysms, nonfoveal exudates, minor vessel attenuation, extrafoveal pigment changes
|
Cotton wool spots
|
Massive macular exudation, focal retinal detachment
|
Opaque vitreous hemorrhage, complete retinal detachment, blindness
|
|
Facial bones
|
Cosmetically undetectable facial asymmetry
|
Minimal cosmetic asymmetry
|
Moderate orbital contracture
|
Severe hypoplasia of orbital bones
|
Table 2
Detection: analytic (LENT SOMA eye-orbit)
|
Management
|
||||
|
Tearing, Cornea, Lacrimation
|
Lubrication as needed
|
Lubrication with or without pressure patch, antibiotics
|
Topical antibiotics with or without cycloplegia
|
Corneal graft, Enucleation
|
|
Pain
|
Occasional non-narcotic
|
Regular non-narcotic
|
Regular narcotic
|
Parenteral narcotics
|
|
Neovascularization
|
Pan-retinal photocoagulation for neovascular changes
|
Medical management of glaucoma, pan-retinal photocoagulation
|
Surgical management of glaucoma, Cytodestructive procedure
|
Enucleation
|
|
Lens
|
Cataract extraction depending on visual potential
|
|||
|
Retina
|
Medical management of glaucoma, focal photocoagulation
|
Surgical management of glaucoma, with or without pan-retinal photocoagulation
|
Cytodestructive procedure, repair of retinal detachment
|
|
|
Facial bones
|
Cosmetic repair ± orbital augmentation for anophthalmic socket
|
Enucleation, orbital augmentation for anophthalmic socket
|
||
|
Analytic
|
||||
|
Slit lamp exam
|
Assessment of intraocular pressure, pupils, ocular motility, dilated fundoscopic exam, and gonioscopy
|
|||
|
Cultures and stains
|
Assessment of corneal infiltrates
|
|||
|
Ultrasound
|
Examination of posterior pole if opaque media, i.e., cornea, lens, vitreous
|
|||
|
Fluorescein angiogram
|
Evaluation of retinal neovascularization, macular edema/exudates
|
|||
|
Color vision
|
Assessment if afferent pupillary defect, or optic nerve asymmetry
|
|||
|
Automated visual field
|
Bilateral-assessment of optic nerve, pupillary or color vision abnormality
|
|||
|
MRI
|
Assessment of sudden visual loss and abnormal optic disc or normal appearing optic disc and no other visible reason for visual loss
|
|||
Table 3
Examples of clinical and subclinical endpoints of radiation induced eye toxicity
|
Focal
|
Diffuse
|
|
|---|---|---|
|
Subclinical
|
Telangiectasia
|
|
|
Pigment deposition
|
–
|
|
|
Neovascularization
|
||
|
Asymptomatic keratopathy
|
||
|
Asymptomatic cataract
|
||
|
Asymptomatic glaucoma
|
||
|
Iris atrophy
|
||
|
Asymptomatic retinopathy
|
||
|
Asymptomatic optic neuropathy
|
||
|
Clinical
|
Eyelid deformities
|
Vision loss
|
|
Symptomatic keratopathy
|
Diplopia
|
|
|
Symptomatic cataract
|
Sympathetic ophthalmia
|
|
|
Uveitis
|
Endophthalmitis
|
|
|
Symptomatic glaucoma
|
||
|
Visual field defect
|
||
|
Symptomatic retinopathy
|
||
|
Symptomatic optic neuropathy
|
||
|
Boney deformities
|
A number of lid deformities may result from higher doses of radiation in the range of 60–75 Gy, such as ectropion (out-turning of lid margin) or entropion (in-turning of lid margin) (Nakissa et al. 1983). However, these complications are rare and seldom seen today. Radiation to the tarsus may result in its atrophy or contracture. Lid necrosis may develop months to years after treatment and is exacerbated by excess sun to previously irradiated tissue (Brady et al. 1989; Ober et al. 2012). Aberrations such as these can impair the eyelids’ ability to adequately cover and maintain the integrity of the ocular surface, leading to its possible compromise (see next section on ocular surface).
Destruction or occlusion of the puncta may occur when the medial portions of the eyelid are irradiated. Radiation induced canaliculitis leads to fibrosis and obstruction, typically to the lateral one-third of the nasolacrimal system (Buatois et al. 1996). Stenosis or obstruction along any part of the nasolacrimal ductal system can cause impaired tear drainage and symptoms of epiphora.
Lacrimal gland dysfunction can lead to a dry eye and associated chronic symptoms such as pain, and may result in corneal ulceration and visual loss. A series of analyses from the University of Florida and elsewhere relate the incidence of dry eye to the dose of radiation delivered to the lacrimal gland. Dry eye is unusual at doses <30 Gy, is occasionally seen at slightly higher doses, is very common following doses >40–45 Gy, and can be severe at doses >57 Gy (doses usually delivered at 1.8–2.0 Gy per fraction). The interval between RT and the onset of dry eye can be years following modest doses (e.g., >4 years after 30–40 Gy), but are often shorter at higher dose levels (e.g. 9–10 months after >57 Gy) (Parsons et al. 1994, 1996; Jeganathan et al. 2011).
4.2 Conjunctiva and Sclera
Radiation effects to the conjunctiva may occur via direct damage or through secondary effects on the ocular surface. At 1–2 years post-treatment, prolonged conjunctival injection may develop, particularly at exposures of 30–50 Gy. Also at doses of 30 Gy, telangiectatic vessels may follow at 3–6 years. With minor trauma or valsalva efforts, the fragility of these vessels may result in their rupture and accumulation of subconjunctival hemorrhage (Fig. 5).
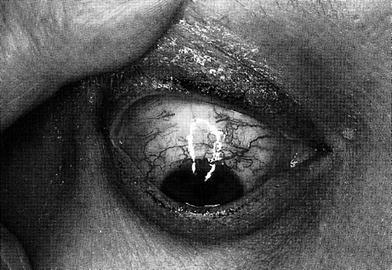

Fig. 5
Clinical syndromes: telangiectasia of conjunctival blood vessels
At radiation doses over 50 Gy, keratinization of the epithelium has been observed (Gordon et al. 1995). These eyes should be evaluated for cornea irritation from adjacent keratin plaques. In severe cases and exposures of 60 Gy, chronic conjunctival ulceration may develop. Feared complications include symblepharon formation (adhesions between the bulbar and palpebral conjunctiva following denuded epithelium) and subsequent forniceal shortening, trichiasis (inward turning of the lashes on the ocular surface) and other eyelid malpositions. Conjunctival necrosis has also been observed in retinoblastoma patients when radioactive plaque therapy provided conjunctiva doses of 90–300 Gy (Haik et al. 1983; Brady et al. 1989; Ober et al. 2012; Donnenfeld et al. 1993). Loss of goblet cells may occur and exacerbate dry eye symptoms through the subsequent dearth of mucin.
The avascular property of sclera renders it relatively radioresistant. For example, the sclera is able to tolerate doses of radiation up to 900 Gy from an iodine or cobalt plaque when administered over 4–7 days. However, scleral atrophy and necrosis has been documented in irradiated eyes, and is most common following the use of brachytherapy. In fact, scleral atrophy is a dose-limiting factor in ocular plaque dosing. Fractionated doses of 20–30 Gy can cause thinning, melting or atrophy of the sclera, while its perforation is a rare complication (Brady et al. 1989). Sclera atrophy should be monitored for infection, corneoscleritis (concomitant inflammation of the sclera and cornea) and perforation.
Pigmented deposits on the episclera have been reported following brachytherapy for uveal melanoma. They developed within 6 months of treatment, occurred in 85 % of patients at 1 year and their quantity was associated with proximity to the tumor or irradiated area (Toivonen and Kivelä 2006).
4.3 Lacrimal Gland, Conjunctiva, Cornea
The health of the ocular surface relies greatly upon the tear film, which is generated by a careful balancing of three components: the aqueous, mucin, and lipid layers. Deficiencies in any one of these may result in a compromised tear film and risk the maintenance of the ocular surface. Radiation can disrupt many of the key structures that produce these tear film elements, and place the eye at risk for ocular surface damage (or dry eyes) via a number of mechanisms. For example, radiation induced atrophy of the lacrimal gland, which occurs at 50–60 Gy, causes a decrease in aqueous tear substance. Furthermore, atrophy of meibomian glands at doses less than 30 Gy (Roth et al. 1976) and damage to conjunctival goblet cells (which also have low radiation tolerance) cause reductions in the lipid and mucin constituent of tears, respectively. The subsequent instability of the tear film and its surface tension results in an evaporative dry state. When all of the three tear film constituents are compromised by radiation, it is inevitable that a dry eye state will follow. The frequency of radiation-induced dry eye is dose-dependent and most likely at doses greater than 40 Gy (Parsons et al. 1994, 1996). However, while changes are evident within 9–10 months at higher doses (>57 Gy), it is suggested that low doses (30–45 Gy) cause later-onset ocular surface disease at an estimated 4–11 years post-treatment (Durkin et al. 2007). Symptoms of keratoconjunctivitis sicca (KCS), or dry eyes, include foreign body sensation, burning, photophobia and blurry vision. High dose radioiodine used in the treatment of thyroid carcinomas results in reduction of lacrimal gland tears; however, the subsequent association to dry eye symptoms is unclear (Fard-Esfahani et al. 2007).
The anterior surface of the cornea can likewise be affected by a number of radiation-induced pathologies. The tear film baths the cornea but when altered by radiation, can lead to an epitheliopathy. Radiation-induced impairment of the fifth cranial nerve causes decreased corneal sensation and neurotrophic harm, while decreased blink mechanism and exacerbation of surface changes can result from aberrations of the seventh cranial nerve. Furthermore, radiation can damage the corneal stem cells located at the limbus (the transition between cornea and conjunctiva along the peripheral edge of the cornea), thereby injuring the cornea’s source of epithelial turnover (Dua et al. 2003). This may cause chronic epithelial defects or filamentary keratitis, which share the symptoms of irritation, photophobia, pain, and epiphora. Without an intact epithelium, the underlying stroma is at risk for damage. In fact, either epithelial or endothelial dysfunction can cause stromal edema. In addition, stromal ulceration results from direct radiation damage to the stromal keratocytes at doses of greater than 60 Gy (Nakissa et al. 1983). Both epithelial and stromal defects can elicit vision-limiting neovascularization and opacification. Years after treatment, keratinization and even lipid deposition in the stroma can occur, both of which further compromise vision. A nonhealing ulcer can be a treatment dilemma and requires close observation for panophthalmitis and perforation.
Without an adequate tear film, the ocular surface has limited contact to the accompanying nourishment, lubrication, immunoglobulins, and enzymes. This may increase the cornea’s susceptibility to colonization or microbial invasion and thereby accelerate ulceration and perforation (Haik et al. 1983; Brady et al. 1989; Donnenfeld et al. 1993; Barabino et al. 2005; Blondi 1958).
4.4 Lens
Cataract is a well known complication following radiation to the eye. A cataract is a loss of optical clarity within the lens. It is usually attributed to a disruption of the regular structure of the lens fibers. Radiation has been primarily associated with posterior subcapsular cataracts secondary to the abnormal epithelial cells migrating posteriorly and centrally. Damage to the radiosensitive germinative epithelium is likely responsible for most post radiation cataracts. The exact mechanism of damage remains unclear. However, evidence exists for free radical formation causing DNA and cell membrane damage and mitotic arrest. Thermal effects of radiation are also likely to contribute to injury of lens fiber cells secondary to the heat being inefficiently dissipated (Fig. 6).
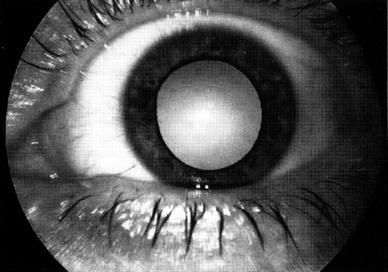

Fig. 6
Clinical syndromes: cataract
An association between cataract formation and exposure to ionizing radiation was first recognized in 1897. In humans an increased incidence of cataracts was noted in nuclear plant workers, survivors of atomic bombs, and later in patients treated with local radiation for head and neck cancers (Abelson and Kruger 1949; Cogan et al. 1949; Merriam and Focht 1957). Cataract formation ranges from a latency period of 6 months to over 30 years with an approximate average of 2–3 years. The development of lens opacity is heavily influenced by patient age, the method of delivery, size and location of the tumor, and total dose, fractionation, energy of the source, and dose rate of radiation.
In patients with choroidal melanoma, tumor size is a risk factor for the development of cataracts with either brachytherapy or external beam radiation. Tumor basal diameter greater than 10–15 mm (Summanen et al. 1996), and height greater than 4–6 mm (Summanen et al. 1996; Beitler et al. 1990; Gunduz et al. 1999a, b, c; Gragoudas et al. 1995), are strong predictors of developing radiation cataracts. Tumor height, which is believed to be a good approximation of tumor volume, has been found to be an independent risk factor and associated with time to cataract formation (Summanen et al. 1996; Kleineidam et al. 1993; Puusaari et al. 2004a). This could be partially secondary to elevated tumors undergoing more necrosis contributing to increased intraocular inflammation, which is known to cause cataracts.
Tumor location also influences cataract formation. It is well accepted that anterior tumors are more likely to develop cataracts after brachytherapy than posterior tumors, secondary to the difference in radiation dose to the lens. For example, one study found cataract rates to be as high as 85 % in anterior tumors treated with Pd103 brachytherapy compared to 17 % for posterior tumors (Finger 1997). Additionally, there is a lower rate and longer time to onset of cataract formation for tumors located posterior to the equator (Fontanesi et al. 1993; Summanen et al. 1996). Cataracts have been noted after an average of 11 months in patients with anterior, iris, or ciliary body lesions compared to 26 months in patients with posterior based lesions (Summanen et al. 1996).
Several studies have investigated the minimal amount of radiation that would induce cataract development. Merriam and Foch, the first to describe the relationship between radiation dose and cataract formation, suggested that the amount of radiation delivered at a single period may be as important as the total dose (Merriam and Focht 1957). A single dose of 2 Gy or a cumulative dose of 5.5 Gy given over longer than 3 months was enough to induce cataract formation. Subsequent studies have reported a slightly higher threshold of single dose of 5 Gy or lower (Henk et al. 1993). In brachytherapy, eyes treated with doses less than 12 Gy of iodine 125 have a significantly lower rate of cataract and cataract surgery compared with doses greater than 24 Gy (COMS 2007). Additionally, for each 10 Gy increase of iodine 125 to the center and posterior pole of the lens, the rate of cataract formation has been reported to increase around 15 % (Puusaari et al. 2004b).
The effect of fractionation can influence cataract incidence. For example, Benyunes et al. reported 85 % of their patients developing cataracts when exposed to a single dose of 10 Gy versus 34–50 % when given fractionated doses of 2 Gy even to a higher total dose of 12 Gy or greater (Benyunes et al. 1995). Fractionation is also associated with delaying the development of cataracts, the severity of lens opacification, and the need for cataract surgery (Tichelli et al. 1993; Aristei et al. 2002; Benyunes et al. 1995). In addition, hyperfractionation with greater than 6 fractions can decrease the incidence of cataracts (Belkacemi et al. 1998).
4.5 Uvea: Iris, Ciliary body and Choroid
Iris neovascularization, or rubeosis iridis, occurs several months to years following RT, particularly with fractionated doses of 70–80 Gy over 6–8 weeks. Ocular ischemia instigates new, abnormal vessel formation through the release of vascular growth factors from ischemic retina, irradiated tumor tissue or direct damage to iris vessels. Without abatement, it is feared these vessels will grow into the anterior-chamber angle accompanied by myofibroblasts. The latter contract, form fibrous adhesions within the angle, and thereby obstruct aqueous outflow and increase pressure in the eye. High intraocular pressure may cause damage to the optic nerve, resulting in neovascular glaucoma, which is thought to occur in 35 % patients treated with brachytherapy (Shields et al. 2003). In plaque brachytherapy treatment, both iris neovascularization and glaucoma may be influenced by involvement of the anterior tumor with the iris, and with high intraocular pressures at diagnosis (Gunduz et al. 1999a, b, c; Puusaari et al. 2004a, b). With helium-ion irradiation in uveal melanoma patients, Daftari et al. found development of neovascular glaucoma correlated with amount of lens and anterior chamber exposed, tumor volume, proximity to the fovea, history of diabetes, and development of vitreous hemorrhage (Daftari et al. 1997).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree