Chapter Outline
Division of the Intraembryonic Celom and Formation of the Diaphragm
Anomalies of Diaphragmatic Development
Normal Gallbladder and Bile Duct Development
Abnormal Gallbladder and Bile Duct Development
Normal Development of the Esophagus
Abnormal Development of the Esophagus
Normal Development of the Stomach, Duodenum, and Lesser Omentum
Abnormal Development of the Stomach and Duodenum
Normal Development of the Pancreas
Abnormal Development of the Pancreas
Normal Development of the Lesser Sac and Greater Omentum
Abnormal Development of the Lesser Sac and Greater Omentum
Normal Development of the Spleen
Abnormal Development of the Spleen
Normal Midgut Rotation and Fixation
Abnormal Midgut Rotation and Fixation
The complex anatomy of the gastrointestinal tract and the peritoneal cavity arises from much simpler origins. The transition from the primitive straight tubular alimentary canal to the elongated and tortuous gut (and its accessory organs) suspended by mesenteries and encased by peritoneal reflections can readily be explained by a well-defined series of events. Although these processes are summarized individually, these multiple events often occur simultaneously with complex interactions.
Early Development
After fertilization, the zygote rapidly undergoes repeated mitotic divisions that result in an increased number of cells. This occurs without a corresponding increase in the cell mass.
Approximately 3 days after fertilization, a solid ball of cells (i.e., morula) develops. The next day, central cavities appear, which separate the cells into the trophoblast (from which part of the placenta develops) and the embryoblast. Two days later, endometrial implantation begins.
During the second week of development, the spherical mass of cells flattens into a bilaminar disk. A primitive yolk sac also develops. During the third week, the embryonic disk rapidly develops into the embryo (i.e., gastrulation). The cells differentiate into the three classic germ cell layers: endoderm, mesoderm, and ectoderm.
The endoderm gives rise to the lining epithelium of the respiratory and gastrointestinal tracts as well as the glandular elements of the liver and pancreas. From the mesoderm arise the smooth muscle of the gastrointestinal tract, the connective tissues, and their associated blood vessels. Blood cells and their progenitors, striated muscle, bone, cartilage, and the reproductive and genitourinary tract are also mesodermal in origin. The ectoderm is the source of the epidermis and the nervous system.
Clefts appear within the lateral aspects of the developing intermediate mesodermal layer, forming the intraembryonic celom. These open laterally into the yolk sac. They lie between and separate the more dorsal somatic mesoderm from the ventral splanchnic mesoderm. The somatic mesoderm, in association with the ectodermal layer, forms the embryonic body wall (i.e., somatopleure); the splanchnic mesoderm, along with the endoderm, forms the embryonic gut (i.e., splanchnopleure) ( Fig. 113-1 ).
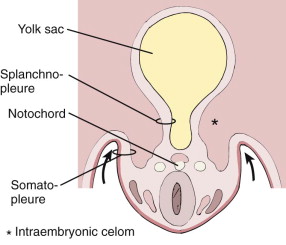
As the lateral margins of the embryonic disk move ventrally and medially, they begin to pinch off the yolk sac and the more laterally placed intraembryonic celom. Continued growth by the somatopleure and its eventual midline fusion complete the encompassment of the intraembryonic celom with formation of a cylindrical body cavity. The more centrally placed splanchnopleure also starts to close ventrally, partially separating the primary yolk sac into the gut and the secondary yolk sac, which is separated by the yolk stalk ( Fig. 113-2 ).
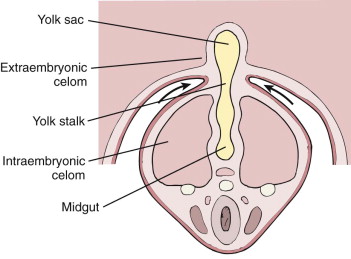
A primitive alimentary tube is formed within the larger surrounding body cavity. The dorsal mesentery and its visceral peritoneum are derived from the splanchnopleure. Most of the ventral mesentery, also derived from the splanchnopleure, degenerates with time, leaving a large embryonic body cavity (i.e., celom).
Division of the Intraembryonic Celom and Formation of the Diaphragm
During the fourth to sixth weeks of development, the large common intraembryonic celomic cavity is partitioned into pleural, pericardial, and peritoneal spaces. By the fourth week, a large pericardial cavity is connected to the peritoneal cavity by two smaller pericardioperitoneal canals. The pressure of the developing head causes the heart and pericardial cavity to be displaced caudally and ventrally. The pericardioperitoneal canals exit from the pericardium along its dorsal aspect to enter the peritoneum.
As the lung buds develop, they grow into the paired pericardioperitoneal canals. This produces two pairs of ridges. The cranial pair gives rise to the pleuropericardial membrane that eventually separates the primitive pericardial cavity into definitive pericardial and pleural spaces. The more caudal pair gives rise to the pleuroperitoneal membrane. This plays an important role in the development of the diaphragm. As the lung buds grow superiorly and the liver and peritoneal space expand inferiorly, these membranes become more prominent. They attach themselves to the abdominal wall along their dorsal and lateral margins. Their free edges project into the pericardioperitoneal canals.
During the sixth week, the free edges of the dorsolateral pleuroperitoneal membrane fuse with the midline dorsal mesentery of the esophagus, forming part of the primitive mediastinum at this point. The anterior half of the primitive diaphragm is formed by the septum transversum, which arises in the third week as a condensation of mesoderm. By the fourth week, it has thickened to form an incomplete division anteriorly between the pericardium and the peritoneal cavity ( Fig. 113-3 ).
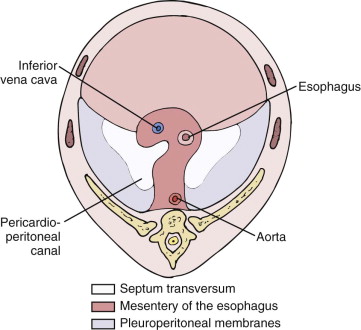
Between weeks 6 and 12, many changes take place in the relative contributions of these structures to the diaphragm that exists at birth. The large dorsolateral component formed by the pleuroperitoneal membranes decreases in size. Myoblasts from the abdominal wall migrate into the peripheral aspects of the membranes. These contribute to the growth of the diaphragm and eventually give rise to the costophrenic sulci. Myoblasts also grow into the primitive dorsal esophageal mesentery, forming the diaphragmatic crura.
In addition to its complex formation, the diaphragm markedly shifts in position. In the fourth week, the septum transversum is at the level of the third to fifth cervical somites. The myoblasts and associated nerve innervation also arise from these levels. From the fourth to the sixth weeks, the dorsal part of the embryo grows rapidly, causing an apparent descent of the diaphragm. The mesenchyme of the septum transversum contributes its own myoblasts to the diaphragm, maintaining its original C3-C5 innervation. In its definitive state, the diaphragm lies at the level of the thoracolumbar junction while maintaining its phrenic nerve, midcervical innervation.
Anomalies of Diaphragmatic Development
Diaphragmatic Hernia
If the pleuroperitoneal membrane fails to close entirely, a patent canal may persist between the pleural and the peritoneal cavities. If this dorsally and laterally placed canal is still present at the reduction of the physiologic herniation of the midgut at week 10 (discussed later), the returning bowel may herniate through this patent foramen of Bochdalek into the chest. This occurs most frequently on the left side.
Foramen of Morgagni Hernia
A natural weakness in the anteromedial portion of the diaphragm (i.e., retrosternal) is caused by the passage of the superior epigastric vessels. Herniation of omentum or intestines may occur at this site, and it occurs most frequently on the right side.
Eventration
Defective muscle development of the dome of the diaphragm may lead to structural weakness and subsequent ballooning. Only a thin, aponeurotic sheet of tissue is then present. Abdominal contents may bulge into the thoracic cavity, simulating a difficult to differentiate eventration from a true herniation.
Normal Liver Development
During the fourth week of development, the caudal portion of the foregut develops a ventral bud called the primordial hepatic diverticulum. This endodermal liver bud enlarges and grows into and between the two layers of the ventral mesentery. More superiorly, it grows in contact with and into the mass of mesenchyme, the septum transversum. The ventral liver bud divides into cranial and caudal portions within the mesentery. The cranial portion gives rise to the liver and intrahepatic bile ducts; the caudal portion forms the gallbladder and cystic duct.
The cranial liver bud further divides into right and left lobes, which are initially of equal size. The right lobe eventually becomes much larger than the left. The distal branches of the right and left lobe cords undergo canalization to become the definitive main right and left hepatic ducts. The intrahepatic biliary tree is thought to arise from the hepatic parenchyma. These cords of tissue extend along the randomly created pattern of portal vein tributaries (discussed later). Consequently, the pattern of intrahepatic bile ducts is variable.
The mature liver has two ligaments that represent remnants of embryologic vascular channels. These are the ligamentum teres (i.e., round ligament) within the falciform ligament and the ligamentum venosum. These develop from the vitelline and umbilical veins, respectively. The paired vitelline veins drain the yolk sac, pass through the developing liver and septum transversum, and empty into the right side of the primitive heart (i.e., sinus venosus). The vitelline veins within the liver create a meshlike network of vascular channels that become the hepatic sinusoids. Some of the lining cells of the sinusoids later differentiate into macrophages (i.e., Kupffer cells), the reticuloendothelial component of the liver. The segments of the vitelline veins proximal and distal to the sinusoids become, respectively, the hepatic veins and the hepatic portion of the portal vein.
The paired umbilical veins drain the placenta and chorion. They pass through the septum transversum, contributing minimally to hepatic sinusoidal development, and then empty into the sinus venosus. The entire right umbilical vein and a segment of the left proximal (cephalad) to the liver atrophy. A large venous channel, the ductus venosus, arises from the hepatic sinusoidal network and carries blood from the distal (caudal) left umbilical vein into the sinus venosus, bypassing the liver. Eventually, the lumen of the ductus venosus is obliterated, and the structure becomes the ligamentum venosum. The distal portion of the left umbilical vein migrates medially to the liver edge, and its lumen closes. It then becomes the round ligament (i.e., ligamentum teres). This ligament, which connects the ligamentum venosum to the umbilicus, is encased within the most anterior portion of the ventral mesentery, the falciform ligament ( Fig. 113-4 ).
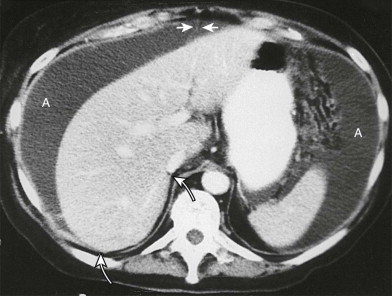
The mesenchymal septum transversum contributes to the ventral mesentery that surrounds the liver and gallbladder. The fibrous tissues of the liver, including Glisson’s capsule, embryonal hepatic hematopoietic tissues, and the Kupffer cells lining the sinusoids, are also derived from the septum transversum.
The membranous ventral mesentery is a double-layered structure that encloses the liver and gallbladder to become their visceral peritoneum. The cephalic portion of the developing liver directly contacts the septum and therefore is not enveloped by the ventral mesentery. This region, devoid of peritoneum, is known as the bare area (see Fig. 113-4 ). The visceral peritoneum reflects off the liver onto the undersurface of the diaphragm as the coronary ligaments outlining this region.
Abnormal Liver Development
Atypical Segmentation
Variations of lobulation sometimes occur. A bipartite liver results from an exaggerated separation of the lobes, which may represent congenital absence of the medial segment of the left lobe.
Absence of the right lobe may result from maldevelopment of the portal vein or the primordial hepatic diverticulum proper. A portal venous abnormality may account for an absent left lobe. Multilobar livers—as many as 16—may be seen. Hepatic anomalies are discussed further in Chapter 85 .
Atypical Location
Hepatic lobes can develop in the thorax. They may undergo torsion when they have a separate mesentery. Heterotopic liver tissue may also be found within structures that share a common embryologic development: gallbladder, pancreas, umbilical cord, and gastrohepatic ligament.
Normal Gallbladder and Bile Duct Development
The fourth week of embryogenesis is marked by the appearance of the hepatic diverticulum. It then divides into cranial and caudal buds. The caudal bud forms the gallbladder, cystic duct, and extrahepatic bile ducts. Their development precedes that of the intrahepatic ducts by several weeks.
The common bile duct develops as a cord connecting the cystic and main hepatic ducts to the descending duodenum. The hollow gallbladder and common bile duct primordia become occluded with proliferating endoderm during the fifth week. They recanalize by vacuolization and cell degeneration by the end of that week. The recanalization of the common bile duct precedes that of the gallbladder and duodenum. As the duodenum rotates 90 degrees to the right, the common bile duct rotates an additional 180 degrees along with the ventral pancreatic anlage. The common duct is carried from its original ventral position to the right, then posteriorly, and finally to the medial aspect of the duodenal sweep.
The gallbladder and intrahepatic ducts also communicate through the cystohepatic ducts of Luschka during fetal life. These ducts usually atrophy in the adult but may remain patent in some patients. When they persist and are not recognized during cholecystectomy, a significant bile leak may result.
Vacuoles in the wall of the duodenum coalesce to form two separate channels and then one single lumen for the adjacent common bile and main pancreatic ducts. Within the wall of the duodenum are primitive ampullary tissues that enlarge and displace the junction of the two ducts away from the duodenal lumen. This displacement is reversed by the growth of the duodenal wall smooth muscle. This accounts for the considerable variability seen in the junction of these two structures.
Abnormal Gallbladder and Bile Duct Development
Phrygian Cap
The phrygian cap is an abnormal shape of the gallbladder. It represents a folding of the fundus on itself and is not truly pathologic.
Gallbladder Diverticulum
The gallbladder diverticulum is the true diverticulum (containing all normal wall elements). It probably is the remnant of the cystohepatic duct. It can be a site of bile stasis and stone formation (see Chapter 76 ).
Positional Anomalies
The most common anomaly of gallbladder position is that of a “wandering” or “floating” gallbladder. Elongation of the gallbladder mesenteric attachment to the undersurface of the liver results in excessive mobility. The gallbladder may herniate into the lesser sac, undergo torsion, or be located in other intra-abdominal locations (see Chapter 76 ).
Bifid Gallbladder or Duplication
A bifid gallbladder has two cavities but only one cystic duct. Each of the duplicate or triplicate gallbladders has its own cystic duct. These entities may result from a persistent outpouching of the extrahepatic duct or incomplete recanalization of the gallbladder. The latter may also give rise to a septate gallbladder (see Chapter 76 ).
Agenesis
Agenesis, a rare entity, results from a lack of development of the caudal portion of the liver bud or improper recanalization of the gallbladder. It may be associated with a host of other anomalies affecting many other organ systems (see Chapter 76 ).
Tracheobiliary Fistula
Tracheobiliary fistula is characterized by the combination of bile-stained sputum and pneumobilia. It results when the biliary tree is connected to the carinal region.
Biliary Atresia
In biliary atresia, the number of intrahepatic bile ducts is decreased. This may develop primarily or result from α 1 -antitrypsin deficiency, cystic fibrosis, or viral hepatitis (see Chapter 120 ).
Alagille’s Syndrome
Alagille’s syndrome is an autosomal dominant syndrome of arteriohepatic dysplasia. It is characterized by a paucity of bile ducts, peripheral pulmonic stenosis, vertebral anomalies, and mental and physical retardation (see Chapter 119 ).
Cystic Disease
The spectrum of cystic diseases ranges from intrahepatic cysts or fibrosis to renal disease. Various patterns of inheritance also occur. Cysts may be attributable to defective development of the intrahepatic ducts. In other forms, bile duct hyperplasia and portal fibrosis may predominate (see Chapters 76 and 119 ).
Caroli’s Disease
Caroli’s disease is a nonfamilial entity characterized by segmental cystic dilation of the intrahepatic bile ducts. It may represent an intermediate form of disease between congenital hepatic fibrosis and choledochal cysts. A possible cause is perinatal hepatic artery occlusion. Multiple episodes of cholangitis may result from bile stasis (see Chapter 76 ).
Choledochal Cysts
Multiple theories have been proposed to explain choledochal cysts. These concepts include distal biliary ductal obstruction with subsequent weakening and ballooning of the wall, anomalous course of the bile duct through the duodenal wall, and deficient development of the bile duct wall. Additional theories include a high junction of the bile and pancreatic ducts proximal to the sphincter of Oddi, which allows reflux of pancreatic enzymes up the common bile duct when the sphincter is contracted, resulting in cholangitis and dilation. Another theory suggests an excess quantity of epithelial cells in the primitive choledochus, followed by recanalization leading to cyst formation. Viral infection leading to infantile obstructive cholangiopathy is another widely favored theory. During the past decade, technologic advances in imaging have greatly enhanced the noninvasive evaluation of the biliary tract. For example, multidetector computed tomography (CT) and magnetic resonance cholangiopancreatography (MRCP) can accurately depict preoperative anatomy and enable delineation of an anomalous pancreaticobiliary junction.
Choledochal cysts are found more often in Asians and in females. Five classic radiographic types have been identified. The most common type is aneurysmal dilation of the common duct, which often extends into the cystic duct and the main hepatic ducts. The rare second form is a diverticulum that projects off the distal common duct. The third form is that of a choledochocele, which represents a dilation of the distal common bile duct that protrudes into the duodenum. This third form may also be a congenital duplication cyst of the duodenum through which the common duct courses (see Chapter 76 ).
Two other types are included in the classification of choledochal cysts. One is multifocal segmental intrahepatic and extrahepatic ductal dilation. The other is Caroli’s disease (discussed earlier).
Normal Development of the Esophagus
During the latter half of the fourth week, the respiratory system develops as a ventral bud of the foregut. The laryngotracheal groove forms at the caudal end of the primitive pharynx and becomes the laryngotracheal diverticulum. Longitudinal (tracheoesophageal) folds eventually separate the ventral respiratory apparatus from the dorsal esophagus.
Initially, the esophagus is relatively short. Growth of the heart and lungs contributes to the elongation of the esophagus. Its epithelium and epithelial glands proliferate and obliterate the hollow lumen. This cellular plug is resorbed by the eighth week, the end of the fetal period.
Abnormal Development of the Esophagus
Tracheoesophageal Fistula
Partial fusion of the tracheoesophageal folds leads to incomplete separation of the respiratory and gastrointestinal tracts (i.e., tracheoesophageal fistula). This is usually accompanied by some narrowing of the associated lumens (i.e., esophageal atresia). This common anomaly is discussed further in Chapter 114 .
Duplication
Incomplete resorption of the endothelial plug may result in a duplication of the esophagus (see Chapter 114 ). Duplication cysts are manifested as submucosal masses and do not usually communicate with the esophageal lumen. CT or endoscopic sonography can document its cystic nature.
Stenosis
Weblike narrowing or long segmental strictures may occur in the distal esophagus. Stenosis results from incomplete recanalization (see Chapter 114 ).
Esophageal Bronchus
An esophageal bronchus is evidence of the common origin of the respiratory and gastrointestinal tracts. In this entity, a branching bronchus arises from the esophagus, usually in association with a pulmonary sequestration.
Congenital Short Esophagus
If the esophagus does not proportionally elongate as the body grows, a congenital short esophagus occurs. Seen at birth in association with a hiatal hernia, it is distinct from the acquired form found in adults.
Normal Development of the Stomach, Duodenum, and Lesser Omentum
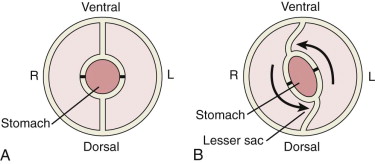
At the end of the fourth week, which is the midpoint of the fetal period, the primitive stomach is still a straight, hollow tube in the midsagittal plane. During the next 2 weeks, the future stomach dilates, first in a fusiform manner and then with preferential growth of its dorsal wall. This causes a dorsal bulge, which is the origin of the greater curvature.
During the sixth to eighth weeks of development, the stomach rotates along two different axes simultaneously. The first and major rotation about the longitudinal axis causes the dorsal bulge to form the lateral border of the stomach. The former ventral aspect forms the medial wall (i.e., lesser curvature). This accounts for the left vagus nerve innervating the anterior wall of the stomach and the right vagus nerve innervating the posterior wall in the adult ( Fig. 113-5 ).

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








