Positron emission tomograph (PET)-magnetic resonance (MR) is a new modality combining PET and MR. In gynecologic cancers it can be used for staging of cervical and endometrial cancer, planning of radiation therapy in cervical cancer, assessing response to chemotherapy in ovarian cancer, and detection of recurrence in most gynecologic cancers. It is being explored for prostate cancer and other genitourinary cancers, but is still in experimental stages.
Key points
- •
PET-magnetic resonance (MR) combines the soft tissue contrast of MR with the functional imaging of PET.
- •
PET-MR uses the sensitivity of PET for nodes together with the local soft tissue assessment of MR.
- •
PET-MR is useful for initial staging of cervical and endometrial cancer, planning of radiation therapy in cervical cancer, and assessing response to chemotherapy in ovarian cancer.
- •
PET-MR can assess recurrence in most gynecologic cancers.
- •
The soft tissue contrast of MR can disambiguate many small PET lesions for a variety of applications.
Applications of PET/MR imaging in gynecologic cancers
Introduction
Cancers of the female gynecologic tract are fairly common. Together, the 3 most common cancers, epithelial ovarian cancer, cervical cancer, and endometrial cancer, will account for an estimated 95,320 cases and 28,830 deaths in 2016, together accounting for 5.7% of all new cases and 4.9% of deaths. The cancers vary in their frequency and severity; endometrial cancer is the most common, whereas ovarian cancer is the most lethal. The relative clinical course of the cancers is affected by their stage at diagnosis: 82% of cervical and 88% of endometrial cancers are diagnosed at the local or regional stage with relatively favorable prognosis, whereas only 34% of ovarian cancers are detected at an early stage and portend a relatively poor prognosis. The authors aim to describe the role of PET/magnetic resonance (MR) in staging, response assessment, and detection of recurrence of gynecologic cancers. The authors also discuss early developments in the role of PET/MR in selected genitourinary cancers.
What Is PET/Magnetic Resonance?
PET/MR is a hybrid imaging modality composed of PET and MR, in a similar fashion to the earlier combination of PET and computed tomography (CT) (vis-a-vis PET/CT, which has now replaced stand-alone PET). The aim is to combine the exquisite soft tissue contrast, adaptable field of view, and functional information of MR, widely in use in structures with a variety of soft tissues in a small space, such as the brain, joints, and the female pelvis, with the physiologic imaging property of whole-body PET, which is widely used for staging, response assessment, and detection of recurrence of a wide variety of cancers. The authors principally discuss the use of PET with fluorodeoxyglucose (FDG), a radiotracer that assesses glucose metabolism that is ubiquitously used in cancer imaging. Although other tracers are sometimes used, they are now mostly in a developmental stage; an important exception is the use of C11-choline PET for prostate cancer, which is discussed in that section.
Integrated PET/MR scanners, which obtain PET and MR images simultaneously or sequentially, have been available in the United States for several years now and in many practices are important emerging clinical and investigative tools. These resources obtain anatomic, functional, and metabolic data from using both modalities in one scan session, with sensitivity and specificity that is much higher than that of a conventional CT scan, at greater safety (ie, much lower cumulative ionizing radiation), and often fewer artifacts. Advances in PET imaging with improvement in detector sensitivity and advent of various novel radiotracers, as well as evolution of ultrafast high-resolution MR sequences and more patient-friendly hardware, simultaneous PET/MR scans are poised to be game changers in the management of gynecologic and genitourinary malignancies.
What Is the Evidence for, and Ongoing Work on, PET/Magnetic Resonance?
With its low ionizing radiation and an adaptable field of view (FOV), PET-MR theoretically seems advantageous; but as it is a relatively new technique, there is relatively little published in medical literature validating its superiority to separately obtaining FDG PET-CT and MR. With that said, PET/MR has been shown to be noninferior to FDG PET-CT and MR imaging obtained separately, for various malignancies, mostly for local and regional staging.
For gynecologic cancers, much of the work has been done by Grueneisen and colleagues at the University Hospital Essen in Duisburg-Essen, Germany. This group, in a study of 44 patients with cervical or ovarian cancer, found improvement of PET/MR over MR alone for detection of recurrence, identifying 99% versus 89% of lesions (and 100% vs 92% of patients with recurrence). Another study of 19 patients with recurrent ovarian or cervical cancer by Beiderwellen and colleagues compared PET/MR with PET/CT and found that both modalities showed all lesions, including 1 patient’s lung metastasis, although there was some improvement in conspicuity of metastases with PET/MR. A similar study of 24 patients with a suspected recurrence of a pelvic malignancy showed no significant difference between the two approaches on a lesion-by-lesion basis. Another study by the same group, analyzing 27 patients with cervical cancer at initial staging, showed 85% correct T staging and 91% sensitivity and 94% specificity with PET/MR N staging; however, the study did not take into consideration the performance of conventional modalities. There were 5 patients with distant metastases to inguinal and para-aortic nodes, all of which were detected by PET/MR. A fourth study by this group was of 48 patients, with a mix of pelvic malignancies, either primary or recurrent, in which PET/MR showed sensitivity and specificity of 93% to 95% and 83% to 87%. The group also found that diffusion-weighted imaging (DWI) offered no incremental value greater than PET/MR using conventional imaging sequences. Finally, a study of 26 patients with a mix of advanced gynecologic cancers by Queiroz and colleagues showed that PET/MR was better at T staging (specifically in the area of primary tumor delineation) than PET/CT, particularly for cervical and endometrial cancers; however, this study did not explore its utility for N or M staging.
Of course, to justify the return on investment of a new resource, many studies have compared results of an integrated PET-MR imaging with results of FDG PET-CT and MR imaging obtained separately. Kitajima and colleagues, in a study of 30 patients with endometrial cancer, retrospectively fused PET-CT and MR and found the result was more sensitive than MR alone and was similar in sensitivity and specificity to PET/CT. Another study by Kitajima and colleagues looked at retrospective fusion of PET/CT and MR studies in 35 patients with cervical cancer, whereby fused PET/MR gained the accuracy of MR (83%) for T staging (more than 50% for PET-CT) and gained the sensitivity of PET-CT (92%, more than 69% for MR) for N staging. Kim and colleagues retrospectively fused PET/CT and MR in 79 patients with cervical cancer in a wide variety of stages and found an increase in sensitivity for nodes (from 44% to 54%). (Of note, neither article looked at lung metastases, which are a known weakness of MR and, thus, PET/MR and which would, therefore, remain an argument for separately performing PET-CT and MR.) Fiaschetti and colleagues retrospectively fused pelvic MRs and PET-CTs of 24 patients with adnexal lesions and found increase in negative predictive value to 83% from 44% for the source PET-CT and 50% for source MR images (sensitivity increased slightly as well). Retrospective fusion may, thus, serve as an alternative for institutions unable to purchase a PET/MR machine, although concerns about misregistration remain as the examinations are performed at different times. What seems apparent is that the incremental value of an integrated simultaneous PET-MR imaging resource comes from its substantial reduction of ionizing radiation and patient convenience of completing 2 advance modalities in one encounter.
Finally, another principal concern with MR and, thus, PET/MR is chest (in particular lung) metastases. In hybrid imaging, PET’s poor spatial resolution is compensated for by the overlaid anatomic imaging modality; but MR has poorer spatial resolution in the chest (where motion is a major concern) compared with CT. Because a PET/MR sacrifices the CT, there is the concern that lung metastases may be missed, as PET/MR shows decreased detectability for nodules less than 1 cm, particularly less than 5 mm. However, as there is some question as to how important these small nodules actually are, avoiding detection of clinically irrelevant incidental nodules may actually be beneficial.
How Is PET/Magnetic Resonance Done? Protocolling PET/Magnetic Resonance and Other Issues
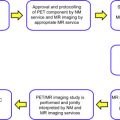
In gynecologic malignancies, 18 F-FDG PET/MR protocols are intended to provide one-stop shopping for treatment planning. The examination assesses the locoregional extent of pelvic tumor and evaluates the whole body (WB) for nodal, peritoneal, extrapelvic visceral, and bone metastases.
A variety of MR protocols are described in the literature. The authors present experience with the simultaneous integrated PET/MR scanner (Biograph mMR; Siemens, Erlangen, Germany) in which advanced, ferromagnetic-friendly PET photodetectors are embedded inside the MR scanner into a single housing. This setup allows the concurrent imaging of the pelvis. Alternately, the sequential Pet/MR scanner construct (eg, Ingenuity TF, Phillips, Cleveland, OH) joins an MR scanner with a PET scanner by a movable table between the two resources. Either setup allows the acquisition of MR and PET data in a single encounter or session and for analysis of fused images.
Now the MR-based photon attenuation techniques are used with the integrated PET/MR scanners. Specifically, tissue decomposition with T1-weighted Dixon sequences are used to back-calculate attenuation by measuring the relative amounts of water and fat content, though this method grossly underestimates attenuating effects, as signal void from bone cannot be differentiated from air. Possible solutions include atlas-based methods that retrospectively add missing tissue information and ultrashort echo time sequences that display tissues with very short T2* (eg, bone). Usually, Dixon methods are sufficient in clinical practice. Misregistration may be somewhat greater than with PET-CT for abdominopelvic organs, but the implication of this seems to be little in routine clinical practice.
The protocol at the Cleveland Clinic (S.N.S.), which uses a Siemens Biograph mMR, is as follows. Following the acquisition of scout images, coronal in-phase and opposed-phase T1-weighted Dixon images of the WB are obtained. These images are postprocessed to provide coronal water and fat images, which are then manipulated to provide a map of attenuation that is used for attenuation correction. Following this, simultaneously during the PET acquisition at each table position, axial half-Fourier acquisition single-shot turbo spin echo (HASTE) and coronal T2 turbo spin-echo (TSE) images are also obtained. Later, these may be viewed individually or overlain with the WB PET data to provide evaluation of fused images. When clinically indicated, following the WB PET/MR scan, a dedicated pelvic MR imaging examination is completed, which includes small FOV multiplanar T2 images of the pelvis, axial DWI, as well as T1 fat-saturated images of the pelvis without and dynamically after the administration of intravenous gadolinium. T2-weighted images help assess parametrial invasion; DWI correlates well with tumor volume for radiation planning, and precontrast and postcontrast images help assess local staging and surrounding structures. The WB PET/MR scan requires about 30 minutes, with each of the approximately 6 bed positions requiring a 4-minute scan time; the table times for the comprehensive examination of both the WB + dedicated MR imaging of the pelvis lasts approximately 1.0 to 1.5 hours. At interpretation, the parameters of the standardized uptake value (SUV), DWI, and tumor dimensions are measured for quantitative as well as qualitative data.
An example of another protocol used on a Siemens simultaneous machine is given by Grueneisen and colleagues, who in their published gynecology PET/MR work, use a fairly similar protocol consisting of Dixon sequences for attenuation correction, coronal 3-dimensional (3D) volume-interpolate breath-hold (VIBE), followed by an axial echo-planar DWI series, a coronal T2-weighted 2-dimensional (2D) turbo inversion recovery magnitude series, and a 2D fat-saturated HASTE series of the WB. PET images are acquired concurrently. This imaging is then followed by pelvis-only 3D VIBE axial, dynamic postcontrast 3D fat-saturated sagittal VIBE images of the pelvis at 20, 60, and 90 seconds, T2-weighted TSE sagittal images; and finally 3D postcontrast VIBE images of the WB. In the authors’ own experience, additional dedicated PET images of the pelvis can be obtained while MR imaging of the pelvis is being performed, which is sometimes useful as it allows for a second set of images, often at a higher count rate, and a second time point as well. The whole series takes about 41 to 51 minutes. This group has also published a faster protocol (appropriately labeled FAST), with a WB DWI echo planar imaging (EPI) sequence, HASTE sequence, and postcontrast VIBE sequence, which can be performed in 27 minutes, apparently with no loss of sensitivity.
An example of a protocol used on a sequential GE machine is given by Queiroz and colleagues, who (using a sequential GE Discovery machine) used a T2-weighted SSFSE of the upper abdomen, followed by T1-weighted liver acquisition with volume acceleration (LAVA) (a 3D gradient-echo sequence), DWI EPI, axial T2-weighted periodically rotated overlapping parallel lines with enhanced reconstruction (PROPELLER), sagittal T2-weighted PROPELLER, and contrast-enhanced T1-weighted LAVA FLEX images of the pelvis. PET imaging is acquired before or afterward. There is no WB image mentioned here.
Nonclinical issues include reimbursement and workflow. No category 3 billing codes currently exist for hybrid FDG PET/MR imaging, and so the WB PET/MR is usually billed as a PET-only examination. When a diagnostic MR of the abdomen or pelvis is obtained in the same session, an additional charge of diagnostic MR scan of the abdomen or pelvis may be applied. (Additional charges are applied similarly for other organs, such as the brain or extremities.) The clinical success of a modality is heavily predicated on ease of access and workflow, both for outpatients and inpatients. In the ideal scenario, a hybrid PET/MR imaging scanner would be located in close proximity to the nuclear medicine and MR imaging departments. Such a location would also have favorable implications in case of patient-related or equipment-related emergencies. If this is not possible, a system of robust communication between imagers and technologists is key for maximum imaging quality and efficiency. Although a discussion of the technical considerations related to image acquisition are beyond the scope of this article, image interpretation on existing vendor platforms remains a challenge because of the large data sets, the need to support multiple modalities, and technical knowledge on the physician side. Specifically, the authors’ institutions differ in this regard; one of us (S.N.S.) uses the vendor-provided software, whereas the other (J.O.) uses a third-party application specialized for image fusion and registration. Interpretation may be carried out separately by nuclear medicine and abdominal or women’s imaging physicians, particularly if separate reports for the PET and MR are necessary, or by the same person if he or she is dually trained and qualified. Many institutions generate 2 separate reports for billing purposes, whereas others generate an integrated structured disease-specific reports. If the examinations are being read separately, it is important that both of the interpreting physicians communicate with each other to avoid discrepancies that can confuse referring clinicians.
When Is PET/Magnetic Resonance Used? Current Guidelines and Applications
In general, there are 2 sets of imaging guidelines presently used, those by the National Comprehensive Cancer Network (NCCN) and those by the American College of Radiology (ACR). The NCCN’s guidelines go through every part of cancer treatment, from staging to recurrence, but often make little distinction among imaging modalities (many times suggesting CT, MR, or PET-CT), whereas the ACR’s guidelines are more detailed in regard to imaging modalities and technical scan details and less expansive in terms of clinical situations covered. No guideline currently covers PET-MR, given the novelty of the modality; but most guidelines do cover PET and MR separately. Any indication for which both PET and MR are suggested as possibly useful is a possible indication for PET-MR.
In general, MR is seen as more effective for local staging and PET-CT for nodal and distant metastases (particularly for endometrial and cervical cancers). In all cases, PET-CT is thought to be optimal to detect suspected recurrences, with MR imaging being exquisite in helping to localize them. MR also has the potential to avoid image artifacts from the bony pelvis. There is, thus, the opportunity for one-stop shopping and better anatomic localization with PET/MR.
As an emerging modality, uses for PET/MR are only now being discovered. Usually, we attempt to combine the local soft tissue contrast of MR with the nodal staging abilities of PET. Specifically, MR is better at assessing tumor size and local issues, such as stromal invasion and parametrial involvement, whereas PET is better at metastases to lymph nodes and bone. MR is also better at assessing liver metastases because of better soft tissue contrast and the moderate background physiologic liver uptake. Additional MR functional-imaging sequences, such as delayed contrast enhancement and DWI, may help in detecting lesions or assessing their importance.
Cervical (and vulvar) cancer
For cervical cancers, the NCCN favors PET for initial staging of tumors greater than stage IB2 to exclude extrapelvic disease; MR is added as indicated. PET and MR are also used to look for distant metastases if patients have para-aortic nodes on initial surgery. MR is the modality of choice to exclude disease high in the endocervix, to determine soft tissue and parametrial involvement, and to rule out gross residual disease after hysterectomy. The ACR also discusses pretreatment staging; and MR imaging is seen as having the best visualization of the primary tumor and extent of soft tissue disease in the central pelvis, with FDG-PET being better for nodal, extrapelvic, and bone metastasis. Thus, in evaluating gynecologic cancers at baseline, FDG PET/MR represents an optimal modality as it combines both these modalities in one examination. The NCCN states that both PET and MR can be used to plan radiation therapy for cervical cancer, with MR used for delineating residual tumor geometry. Both PET and MR can also be used for surveillance after chemoradiotherapy. Finally, as with other cancers, PET and MR are indicated for detecting recurrence if clinically indicated.
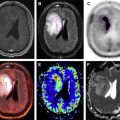
In initial staging of cervical cancer, PET can be used in correlation with pelvic MR imaging to stage the initial extent of disease. For example, the high resolution of small-FOV MR can be used to assess parametrial invasion (stage IB vs IIB), whereas WB PET staging can be used to find distant metastatic disease (stage IVB) ( Fig. 1 ). The small FOV is also adaptable to the clinical question. PET may detect nodes not sufficiently enlarged to be detectable by MR or CT, as shown in a recent meta-analysis of 41 studies, and is particularly useful in the para-aortic region ( Fig. 2 ), as described in another meta-analysis of 22 studies; as discussed, adding a fused MR to a PET/CT increases sensitivity. Using MR rather than CT can resolve the location of PET-detected lesions not easily seen on CT or make subtle lesions more obvious, and MR itself may help assess local invasion ( Fig. 3 ). It may also avoid confusing physiologic accumulations of tracer, such as urine, in a ureter or a functional ovary with disease.