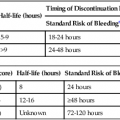
Infection following any procedure that violates the skin’s protective barrier is well known throughout all medical disciplines and contributes to significant morbidity, not to mention increased healthcare costs. Despite the universalization of aseptic technique and the expansive variety of antimicrobial drugs at our disposal, infection remains a frequent complication encountered by the interventional radiologist. In the recent past, numerous authors, including the Society of Interventional Radiology via their Practice Guidelines statement,1 have performed comprehensive reviews of the current literature in an attempt to summarize and condense the body of data available. What these documents have revealed is that the practices currently in place at the majority of institutions have roots founded and supported primarily by the surgical literature.2–5 Additionally noted is that despite the vastly prevailing opinion that antibiotic prophylaxis has become the standard of care, very little substantiation in the form of randomized controlled trials can be found. The issue of appropriate periprocedural antibiotic prophylaxis will certainly continue to arise as the breadth of procedures provided by interventional radiology expands. For all procedures, patient-specific (age, weight, allergies, renal and hepatic function) and community-specific (formulary selection, local resistance) factors, in addition to weighting of the overall risks and benefits of antibiotic prophylaxis, must be considered. Consultation with appropriate specialists (e.g., pharmacy, infectious disease service) are highly encouraged when any questions or inconsistencies arise. It is important to note that the whole of medicine, not excluding the surgical or interventional radiology community, has in the recent past begun to reevaluate the role of periprocedural antibiotic prophylaxis as emerging issues such as resistance and cost-driven delivery of health care move to the forefront.5–7 Ultimately, the responsibility lies with the interventional radiologist, who must conclude whether prophylactic antibiotic use is appropriate, identify the best antimicrobial agent, and oversee timely delivery. Many factors impact whether a patient is at elevated risk for periprocedural infection, and some aspects may not be resolved clinically (i.e., comorbidities, bacteremia) prior to the necessary performance of an intervention. However, preventive measures such as meticulous attention to sterile draping, handwashing, and sterile technique remain crucial to infection control in the interventional suite. Although universal application of these precautions should be independent of the decision for or against prophylactic antibiotic use, the National Academy of Sciences/National Research Council’s surgical procedures classification system can be applied in the interventional suite to guide use of prophylactic antimicrobial agents.8 It is known that infection rates associated with interventional procedures differ from surgical counterparts,9 but the characterization of a procedure as clean, clean-contaminated, contaminated, or dirty can predict infection risk associated with a given anatomic location. Importantly, these groupings take into consideration both the general health of tissues within the operative field and successful execution of aseptic technique for the duration of the procedure. Clean procedures carry a 5% risk of infectious complication. These interventions maintain sterile technique in tissue with no obvious inflammation, and do not involve anatomic structures with potential bacterial colonization/infection (e.g., genitourinary [GU], gastrointestinal [GI], respiratory tract).3,8 Clean-contaminated procedures convey a 10% risk of periprocedural infection. In this group, the GU, GI, or biliary tract may be entered, but inflammation is clearly absent at the time of the procedure, and aseptic techniques are maintained throughout.3,8 Any major break in sterile technique results in a contaminated procedure, as does entry into obviously inflamed tissues,3,8 save for those harboring purulent material. A contaminated intervention’s risk of infection doubles that of clean-contaminated, at approximately 20%. Procedures with clearly infected material, including clinically infected GU or biliary systems or a GI perforation, make up the final conglomerate, dirty procedures. It is predicted that nearly 40% of dirty procedures result in infectious periprocedural complications.3,8 Routine prophylactic antimicrobial agents are currently recommended for all procedures except those considered clean. Institution-specific formulary geographic bacterial resistance tendencies, as well as expense, should be considered prior to selection of an appropriate agent.1,5,9 Efficacious antibiotic prophylaxis has been shown to be a time- and dose-sensitive practice. A clearly defined window spanning from 1 hour prior to the procedure up to the time of initial incision has been validated by the surgical literature. A delay in administrating the prophylactic agent may increase the risk of periprocedural infectious complication by up to a factor of 5,2,10,11 and several studies provide evidence that antimicrobial therapy initiated postoperatively is equivalent to no prophylaxis.2,10 At our institution, we wait until the patient is in the interventional suite to initiate antibiotic prophylaxis. Providing prophylaxis via a reliable route (i.e., intravenously [IV]) and in adherence with temporal guidelines ensures that adequate serum antibiotic concentrations are achieved for maximal inhibitory effect.12 Several studies have disproved the previously held perception of superior efficacy of multidose prophylaxis regimens; a solitary dose achieves adequate prophylactic effect.5,13,14 Important exceptions to single-dose prophylaxis include: (1) prolonged procedures lasting longer than 2 hours and (2) intervention on an obstructed structure such as the biliary tree, GU system, or other similar viscus. An auxiliary dose of the prophylactic agent should be administered under the direction of the interventionist for procedures exceeding 2 hours, taking into account the pharmacokinetics of the specific agent used.5 In patients with a known obstructed system, the risk of bacterial translocation and inoculation of the lymphatic or vascular system, with resultant bacteremia or sepsis, is increased.2,15 Therefore, therapeutic doses of antimicrobial agents should be continued for at least 48 hours following intervention or until adequate drainage has been achieved.5 Particularly in the inpatient setting, it is not uncommon for the referring service to manage antibiotic administration in the immediate postprocedural period. Prompt communication of the necessity for continued treatment must be clearly provided by the interventionist and his/her team to ensure sustained therapy. Similar to other procedural subspecialties, interventional radiology has realized a continuous increase in the overall volume of cases, particularly the placement of invasive devices. Concurrently, infectious periprocedural complications have followed a similar upward-sloping trend.5 One would expect that, all other variables remaining constant, these tendencies would abide to a linear plot. It is striking then that the trends are not collinear, a finding that can be greatly attributed to the emergence of a variety of antibiotic-resistant microbial strains including methicillin-resistant Staphylococcus aureus (MRSA), vancomycin-resistant enterococci (VRE), and β-lactamase–producing gram-negative rods.16 Resistance may be conferred between bacteria by horizontal gene transmission, known as conjugation, or by spontaneous mutation. These impressive microbial strategies are driven by the most clinically relevant contributor to the proliferation of resistant strains: inappropriate, nonspecific use of broad-spectrum antibiotics17 in both healthy and immunocompromised patients.5 The latest generation of antibiotics for treatment of VRE species includes daptomycin, quinupristin-dalfopristin, and linezolid. These agents specifically attack the permeability of the bacterial membrane, or synthesis of protein/peptidoglycans integral to proper membrane function.18 As the battle to develop newer antimicrobial agents continues, a refocusing of the medical community’s efforts towards improved preventive techniques targeted at these dangerous strains and their mode of transmission is necessary. When a prophylactic antimicrobial agent is selected, special consideration of the most likely pathogen and source should guide the interventionist to not only prevent patient morbidity and mortality but also deter further contribution to antibiotic resistance. Complacency in the prevention of horizontal transmission and appropriate therapeutic selection today will inevitably result in a greater number of resistant organisms in the future.19 All too frequently, one is faced with the patient reporting an antibiotic allergy. It is known that many of these self-reported reactions do not represent a true immune-mediated reaction. Anaphylactic reactions are rare and include hives, bronchospasm, hypotension, and laryngospasm. An accurate detailed history should be obtained to determine the true nature of the complaint. The true incidence of hypersensitivity with the penicillins and other related β-lactams is near 2%. Cephalosporin cross-reactivity is a concern but occurs in only 15% of patients with previous adverse reaction to the penicillins. As a result, penicillins and cephalosporins should be withheld in patients with documentation of severe reaction. Instead, antimicrobials lacking the β-lactam fundamental structure, such as vancomycin, should be used,5 with the monobactams and carbapenems other potential choices. Patients reporting minor reactions such as fever and/or a typical maculopapular rash can safely receive β-lactams without concern for anaphylactoid reaction.20 Antimicrobial agents achieve their desired toxicity by exploiting several mechanisms of bacterial homeostasis. Cell wall–specific agents include the penicillins, cephalosporins, and vancomycin (wall synthesis), in addition to amphotericin, polymyxin, and daptomycin (wall permeability). Protein synthesis is targeted by the aminoglycosides, macrolides, tetracycline, and linezolid, while nucleic acid synthesis is inhibited by the sulfonamides and quinolones. Even when correctly selected for the most likely bacteria, delivery of the agent must result in serum and tissue concentrations that exceed a minimum inhibitory concentration (MIC) of the organism so that effective interference with the desired process can occur.12 Penicillins are very well tolerated, readily available, economical, and have a low incidence of hypersensitivity. Primarily renal excretion demands heightened awareness in patients with known renal insufficiency where accumulation can occur. Appropriate alteration in dose amount and/or frequency (“renal dosing”) should be employed for these patients, especially if multiple doses are anticipated.9 These agents provide excellent coverage of gram-positive microbes and can be delivered in conjunction with β-lactamase inhibitors such as clavulanic acid (Augmentin, Timentin), sulbactam (Unasyn), and tazobactam (Zosyn) to counteract resistance conferred by the β-lactamase protein. Penicillins resistant to degradation by β-lactamase are also available (methicillin, oxacillin, cloxacillin, dicloxacillin, nafcillin), and can be used when culture and sensitivity indicate Staphylococcus spp. expressing the β-lactamase gene. Surgical antibiotic prophylactic regimens use the cephalosporins more frequently than any other agents, and interventional radiology use is similar. Like their penicillin relatives, with whom they share a similar β-lactam fundamental structure, cephalosporins are well tolerated. There is versatility among the agents within this family, allowing for use across a variety of interventional procedures, save for those where Enterococcus species are anticipated. Cefazolin (Ancef) provides excellent coverage for a large proportion of the microbes encountered, while being economical and readily available. The third-generation cephalosporin ceftriaxone (Rocephin) achieves excellent penetration into the biliary tree owing to its biliary mode of excretion. It is frequently used when violation of the bile ducts is intentional or a potential side effect.3 If expansion of the spectrum of antimicrobial prophylaxis to include gram-negative organisms is desired, the addition of aminoglycosides should be considered as adjunctive agents. By altering the permeability of the bacterial membrane through inhibition of protein synthesis, excellent synergism can be achieved when combined with the β-lactams.3 However, in patients receiving or potentially receiving vancomycin, nephrotoxicity and ototoxicity are possible and should be closely evaluated during combined regimens. Excretion is primarily renal, and this has resulted in adoption of aminoglycosides as the primary or secondary choice for prophylaxis in the GU system, along with the cephalosporins. Guidance as to appropriate dosage in the patient with renal insufficiency should be sought from infectious disease or pharmacy consultation. While also low in cost, there are limitations and disadvantages to aminoglycoside use. It is known that purulent collections harboring low-pH environments (i.e., abscesses) hinder the activity of this class of medications. Also, as effective as aminoglycosides are at expanding the antimicrobial spectrum of other agents, they are hindered by their own intrinsic narrow breadth of coverage so are rarely used as solitary therapy. TABLE e24-1 Antibiotic Prophylaxis Recommendations for Interventional Radiology Procedures
Antibiotic Prophylaxis in Interventional Radiology
Introduction
Antibiotic Prophylaxis
Antibiotic Resistance
Antimicrobial Hypersensitivity
Antibiotic Agents
Penicillins
Cephalosporins
Aminoglycosides
Procedure-Specific Antibiotic Prophylaxis
Procedure
Recommended Drug
Suspected Organism(s)
Central venous access, implantable
None
Staphylococcus aureus, Staphylococcus epidermidis
Central venous access, implantable (immunocompromised patient)
Cefazolin 1 g IV
S. aureus, S. epidermidis
IVC filter placement
None
S. aureus, S. epidermidis
Superficial venous insufficiency
None
S. aureus, S. epidermidis
Angiography, angioplasty, atherectomy, stent and caval filter placement
None
None
Arterial stent-graft placement (aortic, iliac, superficial femoral)
Cefazolin 1 g IV
S. aureus, S. epidermidis
PTC/PBD
Ceftriaxone 1 g IV, or ampicillin/sulbactam 1.5-3 g IV, or ampicillin 2 g IV + gentamicin 1.5 mg/kg IV
Escherichia coli, Klebsiella, Enterobacter, Enterococcus, Pseudomonas, Clostridium
TIPS
Ceftriaxone 1 g IV, or ampicillin/sulbactam 1.5-3 g IV
Antegrade pyelography and PCN without clinical infection
Cefazolin 1 g IV, or ceftriaxone 1 g IV
None
Antegrade pyelography and PCN with clinical infection
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree

 Get Clinical Tree app for offline access
Get Clinical Tree app for offline access