Arthritides
Arthritis is the most common cause of disability in America. Close to 50 million adults (approximately 22%) in the United States report having physician-diagnosed arthritis. It has been estimated that this number will rise to 67 million by the year 2030, and more than one-third will have limited or restricted activity as a consequence. Arthritis affects all age groups, races, and ethnicities. Overall, it is more common in women than men in every age group. Arthritis is a more prevalent chronic condition than heart disease, hearing impairments, chronic bronchitis, asthma, or diabetes.113 Not only do arthritis and its related conditions have a significant impact on activities of daily living, such as housekeeping, sleeping, driving, and working, they have a substantial economic impact as a result of lost wages and direct health care costs. As more and more research is being devoted to the cause and underlying predispositions to arthritis, genetic and familial factors seem to be increasingly prominent. Early recognition and appropriate management of arthritis are important to limit its progression and related disabilities.
Imaging
Plain film radiography has traditionally been the most widely used and useful imaging modality for diagnosing, differentiating, monitoring the progression, and evaluating the treatment of the various arthropathies.576 Although magnetic resonance imaging (MRI), ultrasonography and computerized tomography are attractive because of their multiplanar ability, conventional radiography remains among the least expensive and most easily accessed imaging modalities. Therefore, it is still difficult to imagine proceeding to more advanced imaging techniques without first evaluating the patient with plain film. Despite some limitations, such as insensitivity to very early joint changes, it adequately demonstrates bone erosions, osteophytes, alterations of joint space, and misalignment.
Advanced imaging modalities are being used alone, and in conjunction with plain film radiography as they provide detection of more subtle findings such as synovitis, periarticular osteoporosis, and early erosions, when plain films are negative or inconclusive. Advanced imaging is useful in evaluating joints with complex anatomy such as the sacroiliac (SI) articulations, which can be difficult to assess by the use of plain film. Computed tomography (CT) provides excellent anatomic detail and images in an axial plane. MRI is used to evaluate the cartilaginous and ligamentous joint structures and marrow of the adjacent bone. Scintigraphy provides a useful method of gathering functional information to aid in morphologic assessments provided by other imaging modalities. Ultrasonography has the capability to define joint effusions and identify edematous connective tissues. No single imaging modality meets every need for every condition. Each can play a vital role in the evaluation and management of a patient with a suspected or known arthropathy.
Classification
In general, arthritis can be grouped into three major categories: inflammatory (rheumatoid), crystal-induced (gout), and degenerative (osteoarthritis). Inflammatory arthritides are subdivided into rheumatoid arthritis (and related diseases) and connective tissue disorders. They also can be subdivided into rheumatoid types (seropositive) and rheumatoid variants (seronegative) based on the likelihood of the serologic presence or absence of the rheumatoid factor, as established by the latex fixation test.
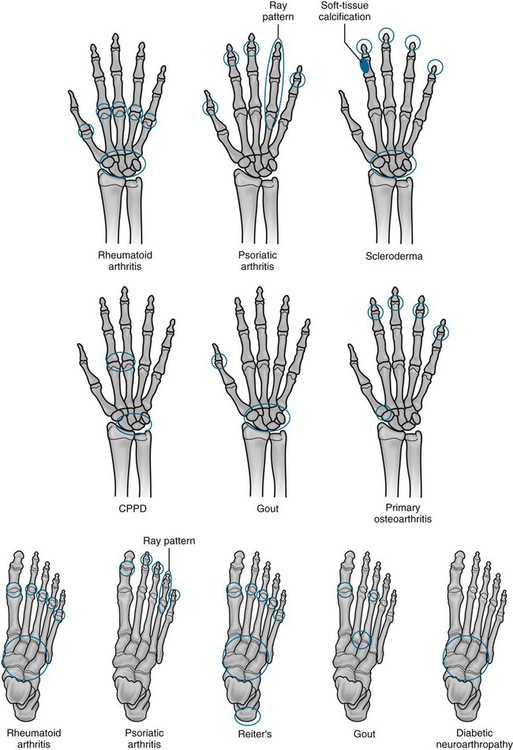
Arthritis may be monoarticular or polyarticular. All types of arthritis may be monoarticular early in their pathologic course. Joint abnormalities that are caused by trauma or infections usually remain monoarticular. Arthritides are differentiated on the basis of clinical data and imaging studies (Table 9-1 and Fig. 9-1). The most useful information about them comes from plain film radiographs or advanced imaging coupled with clinical data that relate to skeletal distribution, patient age, patient gender, joint swelling, joint stiffness, joint range of motion, symptom response to physical activity, and laboratory tests (e.g., erythrocyte sedimentation rate [ESR] or C-reactive protein [CRP], rheumatoid factor [RF], antinuclear antibodies [ANAs], human leukocyte antigen [HLA] typing).
TABLE 9-1
SUMMARY OF SELECTED ARTHRITIDES
| Arthritis/Mode | Female-to-Male Ratio | Age of Onset | Target Joints | Distribution | Radiographic Features | Clinical Features | Lab | Extraarticular Manifestations |
| Rheumatoid/inflammatory | 3 : 1 | 40–70 | MTP, MCP, PIP, knees, hips, cervical spine | Bilateral, symmetric | Capsular swelling, symmetric joint space narrowing, juxtaarticular osteoporosis, marginal erosions; joint deformity | Morning stiffness, joint swelling | +RF (IgM), ↑ESR, 10%–50% ANA, ↑C-reactive protein (CRP) | Rheumatoid subcutaneous nodules, pulmonary, cardiovascular |
| JIA/inflammatory | 3.5 : 1 | <16 | Knees; ankles; elbows; wrists; cervical spine | Bilateral, symmetric if polyarticular | Capsular swelling, symmetric joint space narrowing, juxtaarticular osteoporosis, marginal erosions, ankylosis, growth abnormalities, periostitis | Varies with subtype: systemic fever, adenopathy; arthralgia; rash; limp | ±RF, HLA-B27 and ANA depending on subtype; ↑ESR and CR protein; systemic = leukocytosis | Varies with subtype; hepatosplenomegaly subcutaneous nodules; iridocyclitis |
| SLE/inflammatory | 9 : 1 | 30–50 | MCP; PIP of the hands primarily | Bilateral, symmetric | Nonerosive, reversible, joint deformities; osteonecrosis as possible complication | Arthralgia; butterfly rash; constitutional signs and symptoms | ANA (98%); anti–DNA antibodies; LE cells (70%–85%); RF (20%–35%); ↑ESR and CR protein | Rash; renal disease; interstitial lung disease |
| Scleroderma/inflammatory | 8 : 1 | 35–65 | Distal tufts; DIP; PIP of the hands primarily | Bilateral, symmetric or asymmetric | Acroosteolysis, erosions, subcutaneous calcifications | Raynaud phenomenon; skin fibrosis; arthralgia of hands; telangiectasia | ANA in 60%; +RF (30%); ↑ESR | Skin fibrosis; renal fibrosis; GI fibrosis; interstitial lung disease; pericarditis |
| Polymyositis/dermatomyositis/inflammatory | 2 : 1 | 40–60 | Large muscles of the proximal appendicular skeleton; DIP, PIP, and MCP joints | Bilateral, symmetric muscle involvement | Muscle edema; subcutaneous, intermuscular, and periarticular calcifications; transient osteopenia, soft-tissue swelling | Muscle weakness and atrophy; rash; arthralgia of hands, wrists, and knees | ↑CPK; muscle biopsy = inflammation and degeneration | Interstitial lung disease; pericarditis |
| AS/inflammatory | 1 : 10 | 15–35 | SI; spine: vertebral bodies and apophyseal articulations; hip; shoulder | Bilateral, symmetric | Erosions; periostitis; ankylosis; thin, marginal syndesmophytes | LBP and stiffness; limited chest expansion | +HLA-B27 (90%); −RF, ↑ESR, and CR protein | Interstitial fibrosis; aortic insufficiency; iritis |
| Enteropathic/inflammatory | Varies with underlying bowel disorder | Varies | SI; spine; knee | Symmetric in axial skeleton; asymmetric in extremities | Mimics AS in the axial skeleton; nonerosive in the extremities | LBP and stiffness; IBD | +HLA-B27; −RF; ↑ESR | Underlying bowel disorder |
| Psoriatic/inflammatory | 1 : 1 | 30–50 | Predilection for upper extremity; DIP and PIP; SI; spine | Bilateral, symmetric, or asymmetric in SI joints; asymmetric in extremities | Sausage digit; marginal or central erosions with periostitis; early joint space widening with eventual narrowing; bulky, nonmarginal syndesmophytes; SI erosions and ankylosis | Psoriasis; nail changes (thickening, pitting, discoloration); LBP | +HLA-B27; −RF; ↑ESR | Psoriatic skin and nail changes |
| Reiter/inflammatory | 1 : 5 | 15–35 | Predilection for lower extremity; MTP; calcaneus; SI; spine | Asymmetric in foot; bilateral, symmetric, or asymmetric in SI joints | Mimics psoriatic in the spine and extremities; calcaneal enthesopathy | LBP; heel pain, urethritis; conjunctivitis | Leukocytosis; ↑ESR; HLA-B27 (75%); −RF | Pulmonary fibrosis; urethritis; conjunctivitis |
| Gout/crystal deposition | 1 : 20 | 40–50 | MTP of first digit; other MTP joints, DIP, midfoot, ankle, DIP joints of hand | Asymmetric; often monoarticular | Soft-tissue nodules (tophi) with calcification; paraarticular erosions; overhanging edge; preserved joint space; lack of osteopenia | Red, hot, swollen joint; usually monoarticular | Hyperuricemia; sodium urate crystals in synovial fluid | Tophus in bursa or helix of the ear |
| CPPD/crystal deposition | Varies with site of involvement | Varies; increases with age | Knee; wrist; hip; MCP | Often bilateral, asymmetric | Chondrocalcinosis; DJD-like changes; subchondral cysts | Varies from asymptomatic to acute gout-like pain | Calcium pyrophosphate crystals in synovial fluid | None |
| HADD/crystal deposition | 1 : 1 | 40–70 | Shoulder | Asymmetric; usually monoarticular | Cloud-like periarticular calcification | Varies from asymptomatic to acute joint pain | Hydroxyapatite crystals by electron microscopy | None |
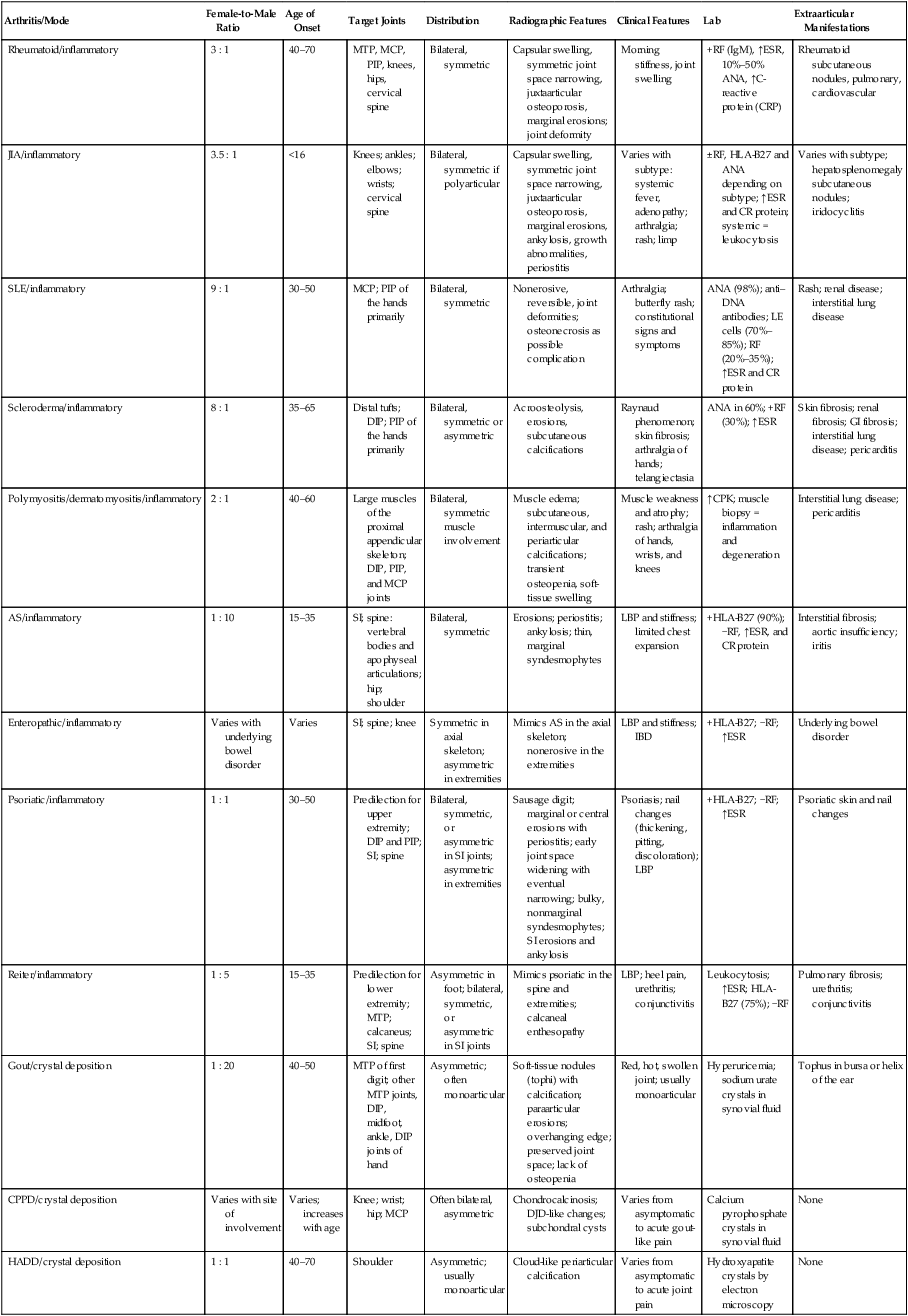
ANA, Antinuclear antibodies; AS, ankylosing spondylitis; CPK, creatine phosphokinase; CPPD, calcium pyrophosphate dihydrate; DIP, distal interphalangeal joint; DJD, degenerative joint disease; ESR, erythrocyte sedimentation rate; GI, gastrointestinal; HADD, hydroxyapatite deposition disease; HLA, human leukocyte antigen; IBD, inflammatory bowel disease; Ig, immunoglobulin; JIA, juvenile idiopathic arthritis; LBP, low back pain; LE, lupus erythematosus; MCP, metacarpophalangeal; MTP, metatarsophalangeal; PIP, proximal interphalangeal; RF, rheumatoid factor; SI, sacroiliac; SLE, systemic lupus erythematosus.
Inflammatory Arthritides
Rheumatoid Arthritis
Adult Rheumatoid Arthritis
Background.
Rheumatoid arthritis (RA) is an autoimmune disorder, and the most common chronic inflammatory arthritide, believed to affect approximately 0.5% to 1.0% of the general population.633 In the United States, prevalence rates estimate that 1.5 million U.S. adults have RA.265,642,645 Incidence rises with age, and peaks among people ages 65 to 74 years. Recent statistics show an increase in incidence rates among women in the United States, and a small decrease in incidence among men.475 From 1995 to 2007, rates increased by 2.5% each year among women, while there was a 0.5% decrease among men. Women are affected more often than men, and although some studies have shown a ratio as high as 7 : 1, it is generally accepted that women are affected two to three times more frequently.63,150,434,645 If the disease onset occurs at a more advanced age (>60 years), this ratio approaches 1 : 1.513,693
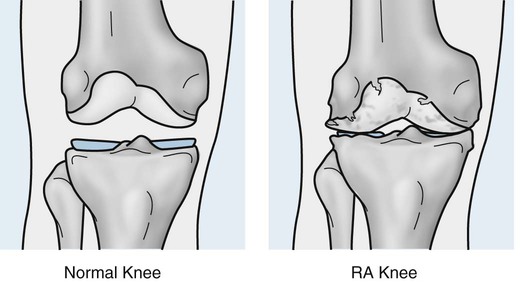
RA primarily affects the synovial lined joints and is frequently associated with a variety of extraarticular manifestations. RA is characterized by an inflammatory, hyperplastic synovitis (pannus), resulting in cartilage and bone destruction, consequent loss of function, and, at times, joint deformity. Although it is known that genetic and immunologic factors play a role in its pathogenesis, the underlying etiology remains uncertain,339,563 and there is no cure. Proposed etiologic theories revolve around a combination of genetic susceptibility (HLA-DRB1 and HLA-DR4), hormonal changes or imbalances (e.g., pregnancy, hormone replacement therapy, oral contraceptives212 and environmental or biologic triggers or risk factors (e.g., smoking, obesity).28,49,364,714 The T and B cells are involved in initiating and possibly propagating the chronic inflammatory disease process. Competing theories hold that the T cell initiates the disease process, but the chronic inflammation is self-perpetuated via macrophages and fibroblasts independent of the T cell.
Joint involvement typically is bilateral and symmetric, involving the peripheral and axial skeleton, with the small joints of the hands and feet, the wrists, knees, elbows, hips, and shoulders particularly affected.92 Changes in SI articulations are relatively infrequent. The natural history of RA varies. It may be monocyclic with the patient having one episode lasting 2 to 5 years without later reoccurrence; polycyclic, in which the levels of disease activity vacillates over the course of the condition; or progressive, where the condition is unremitting, and continues to worsen in severity.
Imaging Findings.
Plain film radiography, predominantly digitized images today, is an inexpensive tool and may be readily used by practitioners on a frequent basis in clinical practice to support clinical and laboratory findings consistent with a diagnosis of RA and aid in eliminating possible differential diagnoses. Although the radiographic changes admittedly fall behind the disease process, plain film does have the advantage of being readily reproducible and permitting easy serial comparison to follow the progression of RA and the effectiveness of its treatment.601,625 Ultrasonography and MRI are superior in detecting synovitis, and many of the initial subtle joint changes are visible much earlier with MRI500; however, studies have not yet shown MRI and/or sonography to sufficiently distinguish between early rheumatoid and nonrheumatoid arthritis.68 In addition, MRI is not yet used routinely because of its high cost, and depending upon the area to be examined, the small size of the joints involved and the inability to image several joints simultaneously (i.e., hand) may limit widespread use.142,188,538 At this time, the major role for MRI in patients with RA is in the preoperative assessment for surgical intervention where patients are experiencing neurologic deficits and/or severe pain.103,341 Consequently, this section primarily focuses on plain film findings.
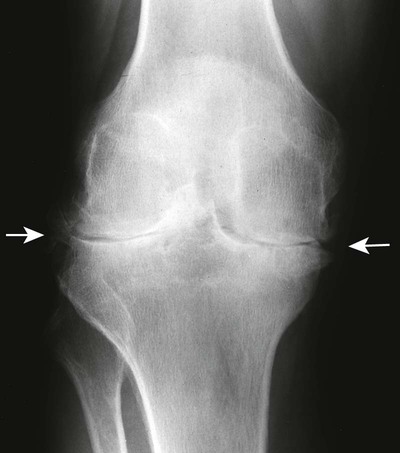
RA is classically symmetric and may involve any synovial joint. The general radiographic findings reflect the underlying pathologic change of chronic synovial joint inflammation with associated hyperemia, edema, and pannus formation. The first feature, which is usually the sole finding for the first few months, is fusiform periarticular soft-tissue swelling arising from capsular distension caused by excessive fluid accumulation seen clinically. Normal fat planes may be displaced or obliterated as a consequence of joint effusion, edema, and/or tenosynovitis. Increased blood flow to the synovium leads to a second early radiographic finding of juxtaarticular osteoporosis, which later becomes more generalized because of patient inactivity.324,405
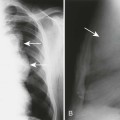
Joint spaces eventually narrow uniformly as the cartilage is destroyed by the enzymatic nature of pannus. Osseous erosions usually become apparent within the first 2 years of the disease,87,237,537 first occurring at the unprotected bone margins or “bare areas” (Fig. 9-2) in which the pannus has direct osseous contact, and later involving the subchondral bone. Radiographic erosions appear to occur most quickly in the first year of the disease.252 The appearance of an alternating pattern of erosions and normal cortical bone has been termed a “dot dash” appearance. Multiple, nonmarginated subchondral cysts or geodes typically develop and may communicate with the synovium.426,686
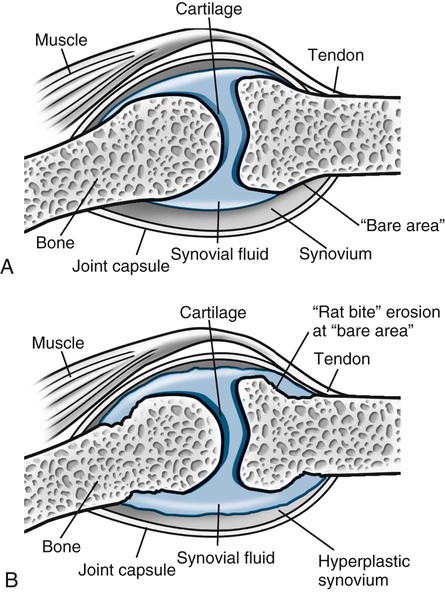
Later stages of the disease give rise to joint deformities resulting from tendon and ligament laxity, ruptures, and contractures. During periods of prolonged remission, radiographic signs of secondary osteoarthritis (OA) may develop, fibrous ankylosis may occur, and bony ankylosis may develop on rare occasions.
Hands and Feet.
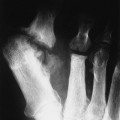
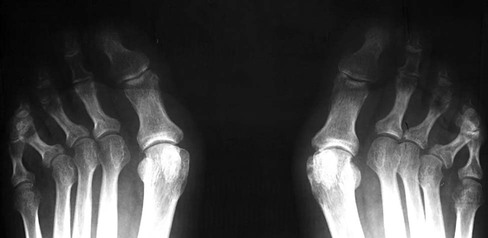
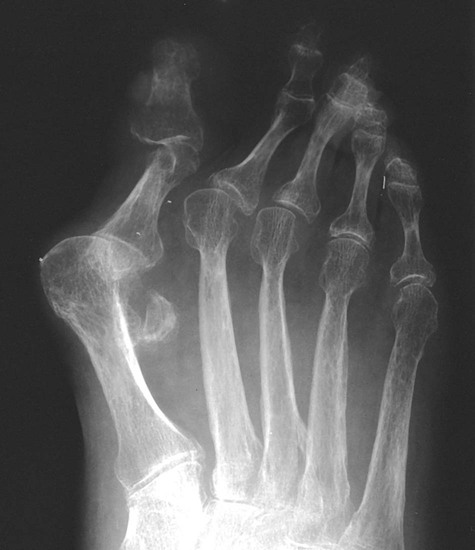
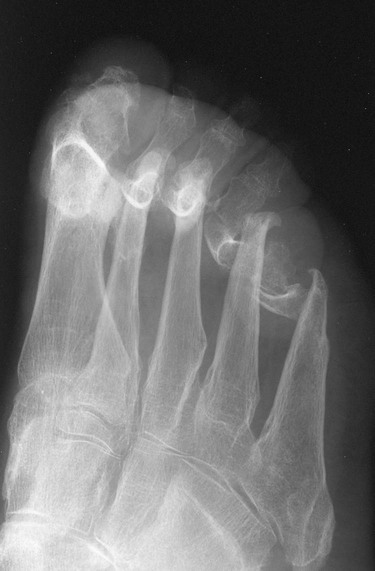
The earliest clinical and radiographic changes are characteristically found in the hands and feet.686 Although traditional evaluation methods of RA focus on radiographs of the hands, the feet appear to show osseous erosions first and to a greater extent.87,301,310,537,685 The initial involvement is of the head of the fifth metatarsal, with erosion and narrowing of the fifth metatarsophalangeal (MTP) joint.
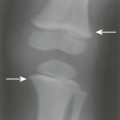
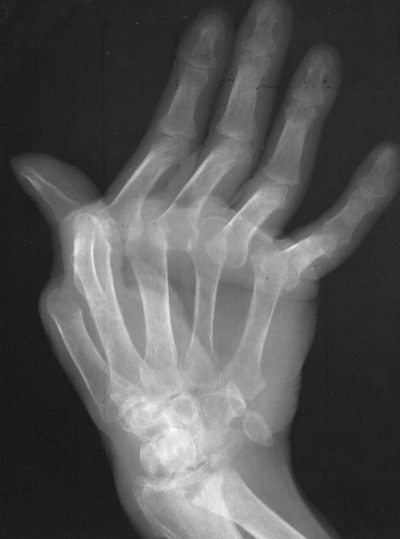
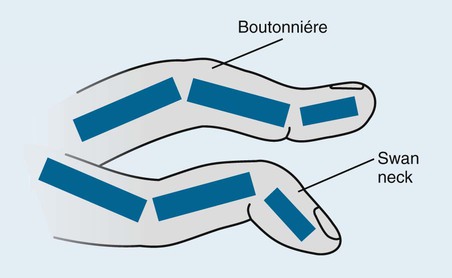
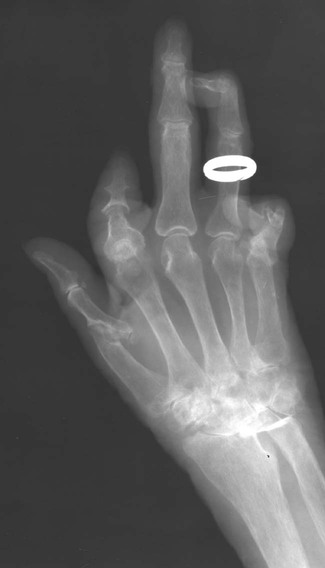
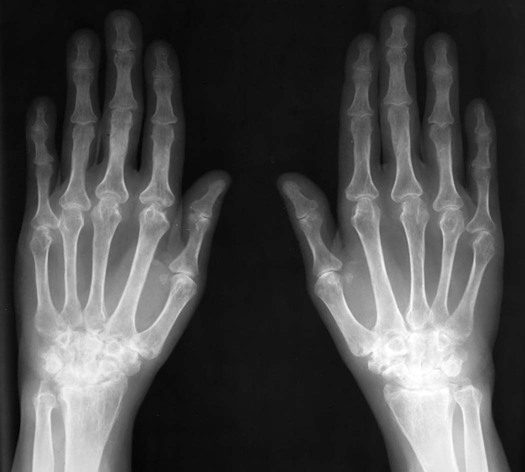
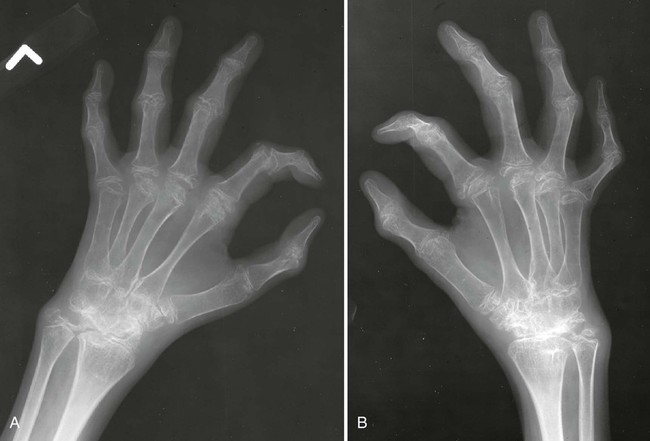
The classic joint distribution of RA in the hands is bilateral and symmetric involvement of the metacarpophalangeal (MCP), proximal interphalangeal (PIP), and thumb interphalangeal (IP) articulations. Some or all of the general radiographic features may be seen. Initially, the joint spaces may appear widened secondary to effusion, however as the cartilage is destroyed, joint space narrowing occurs. The earliest osseous erosions in the hands occur at the MCP joints of the second and third digits of the dominant hand,383 and at the radial aspect of the metacarpal heads (Figs. 9-3 and 9-4). The Norgaard and Brewerton views may aid in visualization of these early joint erosions, although it is difficult to duplicate the exact positioning on serial radiography.686 In the Norgaard view, the hands are 45 degrees supine, with the fingers straight,487 and in the Brewerton view (ball catcher’s view), the MCP joints are flexed at 65 degrees.82 Characteristic joint deformities include the swan neck deformity, boutonnière deformity, ulnar deviation of the fingers (doigts en coup de vent), and the hitchhiker’s or Z-shaped deformity of thumb (Figs. 9-5 to 9-7). The swan neck deformity results from hyperextension of the PIP and hyperflexion of the distal interphalangeal (DIP) joints, whereas the boutonnière deformity represents the opposite configuration of hyperflexion of the PIP joint and hyperextension of the DIP joint. The hitchhiker’s thumb is secondary to MCP flexion and IP extension (Fig. 9-5). Ulnar deviation at the MCP joints is called ulnar drift; when combined with radial deviation in the radiocarpal articulations it results in a “zigzag” deformity (Fig. 9-5). Loosening or disruption of the distal attachment of the extensor tendon to the terminal phalanx leads to a “mallet” finger.
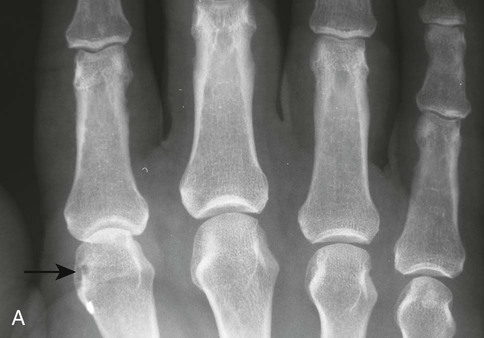
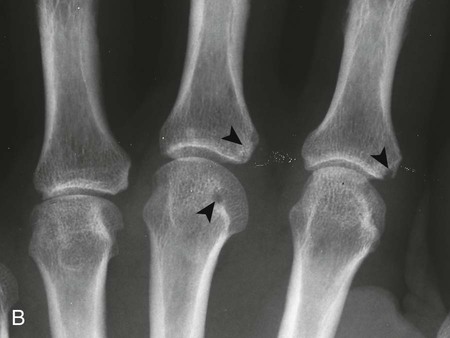
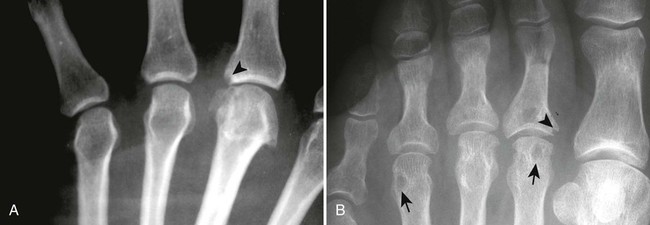
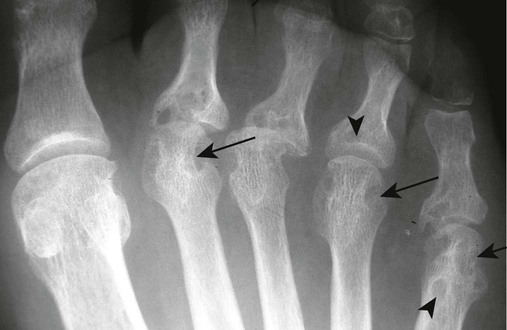
Changes in the feet tend to parallel those of the hands, with the MTP articulations commonly affected first and other deformities, including lateral or fibular deviation at the MTP joints of the first through fourth digits, flexion of the DIP joints (hammer or claw toes), and extension of the MTP joints eventually developing (Figs. 9-8 to 9-11). Pathologic changes in the deep transverse tendons may lead to spreading of the metatarsals or forefoot. Pathologic changes in supporting ligaments also may lead to hallux valgus. Retrocalcaneal bursitis, plantar fasciitis, Achilles tendonitis, or Achilles tendon rupture may occur in the heel.
Wrist.
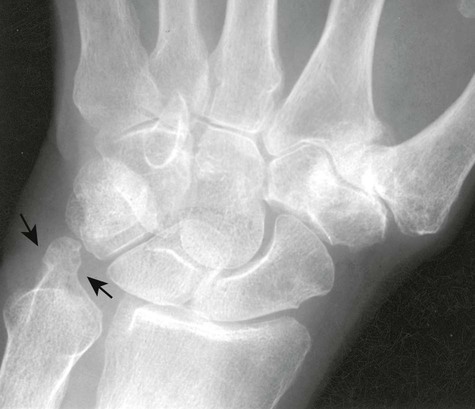
One of the earliest findings of RA may involve tendonitis of the wrist, as suggested in a study by McQueen et al,502 which found evidence on MRI that 81% of patients with early RA demonstrated tendinopathy at baseline. Pancompartmental involvement is typical in the wrist, with the earliest erosions usually involving the radial and ulnar styloid processes; distal radioulnar and radiocarpal joints; and waist of the scaphoid, triquetrum, and pisiform (Figs. 9-12 to 9-15).425 Ligamentous instability results in patterns that parallel posttraumatic lesions, such as scapholunate dissociation, distal radioulnar dissociation, and dorsiflexion or volar flexion instability (Fig. 9-15). Eventual carpal ankylosis is common.
Elbows.
Joint effusion is recognized by a positive fat pad sign. Osseous erosions may be noted at each of the articulating surfaces, although the main destruction usually affects the humeral–ulnar joint. Antecubital cysts may occur in which the synovium herniates through the weakened anterior capsule of the joint.
Shoulders.
The glenohumeral and acromioclavicular (AC) joints may be affected. Resorption of the distal clavicle, in addition to erosions at the coracoclavicular ligament insertion on the undersurface of the clavicle, is not uncommon. Erosions may be observed on the medial aspect of the humeral neck as it abuts the glenoid process from elevation of the humerus, which is caused by a rotator cuff tear secondary to a chronically inflamed supraspinatus tendon. The acromiohumeral distance narrows with progressive elevation of the humeral head, and sclerosis and cyst formation are noted on the adjacent portions of the humeral head and acromion process.
Hips.
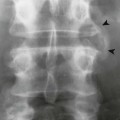
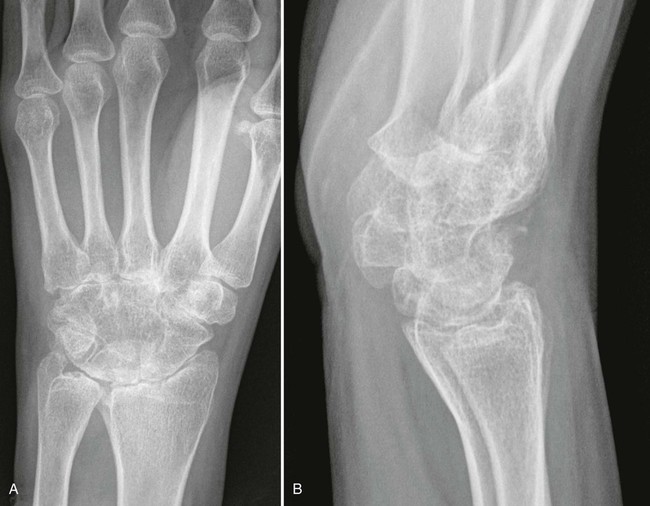
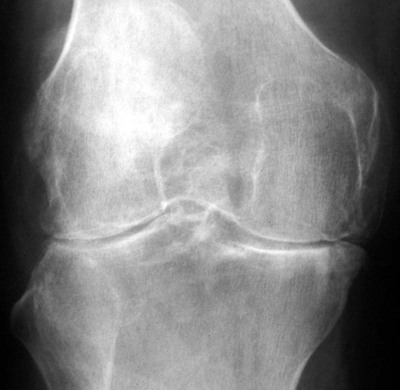
Abnormalities of the hips generally are bilateral and symmetric. Axial migration of the femoral head (concentric joint space narrowing) and bilateral acetabular protrusion are common findings that accompany the other general features of erosions, subchondral cysts, and osteopenia associated with RA (Fig. 9-16).155 There is an absence of sclerosis and osteophyte formation unless secondary OA has developed.
Knees.
Tricompartmental involvement that includes the general features of joint space narrowing and osseous erosions is typical (Figs. 9-17 to 9-19). A genu valgus deformity is more likely to develop than a varus joint deformity (Fig. 9-20). Soft-tissue swelling may be present in the form of suprapatellar effusion or a large popliteal (or Baker) cyst. Subchondral sclerosis and osteophytes may be noted with the development of secondary OA.
Cervical Spine.
RA involvement is rare in other regions of the axial skeleton but affects the cervical spine in more than half the patients within the first 10 years of disease onset,557,686 with a preference for the apophyseal and atlantoaxial joints. The more chronic the disease, the greater is the likelihood of cervical involvement.554 Cervical spine progression appears to parallel clinically and radiologically with peripheral joint involvement. The greater the structural or erosive damage in the peripheral joints, the greater the tendency for severe structural damage in the cervical spine. Radiographs of the cervical spine are a prudent consideration in all patients with RA regardless of symptoms, because many patients are asymptomatic despite cervical involvement.135 These should include lateral views with the patient in flexion and extension to assess level of involvement and evidence of instability.
Atlantoaxial subluxation or instability is the most common radiographic abnormality encountered in the cervical spine, with the prevalence varying from 19% to 80% depending on the patient selection and radiographic examination (Fig. 9-21).334 Movement may occur in several directions (listed in order of decreasing frequency): anterior (most frequent, 9.5% to 36%), lateral, vertical, or posterior.56,115 Movement is usually the result of odontoid erosions or transverse ligament laxity, although the alar and apical ligaments also may be involved.
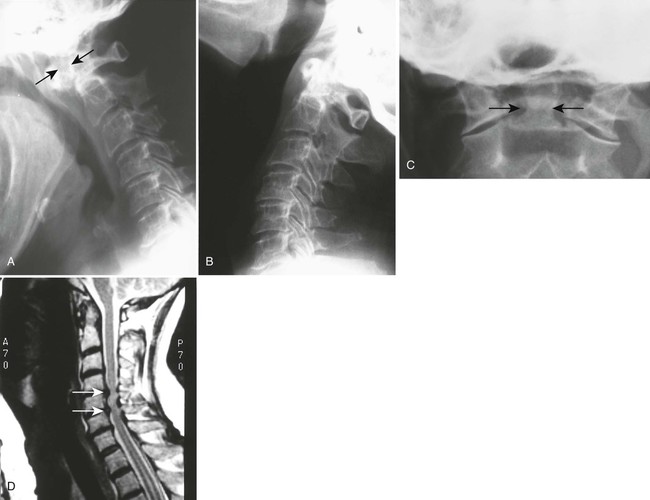
The degree of anterior subluxation shown on conventional radiography correlates poorly with neurologic signs and the presence of cord compression, which is in part a result of the unknown thickness of the synovial pannus not visible on plain films.56,566 The standard method for determining anterior atlantoaxial subluxation has been an anterior atlantodental interval* (ADI) of greater than 3 mm. An ADI of 3 to 6 mm is indicative of early instability with damage of the transverse ligament. An ADI greater than 6 mm indicates the addition of alar ligament damage. Surgical stabilization is likely necessary with an ADI greater than 9 mm.
It has been shown, however, that a posterior atlantodental interval* (PADI) of 14 mm or less may be a more reliable predictor than utilizing the ADI for neural compression, and necessitates an MRI for evaluation of true cord space (Fig. 9-22).56 It has also been shown that neurologic recovery from surgery was likely if the surgery was performed with a PADI of greater than 14 mm, but unlikely if the PADI was less than 10 mm.
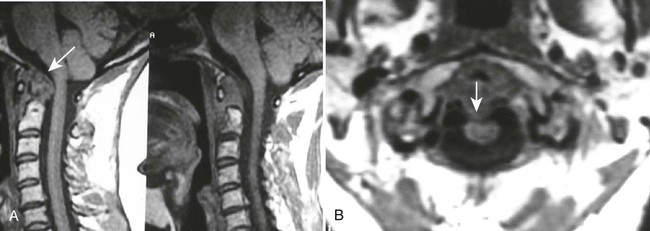
Vertical atlantoaxial subluxation, also known as cranial settling, atlantoaxial impaction (AAI), and pseudobasilar invagination, is superior migration of the odontoid, and occurs with erosion of the occiput-C1 and C1-2 articulations. It results in approximation of the dens and brainstem, which can lead to direct compression or cause neurologic damage by excessive kyphosis.97,566 The McGregor line may be used to assess superior migration, in which vertical subluxation is designated when the odontoid tip lies more than 4.5 mm above the line; however, this is not always accurate as there may be odontoid erosion. The Ranawat index may also be used to evaluate vertical subluxation on the lateral cervical radiograph, in which a measurement is made from the center of the C2 pedicles to a line connecting the midpoint of the anterior and posterior arches of C1. Normal values are greater than 15 mm for men and greater than 13 mm for women. The Sakaguchi-Kauppi method may be used, which was also developed for screening purposes and evaluating the position of C1 in relation to C2. This method divides AAI into four grades based on the relationship of the cranial tip of the C2 pedicle shadows, as visualized on the lateral cervical radiograph, to the line drawn between the caudal parts of the anterior and posterior arches of atlas.335
Subaxial subluxations may occur in 10% to 20% of patients and often are at multiple levels. C3-4 and C4-5 are the most common areas of involvement, producing a “stepladder” or “doorstep” deformity and associated kyphosis on the lateral radiograph.504 Although these are less common than upper cervical spine subluxations, they are potentially more important neurologically because of the smaller spinal canal dimensions.56,115 A canal measurement may be taken from the lateral radiograph in a similar way that the PADI is taken. The normal sagittal diameter from C3 to C7 is 14 to 23 mm. If the subaxial canal diameter measures less than 14 mm, MRI is advised.56 Another method of measuring subaxial subluxations is the cervical height index which evaluates the impact of the combination of changes that may occur throughout the cervical spine (reduced disc height, bony collapse, and subluxations). On the lateral projection, the distance from the center of the sclerotic ring formed from the C2 pedicle to the midpoint of the inferior C7 endplate is measured. This is divided by the measurement taken from the center of the C2 pedicle shadow to the tip of the C2 spinous process. A cervical height index less than 2 mm is specific and sensitive for predicting neurologic compromise.
Disc height narrowing, mild subchondral sclerosis, and erosions of the vertebral endplates, facet joints, and spinous processes are other less-severe signs of rheumatoid involvement that occur in the cervical spine, and much less commonly the thoracic and lumbar spine. These signs often are subtle and difficult to diagnose on plain film radiographs. Patients treated with corticosteroids are susceptible to avascular necrosis which may affect segments of the thoracic and lumbar spine. Vertebral body collapse, fragmentation, and radiolucent fracture lines may be visible.
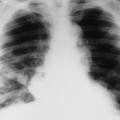
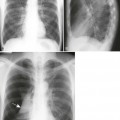
Lungs.
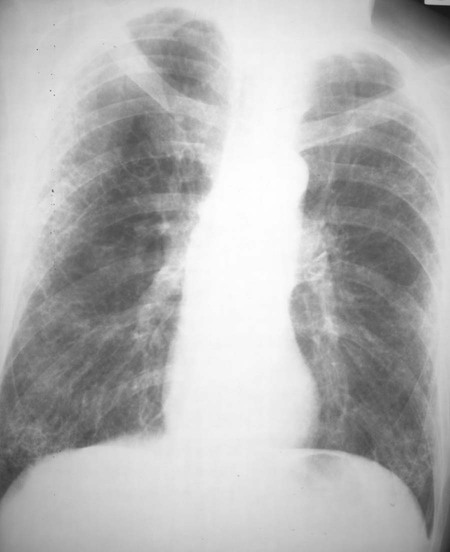
Lung involvement is common, although not always clinically significant. Pleural involvement (e.g., pleurisy, pleural effusion) is the most common lung manifestation and usually is asymptomatic.14 Other pulmonary manifestations include pulmonary fibrosis or honeycombing, constrictive bronchiolitis, bronchiectasis, pulmonary nodules, and subpleural micronodules (Fig. 9-23). High-resolution computed tomography (HRCT) is useful for this avenue of investigation. These findings have been observed on CT in patients with symptoms, and those asymptomatic patients with no abnormalities noted on chest plain film.570 Caplan syndrome describes inflammation and scarring of pulmonary tissue in people with RA who have exposure to coal dust.
Magnetic Resonance Imaging.
As previously noted, MRI is employed in RA patients when severe pain exists or when neurologic compromise is suspected as it is the favored modality for evaluation of the spinal cord and neural elements.481 The primary indication for MRI of the cervical spine are abnormal measurements on plain film,586 and/or signs and symptoms of spinal cord, brainstem, or vertebral artery compression. The presence of pannus and its relationship to the spinal cord can be assessed, as can the relationship of the odontoid to the medulla and brainstem. Edematous changes within the spinal cord can also be recognized.
Cervical spine MRI can be performed with the patient in a neutral position, or with the cervical spine in a flexed or extended position. Functional MRI, obtained with flexion of the cervical spine, has been recommended when neutral position MRI findings are normal, and as a preoperative evaluation.585,711,712
Clinical Comments.
In 2010, in an effort to establish more sensitive parameters for earlier RA recognition and treatment so as to prevent or reduce disease progression and complications, a new classification criteria for RA was established by the American College of Rheumatology (ACR) and European League Against Rheumatism (EULAR).8 Prior to the 2010 criteria, the 1987 ACR criteria were the standard for diagnosis and study of RA. Early diagnosis can be difficult as the symptoms early in the disease process are relatively nonspecific, and may consist mainly of fatigue, muscle soreness, weakness, and joint pain. Comparison between the 2010 and 1987 criteria denote that the new criteria are better at identifying those patients with early RA.8,483
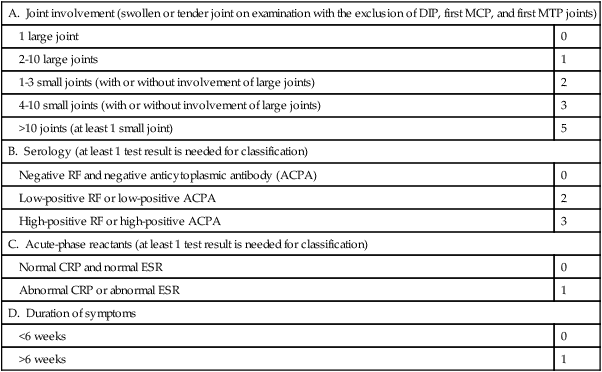
The 2010 ACR/EULAR Classification Criteria for RA
Target population (Who should be tested?): Patients who
1. have at least 1 joint with definite clinical synovitis (swelling),
2. with the synovitis not better explained by another disease (i.e., systemic lupus erythematosus [SLE], psoriatic arthritis, gout).
Classification criteria for RA (score-based algorithm: add score of categories A to D; a score of ≥6/10 is needed for classification of a patient as having definite RA):
| A. Joint involvement (swollen or tender joint on examination with the exclusion of DIP, first MCP, and first MTP joints) | |
| 1 large joint | 0 |
| 2-10 large joints | 1 |
| 1-3 small joints (with or without involvement of large joints) | 2 |
| 4-10 small joints (with or without involvement of large joints) | 3 |
| >10 joints (at least 1 small joint) | 5 |
| B. Serology (at least 1 test result is needed for classification) | |
| Negative RF and negative anticytoplasmic antibody (ACPA) | 0 |
| Low-positive RF or low-positive ACPA | 2 |
| High-positive RF or high-positive ACPA | 3 |
| C. Acute-phase reactants (at least 1 test result is needed for classification) | |
| Normal CRP and normal ESR | 0 |
| Abnormal CRP or abnormal ESR | 1 |
| D. Duration of symptoms | |
| <6 weeks | 0 |
| >6 weeks | 1 |
While the main sites affected by RA are the joints, extraarticular manifestations are common. Rheumatoid nodules are the classic extraarticular lesions. These subcutaneous lesions are found in approximately 25% to 40% of patients and are typically located on the extensor surfaces at sites subject to trauma.434,645 These nodules also can be present in visceral organs. Pulmonary and cardiovascular systems often are affected in patients with RA. In fact, approximately 40% of all deaths in individuals with RA are ascribed to cardiovascular causes, including ischemic heart disease and stroke.658 Two syndromes associated with RA are Felty (a combination of RA, splenomegaly, and neutropenia) and Sjögren (marked by RA and dry eyes and mouth). A laboratory clue to Felty syndrome is decreased white blood cells, which occurs with splenomegaly.
Patients with RA tend to have reduced life expectancies and decreases in their ability to perform activities of daily living and work.228,408,476,535,658 The disease course is variable, ranging from mild with periods of remission to severe and quickly progressive. The highest rate of joint damage and progression occurs early in the disease; therefore treatment should begin immediately after the diagnosis is made so that irreversible joint damage is prevented.686 Known risk factors may increase the possibility of developing the severe stages of the disease and early mortality, so these patients may need a more aggressive treatment.534,535,693,696
Laboratory Tests.
Laboratory tests aid in establishing the diagnosis and assessing disease activity. The test for the presence of RF is positive in approximately 70% to 80% of patients with RA (95% if presenting with subcutaneous nodules), but also is positive in approximately 5% of individuals who do not have RA, and may be present in patients with other nonrheumatoid diseases.394,411 A more recent test, measures antibodies to cyclic citrullinated peptide (CCP). Like RF, a positive CCP not only is associated with RA, but with severe RA.589 It has been suggested, because of the much higher cost of anti-CCP as compared to RF testing, that this testing be utilized only in patients with arthritis who have a negative or low-titer RF (<50 UI/mL).482 A complete blood count, ESR, CRP, and ANA assay also may be performed to initially evaluate and as follow-up for rheumatoid patients.721
Treatment and Management.
RA is one of the most costly musculoskeletal disorders because of loss of work and limitations in daily activities caused by pain and functional disability,7,11,213,730 and because of the associated treatment cost. More aggressive treatment is being used at the initial onset of RA to combat unfavorable long-term outcomes.684,691
Previously, pharmacologic treatment began with nonsteroidal antiinflammatory drugs (NSAIDs) or corticosteroids, progressed to more aggressive nonbiologic disease-modifying antirheumatic drugs (DMARDs), and later for the minority of patients who were unresponsive, to the use of biologic DMARDs.65,645 Currently, to reduce disease activity and prevent joint deformity, a more aggressive approach is being utilized. Nonbiologic DMARDs are being prescribed within 3 months of diagnosis, and there are a variety of biologic DMARDs available. In 2008, the ACR updated RA medical management guidelines to detail which biologic DMARDs are indicated based on RA disease profiles such as disease activity, signs and symptoms, and prognosis.596 In addition, studies have shown the combination treatment of glucocorticoids with DMARDs to be highly effective.640
Treatment is multifaceted and geared toward reducing pain and limiting or slowing the progression of deformities and associated disability. The 2008 guidelines recommend the inclusion of conservative therapy as well pharmaceuticals. Conservative therapy should include patient education (inclusive of pain coping techniques) and emotional support340,393,678; rest; application of heat or cold; dietary changes or supplementation; and resistance training, exercise, and joint mobilization to improve joint range of motion, strengthen muscles, and minimize joint deformity.2,166,174,274,558,716
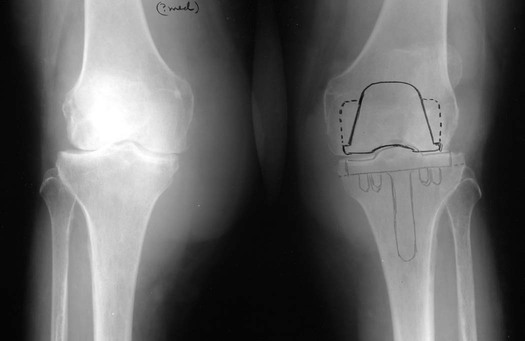
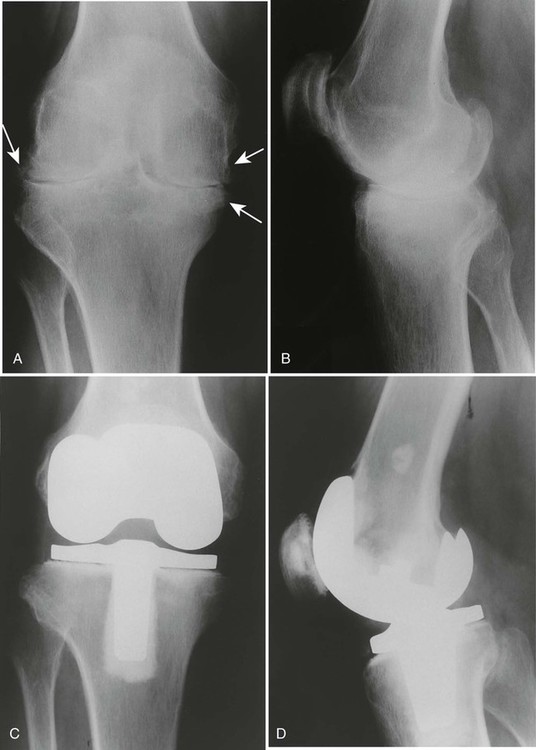
At the disease onset, radiographs of the hands, wrists, and feet should be obtained as a baseline and then repeated at 6-month intervals for a minimum of 2 years to aid in identifying those patients at risk for serious joint damage and deformity.88,518 MRI of the spine should be performed on patients with radiographic abnormalities combined with neurologic deficits, and should be considered in patients with superior migration of the dens, a PADI of less than 14 mm, and a subaxial canal diameter of less than 14 mm regardless of symptoms. MRI with contrast (gadolinium) should be used to differentiate synovial fluid from pannus. Surgical intervention may be indicated to replace joints (Fig. 9-24), correct severe deformity,254 or treat neurologic compromise.230
Juvenile Idiopathic Arthritis
Background
Three sets of criteria exist to classify idiopathic childhood arthritis. The classification system most recently established and internationally accepted utilizes the term juvenile idiopathic arthritis (JIA) and was developed by the International League of Associations for Rheumatology (ILAR). This system unifies and expands upon the previous classifications of juvenile rheumatoid arthritis (JRA) and juvenile chronic arthritis (JCA) established by the ACR and the EULAR, respectively.111,112,531,672,727
Classification according to the ILAR: JIA
1. Systemic arthritis
2. Oligoarthritis
3. Polyarthritis (RF negative)
4. Polyarthritis (RF positive)
5. Enthesitis-related arthritis
6. Psoriatic arthritis
7. Other or undifferentiated
Classification according to the ACR: JRA
Classification according to the EULAR: JCA
1. Juvenile-onset adult type (seropositive)
2. Seronegative chronic arthritis (Still disease)
3. Juvenile spondyloarthropathy (ankylosing spondylitis, psoriatic arthritis, enteropathic arthritis)
The terms JIA, JRA, and JCA all indicate an inflammatory disease of unknown cause, occurring in childhood (<16 years of age), characterized primarily by arthritis (joint inflammation and stiffness) persisting for a minimum of 6 weeks (JCA symptoms must last at least 3 months).
JIA is the most common type of arthritis affecting children.717 It is an autoimmune disorder with an unknown etiology. Incidence and prevalence rates can be wide ranging, likely because of differing definitions and criteria. JIA has a prevalence in the United States ranging from 1.6 to 86.1 per 100,000 children, and an incidence rate of 4 to 14 per 100,000 children.727 Internationally, the prevalence appears to range between 20 and 148 cases per 100,000 children,528,679 and an incidence rate with a range of 3.5 to 22 per 100,000 children.327,528 JIA targets predominantly the synovial joints, in which synovial proliferation leads to joint and soft-tissue destruction. Genetic susceptibility appears to predispose a patient to the disease, and an infectious or environmental factor may trigger its onset. The IL2RA/CD25 and the VTCN1 genes have been implicated as JIA susceptibility loci,287 and an association between specific HLA alleles and clinical subsets of JIA has been noted.728
The first four subsets of the JIA classification encompass the majority of cases and are essentially the same subsets listed by the ACR as representative of JRA. These subsets are divided according to the presence or absence of RF, systemic symptoms within the first 6 months of onset, and the number of joints involved within the first 6 months of onset.
1. Systemic arthritis is the most serious, and least common subtype, comprising approximately 5% of all JIA patients. It occurs in children younger than 5 years of age; affects males and females equally; and presents with spiking fevers of at least 2 weeks, a characteristic evanescent salmon-colored rash on the trunk and thighs, hepatosplenomegaly, serositis, adenopathy, leukocytosis, and mild polyarthritis, usually of the wrist, knees, and ankles. These children typically have negative RF and ANA serology. Approximately 50% of cases show very short stature in adulthood as a result. Macrophage activation syndrome is a rare but serious complication of systemic JIA, the hallmarks of which include hypofibrinogenemia, thrombocytopenia, and elevated aspartate aminotransferase.
2. Oligoarthritis is the most common variety, comprising approximately 30% of JIA patients, and is female predominant (3 : 1). It is found in children younger than 5 years of age, and affects no more than four joints during the first 6 months, generally affecting large joints (e.g., knees, ankles, elbows, wrists). This form frequently is accompanied by asymptomatic chronic iridocyclitis (15% to 20%), uveitis, cataracts, and (in a minority) blindness. There is a large association with ANAs. Two additional subcategories are recognized: persistent oligoarthritis and extended oligoarthritis. Persistent oligoarthritis affects no more than four joints throughout the course of the disease. Extended oligoarthritis affects a cumulative total of five or more joints after the first 6 months of onset.
3. Polyarthritis (RF negative,) involves more than five joints during the first 6 months of the disease, and has a biphasic peak of onset. The first peak is from ages 1 to 4 years, and the second occurs at ages 6 to 12 years. It is more prevalent among females, and comprises approximately 20% of JIA patients. Symmetric small joint involvement of the hands and feet is common, which is similar in distribution to adult RA.
4. Polyarthritis (RF positive,) develops in teenage girls and is similar clinically and radiographically to adult RA.17,682 Juvenile patients may present with subcutaneous nodules, although this symptom occurs less frequently in juveniles than adults. More than five joints are affected, most commonly the hands, wrists, feet, knees, hips, and cervical spine. Polyarticular RF positive comprises approximately 5% of JIA patients.
5. Enthesitis-related arthritis usually occurs in males between 10 and 12 years of age, and indicates the presence of both arthritis and enthesitis, or only arthritis or enthesitis with at least two of the following: SI tenderness or inflammatory spinal pain, presence of HLA-B27, onset of arthritis in a male age 6 years or older, acute asymptomatic anterior uveitis, or family history of confirmed HLA-B27 in at least one first- or second-degree relative. It comprises approximately 25% of JIA patients. Pain and tenderness at the enthesis is the most common presentation. This subset frequently exhibits low back pain in the evening, or after periods of inactivity. Symptoms improve with exercise.
6. Psoriatic JIA indicates arthritis and psoriasis, or arthritis and at least two of the following: dactylitis, nail abnormalities (pitting or onycholysis), or family history of psoriasis in at least one first-degree relative. It encompasses approximately 5% of JIA patients.
7. Undifferentiated arthritis is estimated to make up 10% of JIA patients, and indicates childhood arthritis of unknown cause persisting for at least 6 weeks that does not fulfill criteria for subsets 1 to 6, or fulfills criteria for more than one of the other subsets.
Imaging Findings
Although other imaging modalities may be more sensitive for early detection of joint pathology, plain film radiography continues to be the primary method of imaging for the diagnosis and follow-up evaluation of JIA.323,523 Early plain film radiographs may be negative as plain film radiography does not allow for direct examination of the articular cartilage and synovium, and other imaging modalities, such as technetium bone scan, ultrasonography, CT, or MRI, may be indicated for confirming JIA or eliminating a possible differential diagnosis.
In patients with suspected JIA, bone scintigraphy is useful in determining the joint distribution of the disorder, or when physical findings do not document definite arthritis. The major disadvantage, especially in children, is the high radiation dose. Ultrasonography may reveal inflamed synovium as an area of mixed echogenicity lining the articular cartilage, assess the vascularity of the synovium with Doppler flow studies, and compare serial measurements of synovial thickness and effusion volumes to monitor disease progression.622 However, sonography can be very user dependent. Computerized tomography is still the best modality for visualizing bony abnormalities or complex bony anatomy; however, given the radiation dose, MRI is predominantly used in place of CT. MRI can reveal synovial hypertrophy, soft tissue swelling, and provide outstanding detail of the articular cartilage. Contrast-enhanced sequences are recommended for improved visualization of synovial hypertrophy and cartilaginous erosions. In addition, synovitis and joint effusion may have similar hyperintensity on T2-weighted and short-tau inversion recovery (STIR) images, and contrast enhanced T1-weighted images will aid in indicating active synovitis.
The radiographic presentation in the majority of JIA cases is similar in appearance to adult RA with few exceptions (Table 9-2). Although features within each subset of JIA can differ, there are features that may be common to all, including early manifestations such as fusiform periarticular soft-tissue swelling and juxtaarticular osteopenia (with possible growth recovery lines) that may become diffuse. Intermediate or later-stage manifestations can include joint space narrowing, osseous erosions, growth disturbances, bony ankylosis, joint contractures, subluxation, or dislocation.
TABLE 9-2
RADIOGRAPHIC FINDINGS ASSOCIATED WITH JUVENILE AND ADULT RHEUMATOID ARTHRITIS
| Radiographic Finding | Juvenile Rheumatoid | Adult Rheumatoid |
| Joint space narrowing | Common in late stages of disease | Common in early stages of disease |
| Marginal bony erosion | Common in late stages of disease | Common in early stages of disease |
| Intraarticular fusion | Common | Uncommon |
| Growth abnormalities | Common | Absent |
| Periostitis | Common | Absent |
Distinct features of JIA include periostitis and growth abnormalities, which are the result of the disease process combined with skeletal immaturity. Periostitis commonly involves the diaphysis of the metacarpals, metatarsals, and proximal phalanges and may be an early and prominent finding that is likely explained by the exposure of loosely attached periosteum to an inflammatory and hyperemic process. Growth abnormalities result from hyperemia to the epiphysis and growth plates, causing an overgrown “ballooned” or “squared” epiphysis. Hyperemia also may lead to premature growth plate fusion, resulting in shortening of limbs or limb length discrepancies.
Overall joint involvement mimics that of adult RA in the majority of cases, although JIA has more of a predilection for large joints. Individual joint manifestations are noted in the following explanations.
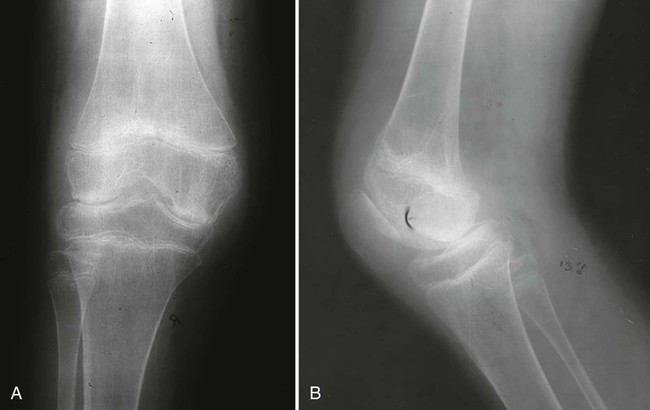
Knees.
The knee is the most commonly affected joint in JIA. Effusion; joint space narrowing; osteopenia and enlargement, or “ballooning” of the metaphysis and epiphysis of the distal femur and proximal tibia; widening or expansion of the intercondylar notch; and patellar squaring are the most distinctive findings associated with JIA in the knee (Fig. 9-25). Growth arrest lines may result from temporary reduction in growth velocity, and frequently are seen around the knee. A discrepancy in leg lengths may occur with knee inflammation. If the inflammation is controlled and occurs before the age of 9 years, the leg lengths equalize and no residual discrepancy remains. If involvement exists after the age of 9 years, early epiphyseal closure may occur and result in a permanently shorter leg on the involved side.635
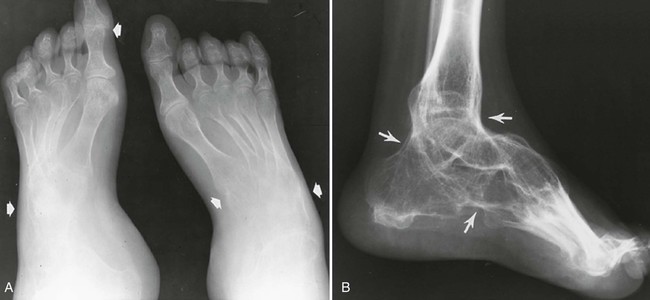
Ankles And Feet.
The expected changes of joint space narrowing and osseous erosions may be noted throughout the articulations of the ankles and feet. Tarsal ankylosis, epiphyseal enlargement, periostitis, and brachydactyly may occur. Swelling may be noted on the dorsum of the foot because of involvement of the subtalar and tibiotarsal joints (Fig. 9-26). Tibiotalar slant, valgus, and varus deformities may result.569
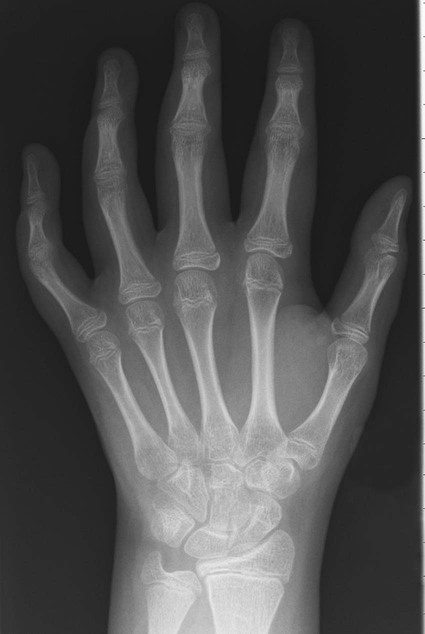
Hands and Wrist.
Early findings include soft-tissue swelling and juxtaarticular osteoporosis of the carpals, MCP joints, and PIP joints; preservation of joint space; lack of erosions; and periosteal new bone formation, resulting in a widened midportion of the phalanges (Figs. 9-27 to 9-29). Accelerated skeletal maturation; osseous erosions in the carpus, distal radius, and ulna; joint space narrowing; carpal and carpometacarpal ankylosis; and boutonnière and swan neck deformities (see description in previous section of adult RA) may present in later stages. Growth defects may be seen in the ulna and the fourth and fifth metacarpal bones (positive metacarpal sign).
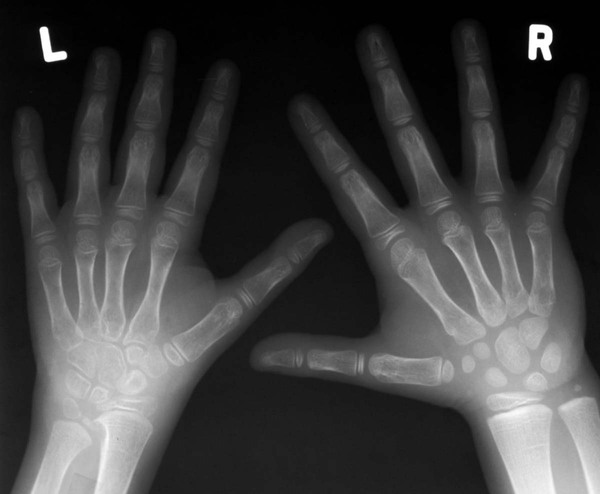
Hips.
JIA may be indicated by lack of growth of the iliac bone, femoral head flattening and enlargement, early growth plate closure, coxa valga deformity, and acetabular protrusion. Osteonecrosis also may develop as a complication of the disease but more often is related to treatment (corticosteroids).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree